Explore the Mechanism of Shanxi Aged Vinegar on High-fat Diet-Induced Antioxidant in Rats Based on PI3K/AKT1 Pathway
-
摘要: 目的:基于PI3K/AKT1通路探究山西老陈醋对高脂饮食诱导大鼠抗氧化作用机制。方法:30只SPF级雄性Wistar大鼠随机分为5组:正常组(C,常规饲料)、高脂模型组(M,高脂乳剂)、低(LV)、中(MV)、高(HV)剂量山西老陈醋干预组(1.35、2.7、5.4 g/(kg·bw))。造模(5周)同时给予陈醋干预后,山西老陈醋继续干预5周。检测血清血脂含量、肝损伤指标及抗氧化酶活性;HE染色观察肝组织病理学状态;qRT-PCR和Western blotting检测大鼠肝脏中PIK3CA、AKT1 mRNA及蛋白水平表达,免疫组织化学技术进行定位分析。结果:与C组相比,M组肝脂肪变性严重,血清中脂质含量和肝损伤程度极显著升高(P<0.01),抗氧化酶活性极显著下降(P<0.01),肝组织中PIK3CA和AKT1 mRNA及蛋白水平表达极显著升高(P<0.01),高脂血症大鼠造模成功,同时PI3K/AKT1通路被激活。血清学检测结果显示,山西老陈醋可显著下调血清中脂质含量、改善肝损伤、增加抗氧化酶活性(P<0.05),使其肝组织病理损伤减轻,且HV和MV组改善效果优于LV组。qRT-PCR、Western blotting及免疫组织化学技术检测结果显示,PIK3CA、AKT1主要定位于肝细胞质及细胞核中,山西老陈醋可极显著下调PI3K/AKT1通路中PIK3CA、AKT1 mRNA及蛋白水平表达(P<0.01),HV和MV组干预效果优于LV组。结论:不同剂量山西老陈醋可不同程度改善高脂饮食大鼠脂质、抗氧化及肝功能指标,最佳剂量可能在2.7~5.4 g/(kg·bw)之间,山西老陈醋可能通过调控PI3K/AKT1通路发挥抗氧化应激损伤作用,进而改善高脂饮食诱发的大鼠高脂血症。Abstract: Objective: To explore the antioxidant mechanism of Shanxi aged vinegar on rats induced by high fat diet based on PI3K/AKT1 pathway. Methods: Thirty SPF male Wistar rats were randomly divided into 5 groups: Normal group (C, conventional diet), high fat model group (M, high fat emulsion), low (LV), medium (MV) and high (HV) Shanxi aged vinegar intervention groups (1.35, 2.7, 5.4 g/(kg·bw)). Modeling (5 weeks) at the same time given Shanxi aged vinegar intervention, the aged vinegar intervention continued for 5 weeks. Serum lipid content, liver injury index and antioxidant enzyme activity were detected. The histopathological status of liver was observed by HE staining. The mRNA and protein levels of PIK3CA and AKT1 in rat liver were detected by qRT-PCR and Western blotting, and the localization was analyzed by immunohistochemistry. Results: Compared with group C, liver steatosis was severe in group M, serum lipid content and degree of liver injury were significantly increased (P<0.01), antioxidant enzyme activity was significantly decreased (P<0.01), mRNA and protein expressions of PIK3CA and AKT1 in liver tissue were significantly increased (P<0.01). The hyperlipidemia rats were successfully modeled and PI3K/AKT1 pathway was activated. The results of Serological showed that Shanxi aged vinegar could significantly down-regulate serum lipid content, improve liver injury, increase antioxidant enzyme activity (P<0.05), and reduce pathological injury of liver tissue, and the improvement effect of HV and MV groups was better than that of LV group. The results of qRT-PCR, Western blotting and immunohistochemical detection showed that PIK3CA and AKT1 were mainly located in the cytoplasm and nucleus of liver cells. Shanxi aged vinegar could significantly down-regulate the mRNA and protein expressions of PIK3CA and AKT1 in PI3K/AKT1 pathway (P<0.01), and the intervention effect of HV and MV groups was better than that of LV group. Conclusion: Different doses of Shanxi aged vinegar can improve lipid, antioxidant and liver function indexes of rats with high fat diet to different degrees, and the best dose may be between 2.7~5.4 g/(kg·bw). Shanxi aged vinegar may play a role in anti-oxidative stress injury by regulating PI3K/AKT1 pathway, so as to improve hyperlipidemia induced by high fat diet in rats.
-
Keywords:
- Shanxi aged vinegar /
- PI3K/AKT1 /
- high fat diet /
- rat /
- antioxidant
-
高脂血症通常由长期高脂饮食所致,表现为外周血中脂质水平异常[1],肝脏脂代谢紊乱,肝细胞变性[2],可引发一系列心血管疾病[3]。山西老陈醋是中国传统的全谷物固态发酵醋,发酵过程中形成了多酚、类黑素、益生菌等多种具有降脂抗氧化功效的有机物[4]。大量研究发现,山西老陈醋浓缩物可有效降低高脂血症小鼠的血脂水平[5],陈醋源乳杆菌通过改变肝脏脂肪酸的构成,改善高脂饮食大鼠肝脂质损伤[6]。进一步研究发现,山西老陈醋多酚提取物能有效减轻高脂血症大鼠的血脂异常和氧化应激[7]。截止目前,山西老陈醋改善高脂血症的作用机制及最适剂量范围尚不明确。
调节糖脂代谢相关通路是最常见的高脂血症防治手段[8]。研究表明脂代谢紊乱与磷脂酰肌醇3-激酶(Phosphatidylinositol 3-kinase,PI3K)异常表达密切相关[9]。PI3K是细胞代谢关键调节因子,PIK3CA是其催化亚基的一种,蛋白激酶B(Protein kinase B,Akt1)作为PIK3CA的下游靶标响应激活[10],PI3K/AKT1信号转导途径通过调节甘油三酯合成关键酶的表达影响脂代谢[11],抑制该通路可改善氧化应激损伤[12],研究证实,食醋加工后的姜黄精油通过调节PI3K/AKT1通路改善肝脏纤维化[13]。固态发酵醋多酚提取物通过抑制PI3K/AKT1通路来调节脂代谢,减轻细胞氧化应激[14]。鉴于此,推测山西老陈醋可能通过调控PI3K/AKT1通路相关基因表达,进而发挥预防高脂血症发生和发展的作用。
本研究通过构建高脂血症大鼠模型,并设置不同浓度山西老陈醋干预组,观察其对大鼠血清内脂质及肝脏相关生化指标的影响,研究山西老陈醋通过PI3K/AKT1通路对高脂饮食诱导大鼠发挥的降脂抗氧化作用,为后期进一步探究其调控机制提供一定的试验基础。
1. 材料与方法
1.1 材料与仪器
SPF级雄性Wistar大鼠 6周龄,体重相近(170±20 g),斯贝福生物技术有限公司,许可证号:SCXK(京)2019-0010,饲养于山西农业大学实验动物中心(SYXK(晋)2020-0003),试验动物使用方案严格执行山西农业大学试验动物伦理委员会对动物试验伦理的要求(IACUC审查号:SXAU-EAW-2020SD0201);山西老陈醋 山西紫林醋业股份有限公司;玉米胚芽油 山东西王食品有限公司;吐温-80(T8360)、胆固醇(C8280)、胆酸钠(C1291697)、丙基硫氧嘧啶(P7161)、总RNA提取试剂盒(R1200)、通用SP检测试剂盒(SP0041)、RIPA组织/细胞裂解液(R0020)、BCA蛋白浓度试剂盒(PC0020)、山羊抗兔二抗及山羊抗鼠二抗(SE134、SE132) Solarbio公司;甘油三酯(Triglyceride,TG,A110-1-1)、总胆固醇(Total cholesterol,TC,A111-1-1)、高密度脂蛋白(High-density lipoprotein cholesterol,HDL-C,A112-1-1)、谷丙转氨酶(Alanine aminotransferase,ALT,C009-2-1)、谷草转氨酶(Aspartate aminotransferase,AST,C010-2-1)、碱性磷酸酶(Alkaline phosphatase,AKP,A059-2)、谷胱甘肽还原酶(Glutathione reductase,GR,A062-1-1)、超氧化物歧化酶(Superoxide dismutase,SOD,A001-3)、总抗氧化能力(Total antioxidant capacity,T-AOC,A015-2-1)检测试剂盒 南京建成生物工程研究所;HiScript Ⅲ RT SuperMix for qPCR(R323) 诺唯赞生物;2×Realtime PCR Super mix试剂盒(MF013) 北京聚合美生物科技有限公司;抗PIK3CA兔多克隆抗体(bs-2067R)、抗AKT1兔多克隆抗体(bs-0115R)、抗GAPDH鼠单克隆抗体(bsm-33033M) 北京博奥森生物有限公司。
B11-3恒温磁力搅拌器 上海司乐仪器有限公司;ELX808全自动酶标仪 BioTek Instruments公司;UV-1200紫外可见分光光度计 上海美谱达仪器有限公司;NanoDrop One/OneC核酸蛋白检测仪 Thermo Fisher;T100普通PCR仪 美国Bio-Rad公司;LightCycler 96荧光定量PCR仪 Roche生物科技有限公司;DYY-8C电泳仪 北京六一生物科技有限公司;YD-6L生物组织包埋机 金华市益迪医疗设备有限公司;DM4000光学显微镜、RM2265轮转式切片机 徕卡显微系统(上海)有限公司。
1.2 实验方法
1.2.1 不同浓度山西老陈醋制备
山西老陈醋原醋为中浓度醋(2.7 g/(kg·bw));蒸馏水稀释至原体积2倍即低浓度醋(1.35 g/(kg·bw));恒温50 ℃下使用磁力搅拌器将原醋浓缩至原体积1/2即高浓度醋(5.4 g/(kg·bw))。
1.2.2 高脂饮食诱导大鼠模型构建
高脂乳剂由60%吐温-80,15%玉米胚芽油,6%胆固醇,2%胆酸钠,0.2%丙基硫氧嘧啶和16.8% H2O配制而成(图1),造模5周[15]。
1.2.3 试验动物分组及样品采集
选取30只6周龄健康状况良好SPF级大鼠,适应性饲喂1周,根据每组间平均体重相近原则,随机分为5个试验组(n=6):正常组(C,常规饲料)、高脂模型组(M,高脂乳剂)、1.35 g/(kg·bw)陈醋干预M组(LV)、2.7 g/(kg·bw)陈醋干预M组(V)、5.4 g/(kg·bw)陈醋干预M组(HV)。正常组各大鼠2.0 mL/d生理盐水持续灌胃10周,试验组高脂乳剂10 mL/kg·d灌胃5周后生理盐水继续灌胃5周,山西老陈醋灌胃组各大鼠陈醋(2.0 mL/d)及高脂乳剂(10 mL/kg·d)灌胃5周后使用陈醋继续灌胃(2.0 mL/d)5周。于温度(22±2 ℃)、湿度(50%±5%)标准动物房内常规饲料喂养,自由采食。
11周试验结束后,禁食16 h,乙醚麻醉,腹主动脉采血,全血样本室温2 h,4 ℃ 2 h,3000 r/min离心15 min,取血清于−80 ℃保存。采集各组大鼠同一位置肝脏组织,4%多聚甲醛固定,剩余组织−80 ℃保存。
1.2.4 不同试验组大鼠血清中脂质含量、抗氧化及肝功能检测
根据说明书,使用酶标仪和紫外分光光度计检测各组大鼠血清中TC、TG、HDL-C含量,SOD、GR、T-AOC酶活性,ALT、AST、AKP的酶活力单位,M组与C组血脂含量(TC、TG、HDL-C)有统计学差异即为造模成功[16]。
1.2.5 不同试验组大鼠肝脏组织病理学检测
大鼠肝组织4%多聚甲醛固定24 h后,取出修块,经常规脱水、透明、浸蜡、包埋、切片(厚度5 μm)后,苏木素-伊红(HE)染色,中性树胶封片,在200倍光学显微镜下观察组织病理变化。
1.2.6 不同试验组大鼠肝脏中PI3K/AKT1通路mRNA水平表达检测
根据NCBI(https://www.ncbi.nlm.nih.gov)上检索的大鼠PIK3CA、AKT1和GAPDH CDS区,利用Primer Premier 3.0分别设计3对特异性引物(表1),由上海生工生物工程股份有限公司合成。
表 1 引物序列信息Table 1. Primer sequence information基因 登录号 引物序列(5'→3') 产物(bp) PIK3CA NM_133399.3 AAGTGGCCCAGATGTACTGC
CAGCAAAGCTCCGAACCATG111 AKT1 NM_033230.3 GGAAGGTGATCCTGGTGAAG
CGGTTCTCAGTAAGCGTGTG115 GAPDH NM_017008.4 ATGACTCTACCCACGGCAAG
TGGGTTTCCCGTTGATGACC75 总RNA提取试剂盒提取各组大鼠肝脏总RNA,核酸蛋白检测仪测定浓度和纯度,根据HiScript Ⅲ RT SuperMix for qPCR说明书逆转录RNA为cDNA。相对荧光定量法(qRT-PCR)检测大鼠肝脏中PIK3CA、AKT1 mRNA表达,根据2×Realtime PCR Super mix试剂盒说明构建反应体系(20 μL):2×Realtime PCR Super mix 10 μL、cDNA模板2.0 μL、正反向引物各0.5 μL、ddH2O 7.0 μL,每个指标重复3次;设置反应程序:94 ℃预变性5 min;94 ℃ 30 s,60 ℃ 30 s,72 ℃ 30 s,循环40次;72 ℃ 5 min。利用2−ΔΔCT法计算不同实验组大鼠肝脏中PIK3CA及AKT1 mRNA水平表达。
1.2.7 不同实验组大鼠肝脏中PI3K/AKT1通路蛋白水平表达检测
使用组织裂解液(含PMSF)提取不同试验组大鼠肝脏组织总蛋白,4 ℃ 14000 r/min离心10 min,BCA试剂盒检测蛋白浓度。SDS-PAGE电泳时,上样量为50 μg/孔,100 ℃水浴变性5 min,按照10%分离胶80 V 20 min,5%浓缩胶120 V 60 min电泳,200 mA 60 min恒流湿转至NC膜,5%脱脂奶粉室温摇床封闭2 h,TBST洗膜5次,抗PIK3CA兔多克隆抗体及抗AKT1多克隆抗体(1:300)、抗GAPDH鼠单克隆抗体(1:5000)4 ℃孵育过夜(12~16 h),移去一抗,山羊抗鼠二抗及山羊抗兔二抗(1:15000)室温孵育1 h,TBST漂洗5次,ELC Plus超敏发光液显色,ImageJ 8.0分析蛋白免疫印迹灰度值。
1.2.8 不同试验组大鼠肝脏中PI3K/AKT1通路定位分析
取前期烘干备用且组织完整的切片,按照通用SP检测试剂盒步骤依次进行脱蜡,柠檬酸钠缓冲液抗原修复,过氧化氢酶灭活,非特异性抗原位点封闭,抗PIK3CA兔多克隆抗体及抗AKT1多克隆抗体(1:100)4 ℃孵育过夜,复温后,PBS漂洗,室温孵育二抗,PBS漂洗,DAB显色,蒸馏水终止反应,苏木素复染,盐酸酒精分化,常规脱水后中性树胶封片,光学显微镜下观察,Image-Pro Plus 6.0进行光密度值分析。
1.3 数据处理
使用Graphpad Prism 8.3.0分析作图,所有数据以平均数±标准差(±SD)表示,采用单因素方差分析,两两比较采用独立t检验。P<0.01作为差异极显著的界值,P<0.05作为差异显著的界值。
2. 结果与分析
2.1 不同试验组大鼠血清中脂质含量、抗氧化活性及肝损伤指标检测结果
2.1.1 血清脂质含量检测结果
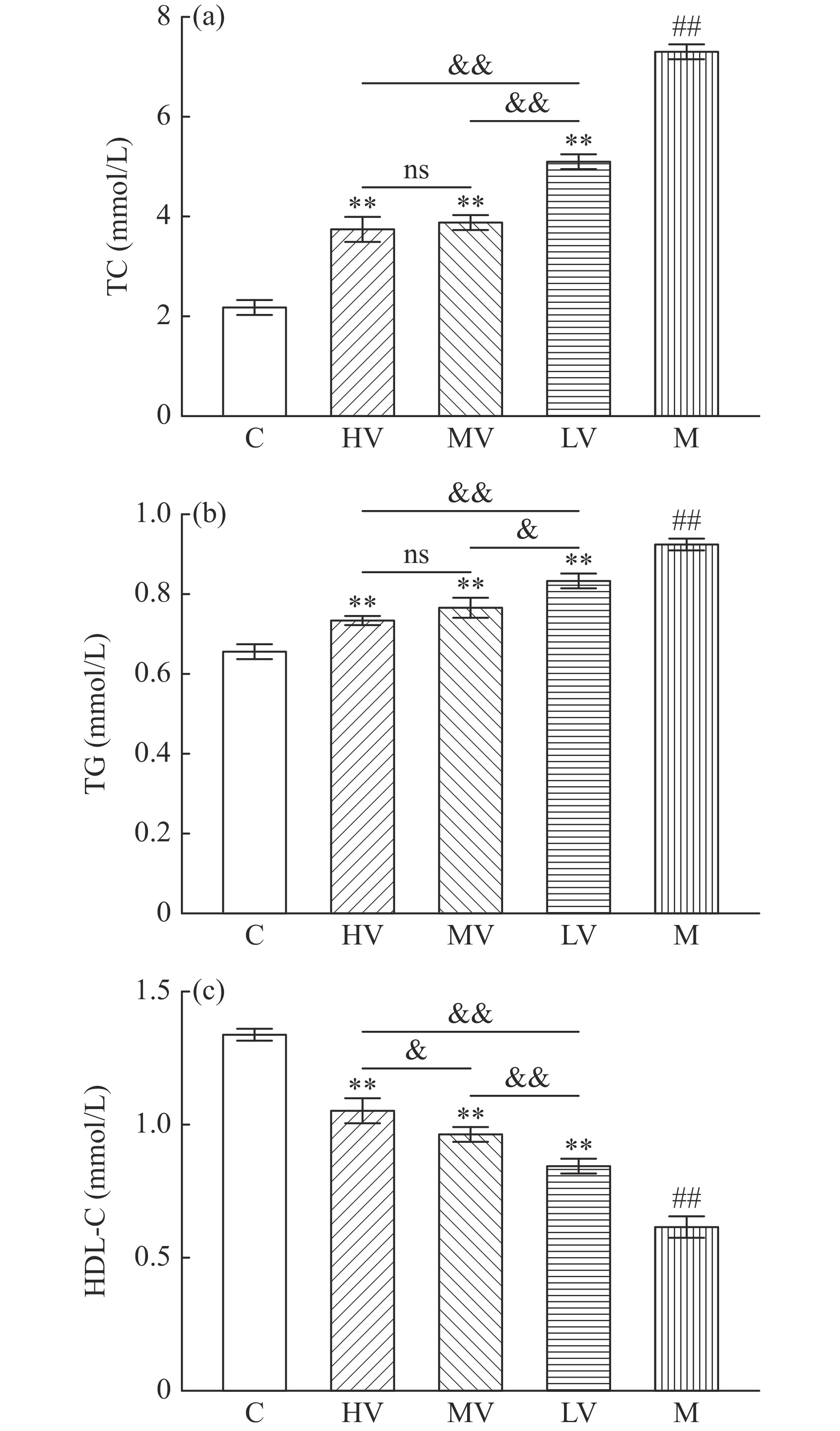
高脂饮食会导致大鼠体内脂质过度沉积,脂代谢紊乱,引发高脂血症,显著特征是血清内TC,TG含量升高,HDL-C含量低于正常值[17]。如图2所示,与C组相比,M组TC及TG极显著增加(P<0.01),HDL-C极显著降低(P<0.01),说明,高脂血症大鼠模型构建成功[16];与M组相比,HV、MV和LV组TC及TG均极显著降低(P<0.01),HDL-C极显著升高(P<0.01);与LV组相比,HV和MV组TC及TG(极)显著降低(P<0.05,P<0.01),HDL-C极显著升高(P<0.01);与MV组相比,HV组HDL-C显著升高(P<0.05),TC、TG差异不显著(P>0.05),结果表明,不同剂量山西老陈醋均可改善高脂饮食引起的脂代谢紊乱,中高剂量组改善效果优于低剂量组。
2.1.2 抗氧化酶活性检测结果
血清内脂质升高,会导致氧自由基(Reactive oxygen species,ROS)增加,使机体发生一定程度的脂质过氧化,T-AOC表示机体内总抗氧化能力,SOD和GR是氧自由基清除剂[18]。如图3所示,与C组相比,M组SOD、GR及T-AOC均极显著降低(P<0.01),说明脂质堆积使大鼠机体抗氧化酶系统紊乱;与M组相比,HV、MV和LV组SOD和T-AOC均(极)显著升高(P<0.05,P<0.01),MV和HV组GR极显著升高(P<0.01),LV组GR无显著差异;与LV相比,HV组SOD及GR均极显著升高(P<0.01),MV组T-AOC和GR(极)显著升高(P<0.05,P<0.01);与MV组相比,HV组SOD极显著升高(P<0.01),T-AOC极显著降低(P<0.01),GR无显著差异(P>0.05),结果表明,山西老陈醋可提高机体抗氧化能力,降低高脂饮食大鼠氧化应激水平。
2.1.3 肝损伤指标检测结果
血清内ALT和AST含量升高是肝细胞受损的重要标志,AKP含量和肝损伤的程度呈负相关[19]。如图4所示,与C组相比,M组ALT及AST均极显著升高(P<0.01),AKP极显著降低(P<0.01),说明血脂过高使肝细胞受损,使细胞质内ALT和AST释放至血液内[20],AKP合成减少;与M组相比,HV、MV和LV组ALT及AST均极显著降低(P<0.01),AKP均极显著升高(P<0.01);与LV组相比,MV和HV组ALT及AST均(极)显著降低(P<0.05,P<0.01),AKP极显著升高(P<0.01);与MV组相比,HV组AST极显著降低(P<0.01),ALT及AKP无显著差异(P>0.05),结果表明,山西老陈醋对肝脏具有一定保护作用,可有效减轻高脂饮食诱发的肝脂质损伤。
2.2 肝脏组织病理学检测结果
HE染色结果显示(图5),与C组相比,M组肝索排列紊乱,肝细胞脂肪变性及空泡样变增多,肝细胞肿胀,细胞核移位,胞浆内出现致密且较大的脂滴空泡;与M组相比,LV组、MV组、HV组细胞变性及脂滴空泡明显减少,HV组和MV组干预效果优于LV组,结果表明,山西老陈醋可有效改善大鼠肝脏脂肪蓄积现象,缓解肝组织病变。
2.3 山西老陈醋对各组大鼠PIK3CA、AKT1 mRNA水平表达的影响
qRT-PCR检测结果显示(图6),与C组相比,M组PIK3CA及AKT1 mRNA水平表达均极显著升高(P<0.01),说明大鼠脂代谢异常导致肝脏内PI3K/AKT1信号传导通路被激活;与M组相比,LV、MV和HV组PIK3CA及AKT1 mRNA水平表达均极显著降低(P<0.01);与LV组相比,MV和HV组PIK3CA及AKT1 mRNA水平表达均极显著降低(P<0.01);与MV组相比,HV组PIK3CA及AKT1 mRNA水平表达无显著差异(P>0.05),结果表明,山西老陈醋可显著改善高脂饮食诱发大鼠肝脏内PIK3CA及AKT1基因表达异常。
2.4 山西老陈醋对各组大鼠PIK3CA、AKT1蛋白水平表达的影响
Western blotting检测结果显示(图7),与C组相比,M组PIK3CA及AKT1蛋白表达均极显著升高(P<0.01),说明肝脂质蓄积使大鼠肝脏内PIK3CA及AKT1蛋白表达异常;与M组相比,HV、MV和LV组PIK3CA及AKT1蛋白表达极显著降低(P<0.01);与LV组相比,HV和MV组PIK3CA及AKT1蛋白表达均极显著降低(P<0.01);与MV组相比,HV组PIK3CA蛋白表达极显著降低(P<0.01),AKT1表达无显著差异(P>0.05),结果表明,山西老陈醋可有效抑制高脂血症大鼠肝脏内PIK3CA及AKT1蛋白表达。
2.5 山西老陈醋对各组大鼠PIK3CA、AKT1定位的影响
免疫组化分析结果显示(图8),各组大鼠肝脏细胞核和细胞质中均有PIK3CA和AKT1表达,与C组相比,M组PIK3CA及AKT1表达极显著升高(P<0.01)。与M组相比,HV、MV和LV组PIK3CA及AKT1表达极显著降低(P<0.01),与LV组相比,HV和MV组PIK3CA与AKT1表达(极)显著降低(P<0.05,P<0.01),结果表明,山西老陈醋通过抑制大鼠肝脏PIK3CA及AKT1的表达进而调控PI3K/AKT1通路。
3. 讨论与结论
目前,高脂血症的治疗主要以中药和膳食辅助治疗为主[21],山西老陈醋富含多种活性物质,可调节血脂保护肝脏[22]。研究证实,山西老陈醋可以降低醉酒小鼠肝功酶活性[19],提取的多酚通过抑制TC、TG分泌调节脂代谢[23]。本次研究中,高脂血症大鼠体内PI3K/AKT1信号转导途径被激活,不同剂量山西老陈醋均可抑制该通路。PI3K/AKT1通路参与胆固醇的摄入与转导,调控该通路及其下游脂质合成基因表达[11],可调节脂代谢。研究结果表明,山西老陈醋可能通过下调PIK3CA及AKT1基因和蛋白的表达,改善高脂饮食诱发的血脂水平、肝功酶活性异常和肝脏病理性损伤,且浓度为2.7和5.4 g/(kg·bw)山西老陈醋对血脂、肝功酶水平异常和肝脂肪性病变改善效果均优于1.35 g/(kg·bw)低剂量干预组,中高剂量组改善效果无显著差异。
高脂血症通常伴随着氧化应激的发生,高脂饮食诱导脂质积累,肝脏脂代谢紊乱[24],游离脂肪酸堆积,肝细胞脂肪变性,抗氧化酶系统紊乱,过量产生的ROS无法被清除,引发氧化应激损伤[25],研究证实山西老陈醋多酚类提取物具有强抗氧化活性[26],陈醋源类黑素通过调控AKT信号传导途径影响ROS生成[27]。本次研究结果表明,山西老陈醋通过提高抗氧化酶活性,增加大鼠抗氧化能力。PI3K/AKT1信号转导途径与多种细胞氧化应激密切相关[28],可通过调节ROS的生成调节氧化应激[29],进一步研究发现多种活性物质均可通过抑制PI3K/AKT1的表达来改善氧化应激[30-31],本次研究结果显示,干预组大鼠肝脏内PIK3CA及AKT1在基因和蛋白水平上均显著下降,说明山西老陈醋改善氧化应激可能是通过抑制PI3K/AKT1的表达来实现的,且2.7 g/(kg·bw)和5.4 g/(kg·bw)山西老陈醋干预组抗氧化及抑制PI3K/AKT1通路能力最为显著。进一步研究发现山西老陈醋浓缩物的降脂作用,优于陈醋原液[5]。这可能是由于山西老陈醋内富含的有机物均具有挥发性[32],浓缩过程中以乙酸为主的有机酸[33]和黄酮类物质损失量最大[34],且本研究中,高剂量组浓度及大鼠摄入量均高于前人研究,综上推测,山西老陈醋的最适浓度可能在2.7~5.4 g/(kg·bw)之间。
综上,不同剂量山西老陈醋均可改善高脂血症大鼠的脂代谢紊乱和氧化应激损伤,高脂饮食诱导大鼠体内PI3K/AKT1通路高度活化,山西老陈醋干预下肝脏内PI3K/AKT1通路受到不同程度抑制,浓度为2.7~5.4 g/(kg·bw)山西老陈醋改善效果最佳。由此,本研究推测山西老陈醋可能通过抑制PI3K/AKT1通路,进而改善高脂血症大鼠抗氧化应激和脂代谢紊乱的能力,但其靶向抑制PI3K/AKT1信号通路的机制尚不明确,有待进一步研究。
-
表 1 引物序列信息
Table 1 Primer sequence information
基因 登录号 引物序列(5'→3') 产物(bp) PIK3CA NM_133399.3 AAGTGGCCCAGATGTACTGC
CAGCAAAGCTCCGAACCATG111 AKT1 NM_033230.3 GGAAGGTGATCCTGGTGAAG
CGGTTCTCAGTAAGCGTGTG115 GAPDH NM_017008.4 ATGACTCTACCCACGGCAAG
TGGGTTTCCCGTTGATGACC75 -
[1] FREDERICK M I, SIDDIKA T, ZHANG P, et al. miRNA-Dependent regulation of AKT1 phosphorylation[J]. Cells,2022,11(5):821. doi: 10.3390/cells11050821
[2] TANG H, ZENG Q, TANG T, et al. Kaempferide improves glycolipid metabolism disorder by activating PPARγ in high-fat-diet-fed mice[J]. Life Sciences,2021,270:119133. doi: 10.1016/j.lfs.2021.119133
[3] NIE Y, LUO F, FIBER D, An opportunity for a global control of hyperlipidemia[J]. Oxid Med Cell Longev, 2021, 5542342.
[4] DU P, SONG J, QIU H, et al. Polyphenols extracted from Shanxi-aged vinegar inhibit inflammation in LPS-induced RAW264.7 macrophages and ICR mice via the suppression of MAPK/NF-κB pathway activation[J]. Molecules,2021,26(9):2745. doi: 10.3390/molecules26092745
[5] 丁月娣, 闫裕峰, 陈亨业, 等. 浓缩山西老陈醋对高脂血症小鼠血脂及血糖的调节作用[J]. 食品工业科技,2020,41(12):286−290. [DING Y D, YAN Y F, CHEN H Y, et al. Regulation effect of Shanxi aged vinegar concentrate on blood lipid and blood glucose in hyperlipidemia mice[J]. Science and Technology of Food Industry,2020,41(12):286−290. DING Y D, YAN Y F, CHEN H Y, et al. Regulation effect of Shanxi aged vinegar concentrate on blood lipid and blood glucose in hyperlipidemia mice[J]. Science and Technology of Food Industry, 2020, 41(12): 286-290.
[6] 许女, 王超宇, 李雅茹, 等. 山西老陈醋源植物乳杆菌CP234对大鼠非酒精性脂肪肝的保护作用及机制[J]. 中国食品学报,2020,20(11):26−34. [XU N, WANG C Y, LI Y R, et al. Protective effect of Lactobacillus plantarum CP234 isolated from Shanxi aged vinegar on nonalcoholic fatty liver disease in rats[J]. Journal of Chinese Institute of Food Science and Technology,2020,20(11):26−34. doi: 10.16429/j.1009-7848.2020.11.004 XU N, WANG C Y, LI Y R, et al. Protective effect of Lactobacillus plantarum CP234 isolated from Shanxi aged vinegar on nonalcoholic fatty liver disease in rats[J]. Journal of Chinese Institute of Food Science and Technology, 2020, 20(11): 26-34. doi: 10.16429/j.1009-7848.2020.11.004
[7] DU P, ZHOU J, ZHANG L, et al. GC×GC-MS analysis and hypolipidemic effects of polyphenol extracts from Shanxi aged vinegar in rats under a high fat diet[J]. Food Function,2020,11(9):7468−7480. doi: 10.1039/D0FO01266A
[8] GANESAN K, XU B. Anti-diabetic effects and mechanisms of dietary polysaccharides[J]. Molecules,2019,24(14):2556. doi: 10.3390/molecules24142556
[9] GUO X X, ZENG Z, QIAN Y Z, et al. Wheat flour, enriched with γ-oryzanol, phytosterol, and ferulic acid, alleviates lipid and glucose metabolism in high-fat-fructose-fed rats[J]. Nutrients,2019,11(7):1697. doi: 10.3390/nu11071697
[10] ZHENG Y, XIE L, XU S, et al. Effects of miR-202-5p silencing PIK3CA gene expression on proliferation, invasion, and epithelial-mesenchymal transition of cervical cancer SiHa cells through inhibiting PI3K/Akt/mTOR signaling pathway activation[J]. Molecular and Cellular Biochemistry,2021,476(11):4031−4044. doi: 10.1007/s11010-021-04211-4
[11] CANAUD G, HAMMILL AM, ADAMS D, et al. A review of mechanisms of disease across PIK3CA-related disorders with vascular manifestations[J]. Orphanet J Rare Dis,2021,16(1):306. doi: 10.1186/s13023-021-01929-8
[12] LIU Z, LI J, LIN S, et al. PI3K regulates the activation of NLRP3 inflammasome in atherosclerosis through part-dependent AKT signaling pathway[J]. Exp Anim,2021,70(4):488−497. doi: 10.1538/expanim.21-0002
[13] CHEN Y, LIAO W, ZHU Z, et al. Essential oil from the raw and vinegar-processed Rhizoma Curcumae ameliorate CCl-induced liver fibrosis: Integrating network pharmacology and molecular mechanism evaluation[J]. Food Function,2021,12(9):4199−4220. doi: 10.1039/D0FO03052J
[14] XIA T, DUAN W, ZHANG Z, et al. Polyphenol-rich extract of Zhenjiang aromatic vinegar ameliorates high glucose-induced insulin resistance by regulating JNK-IRS-1 and PI3K/Akt signaling pathways[J]. Food Chem,2021,335:127513. doi: 10.1016/j.foodchem.2020.127513
[15] PANG M, FANG Y, CHEN S, et al. Gypenosides inhibits xanthine oxidoreductase and ameliorates urate excretion in hyperuricemic rats induced by high cholesterol and high fat food (lipid emulsion)[J]. Medical Science Monitor,2017,23:1129−1140. doi: 10.12659/MSM.903217
[16] HU N, CHEN C, WANG J, et al. Atorvastatin ester regulates lipid metabolism in hyperlipidemia rats via the PPAR-signaling pathway and HMGCR expression in the liver[J]. Int J Mol Sci,2021,22(20):11107. doi: 10.3390/ijms222011107
[17] 张明昊, 郭申, 杜婧雯, 等. 灯盏花素通过调控LKB1/AMPK和Notch1/Jagged1通路改善高脂血症大鼠血脂水平及肝肾功能的作用机制研究[J]. 世界科学技术-中医药现代化,2022,24(6):2340−2352. [ZHANG M H, GUO S, DU J W, et al. Restoring the blood lipid levels and liver and renal functions of breviscapine on hyperlipidemia rats by regulating LKB1/AMPKand Notch1/Jagged1 signal pathway[J]. Modernization of Traditional Chinese Medicine and Materia Medica-World Science and Technology,2022,24(6):2340−2352. ZHANG M H, GUO S, DU J W, et al. Restoring the blood lipid levels and liver and renal functions of breviscapine on hyperlipidemia rats by regulating LKB1/AMPKand Notch1/Jagged1 signal pathway[J]. Modernization of Traditional Chinese Medicine and Materia Medica-World Science and Technology, 2022, 24(6): 2340-2352.
[18] SANTOS H O, DE-MORAES W M A M, DA-SILVA G A R, et al. Vinegar (acetic acid) intake on glucose metabolism: A narrative review[J]. Clin Nutr ESPEN,2019,32:1−7. doi: 10.1016/j.clnesp.2019.05.008
[19] 田颖蕾, 夏婷, 康超艳, 等. 山西老陈醋对急性醉酒小鼠防醉及护肝作用研究[J]. 中国酿造,2021,40(12):40−44. [TIAN Y L, XIA T, KANG C Y, et al. Anti-drunk and hepatoprotective effects of Shanxi aged vinegar on acute drunkenness mice[J]. China Brewing,2021,40(12):40−44. doi: 10.11882/j.issn.0254-5071.2021.12.008 TIAN Y L, XIA T, KANG C Y, et al. Anti-drunk and hepatoprotective effects of Shanxi aged vinegar on acute drunkenness mice[J]. China Brewing, 2021, 40(12): 40-44. doi: 10.11882/j.issn.0254-5071.2021.12.008
[20] WANG M, ZHANG S, ZHONG R, et al. Olive fruit extracts supplement improve antioxidant capacity via altering colonic microbiota composition in mice[J]. Front Nutr,2021,8:645099. doi: 10.3389/fnut.2021.645099
[21] GUO W L, PAN Y Y, LI L, et al. Ethanol extract of Ganoderma lucidum ameliorates lipid metabolic disorders and modulates the gut microbiota composition in high-fat diet fed rats[J]. Food Function,2018,9(6):3419−3431. doi: 10.1039/C8FO00836A
[22] LIM J, HENRY CJ, HALDAR S. Vinegar as a functional ingredient to improve postprandial glycemic control-human intervention findings and molecular mechanisms[J]. Mol Nutr Food Res,2016,60(8):1837−49. doi: 10.1002/mnfr.201600121
[23] ZHOU Y J, XU N, ZHANG X C, et al. Chrysin improves glucose and lipid metabolism disorders by regulating the AMPK/PI3K/AKT signaling pathway in insulin-resistant HepG2 Cells and HFD/STZ-induced C57BL/6J mice[J]. Agric Food Chem,2021,69(20):5618−5627. doi: 10.1021/acs.jafc.1c01109
[24] ZHANG R, YU Y, DENG J, et al. Sesamin ameliorates high-fat diet-induced dyslipidemia and kidney injury by reducing oxidative stress[J]. Nutrients,2016,8(5):276. doi: 10.3390/nu8050276
[25] XU Q, LI Y C, DU C, et al. Effects of apigenin on the expression of LOX-1, Bcl-2, and bax in hyperlipidemia rats[J]. Chem Biodivers,2021,18(8):e2100049.
[26] HE H, WANG L, QIAO Y, et al. Vinegar/tetramethylpyrazine induces nutritional preconditioning protecting the myocardium mediated by VDAC1[J]. Oxidative Medicine and Cellular Longevity,2021,2021:6670088.
[27] YANG L, WANG X, YANG X. Possible antioxidant mechanism of melanoidins extract from Shanxi aged vinegar in mitophagy-dependent and mitophagy-independent pathways[J]. Journal of Agricultural and Food Chemistry,2014,62(34):8616−22. doi: 10.1021/jf501690e
[28] SHEN B, ZHAO C, WANG Y, et al. Aucubin inhibited lipid accumulation and oxidative stress via Nrf2/HO-1 and AMPK signalling pathways[J]. Journal of Cellular and Molecular Medicine,2019,23(6):4063−4075. doi: 10.1111/jcmm.14293
[29] CHEN J, NAN R, WANG R, et al. Ester-producing mechanism of ethanol o-acyltransferase EHT1 gene in pichia pastoris from Shanxi aged vinegar[J]. BioMed Research Internationalt,2019,2019:4862647.
[30] KANDYLIS P, BEKATOROU A, DIMITRELLOU D, et al. Health promoting properties of cereal vinegars[J]. Foods,2021,10(2):344. doi: 10.3390/foods10020344
[31] HUANG J, ZHENG L, WANG F, et al. Mangiferin ameliorates placental oxidative stress and activates PI3K/Akt/mTOR pathway in mouse model of preeclampsia[J]. Archives of Pharmacal Research,2020,43(2):233−241. doi: 10.1007/s12272-020-01220-7
[32] LI S, LI P, LIU X, et al. Bacterial dynamics and metabolite changes in solid-state acetic acid fermentation of Shanxi aged vinegar[J]. Appl Microbiol Biotechnol,2016,100(10):4395−411. doi: 10.1007/s00253-016-7284-3
[33] OUSAAID D, MECHCHATE H, LAAROUSSI H, et al. Fruits vinegar: Quality characteristics, phytochemistry, and functionality[J]. Molecules,2021,27(1):222. doi: 10.3390/molecules27010222
[34] XIE S, SONG J, FAN B, et al. Elucidation and regulation of polyphenols in the smoking process of Shanxi aged vinegar[J]. Foods,2021,10(7):1518. doi: 10.3390/foods10071518
-
期刊类型引用(4)
1. 刘明珠,华玲,张秀萍,丁燃容,刘媛媛. 微波花椒鸡丁预制菜的工艺优化及其挥发性风味物质分析. 四川旅游学院学报. 2025(01): 30-35 .  百度学术
百度学术
2. 王美仁,王柏辉,杨帆,刘学勤,苏春霞,段晓蓉. 不同加工方式对牛肉干产品风味物质的影响. 食品安全质量检测学报. 2025(02): 127-137 .  百度学术
百度学术
3. 玛尔哈巴·帕尔哈提,宋占腾,杨睿娜,李静,安静,刘敏,徐贞贞,朱靖蓉. 不同品种恰玛古风味品质分析. 食品工业科技. 2025(04): 306-315 .  本站查看
本站查看
4. 郑佳琦,宫雅姝,牛梦迪,王军. 鲫鱼冷藏期间新鲜度与挥发性物质相关性分析. 食品与发酵工业. 2024(23): 226-233 .  百度学术
百度学术
其他类型引用(1)






 下载:
下载:






 下载:
下载:
