Optimization of Aqueous Two-phase Extraction Technology of Total Flavonoids from Lonicera japonica Thunb. Leaves and Its Antioxidant Activity
-
摘要: 研究了乙醇-(NH4)2SO4双水相提取金银花叶总黄酮工艺,并探讨了总黄酮的抗氧化活性。以(NH4)2SO4质量分数、液料比、浸提温度和浸提时间等因素为自变量,以总黄酮得率为指标,采用正交试验对工艺参数进行了优化。通过对DPPH自由基、羟基自由基和超氧阴离子自由基清除率的测定,评价总黄酮的抗氧化性能。最佳实验条件为:(NH4)2SO4质量分数15%,液料比25:1 mL/g,浸提温度50 ℃,浸提时间30 min。在该条件下,总黄酮得率为4.98%。当金银花叶总黄酮浓度为0.48 mg/mL时,DPPH自由基的清除率为97.61%,浓度为0.72 mg/mL时,羟基自由基的清除率为86.53%,超氧阴离子自由基的清除率为75.29%,表现出良好的抗氧化性能。Abstract: The ethanol-ammonium sulfate aqueous two-phase extraction of total flavonoids from the leaves of Lonicera japonica Thunb. leaves was studied, and the antioxidant activity of the total flavonoids was further discussed. The extraction process was optimized by orthogonal test with the parameters including mass fraction of ammonium sulfate, liquid-to-material ratio, extraction temperature and extraction time as independent variables and the yield of the flavonoids as index. The antioxidant activity of the total flavonoids was evaluated by measuring the clearance rates of DPPH free radicals, hydroxyl free radicals and superoxide anion free radicals. The optimum experimental conditions were as follows: The mass fraction of ammonium sulfate was 15%, the liquid-to-material ratio was 25:1 mL/g, the extraction temperature was 50 ℃, and the extraction time was 30 min. Below this term, the yield of flavonoids was 4.98%. The clearance rate of DPPH free radicals was 97.61% when the concentration of total flavonoids from the leaves of Lonicera japonica Thunb. was 0.48 mg/mL, and those of the hydroxyl free radicals and superoxide anion free radicals were 86.53% and 75.29%, respectively, when the concentration of flavonoids was 0.72 mg/mL, showing good antioxidant activity.
-
金银花(Lonicera japonica Thunb.)为忍冬科忍冬属植物,分布广泛,适应性强,是一种药食同源的绿色产品,具有清热解毒、抗病毒、消炎等功效[1-2]。金银花叶作为金银花的副产品,产量高,但长期以来尚未得到充分合理地利用[3]。文献研究资料表明,金银花叶中含有黄酮、有机酸、挥发油、环烯醚萜苷及其它等多种活性成分[4]。事实上,金银花叶相较于其花中的黄酮含量更高[5],因此极具开发应用前景。
近年来,学者们主要以乙醇/水为溶剂,对金银花叶总黄酮的提取工艺进行了研究。田存章等[3]采用乙醇回流法提取金银花叶总黄酮,得率为0.487%;李康强等[6]采用超声波辅助乙醇提取金银花叶总黄酮,得率为1%;谢威等[7]采用复合酶辅助水提取金银花叶总黄酮,得率为4.60%。双水相萃取技术是一种新型的提取技术,它结合了萃取、分离以及目标产品部分纯化等功能[8],具有条件温和、处理量大、易于连续操作等优点[9],并且已有效应用于黄酮类物质的提取[10-12],与传统溶剂提取法相比,双水相法黄酮提取率更高,有机溶剂用量更少[13-14]。然而,目前双水相技术在金银花叶总黄酮的提取中尚未见公开报道。
与聚合物/盐、表面活性剂/盐以及离子液体/盐等其它类型的双水相体系相比,小分子醇/盐双水相体系则具有黏度小、不易乳化、分离迅速、操作简便、易于回收等优点[15],与传统有机溶剂相比,则具有不易挥发、污染小等优点,从而具有更强的工业化应用价值。
综上,本研究采用廉价、易得的乙醇和硫酸铵构建双水相体系,研究金银花叶总黄酮提取工艺,以黄酮得率为指标,考察了硫酸铵质量分数、液料比、浸提温度、浸提时间等因素对黄酮得率的影响,采用正交试验优化工艺参数,并分析探讨金银花叶总黄酮的抗氧化活性,以期为金银花叶总黄酮的简便、高效提取及其资源化利用提供理论依据。
1. 材料与方法
1.1 材料与仪器
金银花叶(采收时间为6月) 重庆秀山;1,1-二苯基-2-三硝基苯肼(DPPH)、九水合硝酸铝、芦丁、氢氧化钠、水杨酸、硫酸铵、亚硝酸钠、七水合硫酸亚铁、邻苯三酚、三羟甲基氨基甲烷(Tris)、抗坏血酸(VC)、无水乙醇、30%双氧水溶液、浓盐酸、木犀草苷、木犀草素、槲皮素 均为分析纯,上海麦克林生化科技有限公司;蒸馏水 实验室自制。
FW177中药粉碎机 杭州正迈科技有限公司;FA2204B分析天平 杭州仪速通科技有限公司;L6S型紫外可见分光光度计 上海精密科学仪器有限公司;KL04A低速离心机 上海双旭电子有限公司;DF-101S集热式恒温加热磁力搅拌器 杭州慧创仪器设备有限公司;SK220H型超声波清洗器 上海科导超声仪器有限公司;TENSOR27红外光谱仪 德国布鲁克分析仪器公司;P230II液相色谱 大连依利特分析仪器有限公司。
1.2 实验方法
1.2.1 金银花叶预处理
将金银花叶自然晾干,再用粉碎机将其研磨成粉状,过60目筛。将过筛后的金银花叶粉末保存于样品袋中备用。
1.2.2 金银花叶总黄酮双水相提取工艺
精确称取一定质量的(NH4)2SO4,加入至容积为15 mL的具塞试管中,然后加入适量质量分数为30%的乙醇溶液,在室温(25 ℃)下,超声(200 W,15 min)作用使固体微粒全部溶解,然后静置直至体系形成双水相。固定双水相的体积为10 mL,按照所设定的液料比称取一定质量的金银花叶粉末,加入试管中,水浴加热一定时间后,离心分离(3000 r/min,5 min),并测定上下相中的总黄酮得率[10]。
1.2.3 单因素实验
探究不同(NH4)2SO4质量分数(12%、13%、14%、15%、16%)、液料比(20:1、25:1、30:1、35:1、40:1 mL/g)、浸提温度(30、40、50、60、70 ℃)、浸提时间(10、20、30、40、50 min)等因素对总黄酮得率的影响。各个因素的固定水平值为(NH4)2SO4质量分数14%,液料比30:1 mL/g,浸提温度50 ℃,浸提时间30 min。
1.2.4 正交试验
正交试验选取(NH4)2SO4质量分数、液料比、浸提温度、浸提时间等因素为自变量,各因素分别设置三个水平,依据金银花叶总黄酮得率的考察结果,确定最佳工艺参数。表1为L9(34)正交优化试验因素水平设计表。
表 1 正交试验因素和水平设计Table 1. Design of factors and levels of orthogonal experiment水平 A (NH4)2SO4
质量分数(%)B 液料比
(mL·g−1)C 浸提温度
(℃)D 浸提时间
(min)1 13 25:1 40 20 2 14 30:1 50 30 3 15 35:1 60 40 1.2.5 金银花叶总黄酮得率的测定
芦丁标准曲线溶液的绘制:准确称取10 mg芦丁,用无水乙醇溶解,以质量分数为60%的乙醇溶液定容至50 mL,配制成浓度为0.2 mg/mL的芦丁标准溶液。参考文献[16-17]中的方法,以510 nm为检测波长,测定并记录芦丁标准溶液的吸光度值,绘制标准曲线,得到吸光度值和浓度的线性关系方程:y=12.172x−0.0022(R2=0.9991)。
金银花叶总黄酮得率的测定:取金银花叶黄酮提取液1 mL,加入10 mL容量瓶中,按上述标准曲线方法进行检测,对提取液中总黄酮的含量进行测定,再由式(1)计算金银花叶总黄酮得率(Y)。并根据式(2)计算总黄酮萃取率(E)。
(1) (2) 式中:C1表示上相中的总黄酮含量,mg/mL;V1表示上相体积,mL;C2表示下相中的总黄酮含量,mg/mL;V2表示下相体积,mL;m表示金银花叶粉末的用量,mg。
1.2.6 液相色谱分析
采用正交试验优选出的最佳提取工艺,提取金银花叶总黄酮。采用HPLC法对金银花叶总黄酮提取物中的木犀草苷、木犀草素和槲皮素三个黄酮化合物的含量进行测定。
标准品溶液的配制和标准曲线的绘制:准确称取适量木犀草苷、槲皮素和木犀草素标准品,分别配制成浓度为10、20、30、40、50 μg/mL的乙醇溶液。在上述色谱条件下进样,进样量分别为10 μL,以浓度为横坐标,峰面积为纵坐标,绘制标准曲线,得到木犀草苷的线性方程为y=27.484x+346.74(R2=0.9993),槲皮素的线性方程为y=35.234x+72.933(R2=0.9997),木犀草素的线性方程为y=38.451x+37.682(R2=0.9995)。
样品溶液的制备:按正交试验所选出的优化条件,提取金银花叶总黄酮,并用无水乙醇稀释,过滤膜(0.22 μm)后,得到金银花叶总黄酮样品溶液。
色谱条件:Hypersil ODS2 C18色谱柱(4.6 mm×200 mm,5 μm);流动相(A)为0.2%磷酸溶液,流动相(B)为甲醇;梯度洗脱(0~5 min,20%~22% B;5~10 min,22%~25% B;10~20 min,25%~35% B;20~25 min,35%~40% B;25~30 min,40%~55% B;30~50 min,55%~20% B);检测波长为350 nm;流速为1.0 mL/min;柱温为25 ℃;运行时间为50 min。
1.2.7 金银花叶总黄酮的抗氧化活性分析
1.2.7.1 样品溶液制备
采用正交试验优选出的最佳提取工艺,提取金银花叶总黄酮,并用无水乙醇进行稀释,获得总黄酮质量浓度分别为0.12、0.24、0.36、0.48、0.60、0.72 mg/mL的样品溶液,冷藏待用。
1.2.7.2 金银花叶总黄酮对DPPH自由基的清除作用
采用DPPH法测定金银花叶总黄酮对DPPH自由基的清除率[17]。分别吸取2 mL样品溶液,加入10 mL离心管中,再分别加入2 mL浓度为0.15 mmol/L的DPPH溶液,摇匀,置于暗处处理30 min,以517 nm为检测波长,测定吸光度值,即为Ai;同时,以无水乙醇取代DPPH溶液,测定吸光度值,即为Aj;以无水乙醇取代待测样品溶液,测定吸光度值,即为A0。并根据式(3)计算DPPH自由基清除率(I)。
(3) 1.2.7.3 金银花叶总黄酮对羟基自由基的清除作用
采用Fenton法测定金银花叶总黄酮对羟基自由基的清除率[18]。分别吸取2 mL待测样品溶液加入10 mL离心管中,并加入2 mL浓度为9 mmol/L的FeSO4溶液,然后依次加入2 mL浓度为9 mmol/L的水杨酸溶液和2 mL浓度为8.8 mmol/L的双氧水溶液,摇匀,静置30 min,以510 nm为检测波长,测定吸光度值,即为Ai;同时,以去离子水取代双氧水溶液,测得吸光度值,即为Aj;以去离子水取代待测样品溶液,测得吸光度值,即为A0。并根据式(3)计算羟基自由基清除率(I)。
1.2.7.4 金银花叶总黄酮对超氧阴离子自由基的清除作用
采用邻苯三酚法测定金银花叶总黄酮对超氧阴离子自由基的清除率[19]。吸取4.5 mL pH为8.2,浓度为50 mmol/L的Tris-HCl缓冲液,加入至10 mL离心管中,在30 ℃的水浴中放置25 min,然后分别加入2 mL待测样品溶液,再加入0.4 mL浓度为25 mmol/L的邻苯三酚溶液,摇匀,反应4 min,随后立即滴加2滴浓度为8 mol/L的盐酸溶液,测得其在320 nm处的吸光度值,即为Ai;同时,以去离子水取代邻苯三酚溶液,测得吸光度值,即为Aj;以去离子水取代待测样品溶液,测得吸光度值,即为A0。并根据式(3)计算超氧阴离子自由基清除率(I)。
1.3 数据处理
采用Origin数据处理软件进行图表制作,采用正交设计助手II3.2软件进行正交优化试验设计和分析。实验数据为重复3次测定的平均值。
2. 结果与分析
2.1 单因素实验结果
2.1.1 (NH4)2SO4质量分数对金银花叶总黄酮得率和萃取率的影响
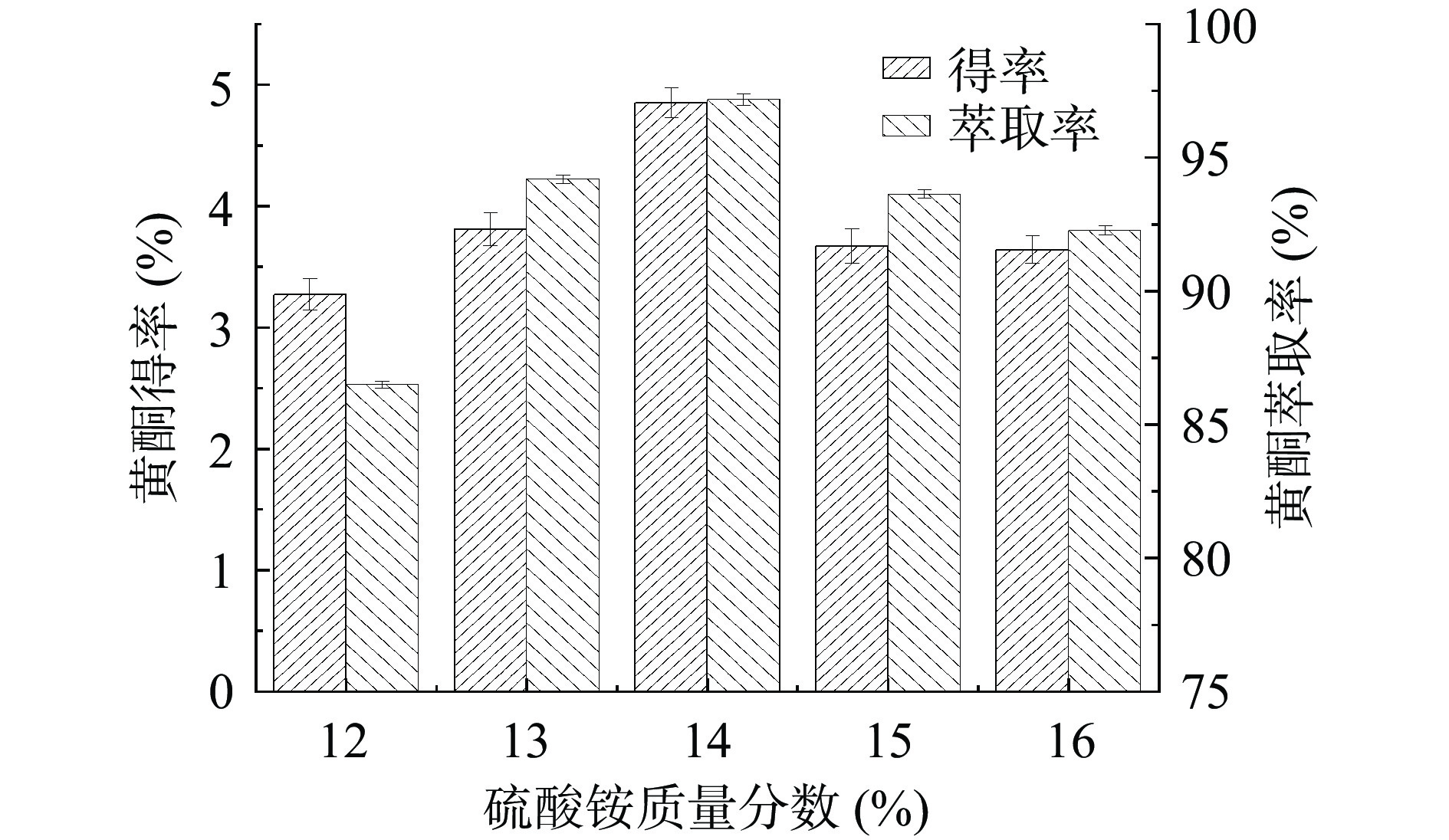
(NH4)2SO4质量分数对金银花叶总黄酮得率和萃取率的影响如图1所示。总黄酮得率和萃取率随着(NH4)2SO4质量分数的增加先增加后降低。当质量分数为14%时,总黄酮得率和萃取率最高。(NH4)2SO4对体系中水分子的束缚作用随着其质量分数的增大而增大,从而提高了上相中乙醇的质量分数,这对促进黄酮的溶出是有利的[20]。随着(NH4)2SO4的质量分数继续增加,其在上相中的含量也相应提高,从而不利于黄酮的萃取。综上,较佳的(NH4)2SO4质量分数为14%。
2.1.2 液料比对金银花叶总黄酮得率和萃取率的影响
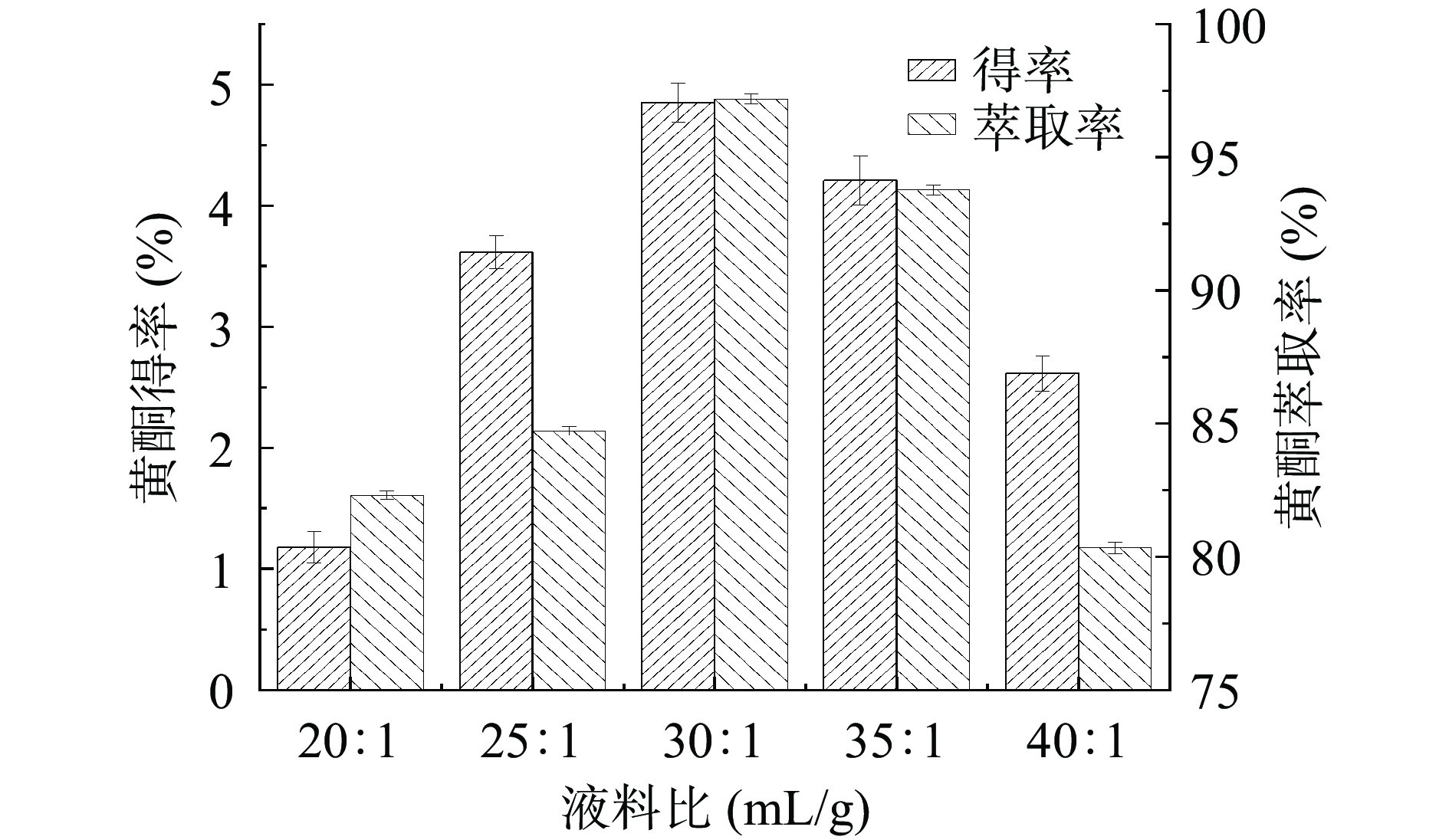
液料比对金银花叶总黄酮得率和萃取率的影响如图2所示。随着液料比的增加,总黄酮得率和萃取率先上升后降低。当液料比为30:1 mL/g时,总黄酮得率和萃取率最高。随着溶剂用量的增加,溶剂渗透、扩散至细胞内以及黄酮物质扩散至溶剂中的速率增大,故得率和萃取率增加[21]。随着液料比的继续增加,推测杂质的溶出会使得黄酮的溶出受到不利影响,黄酮物质在体系中达到溶解平衡[22],此外,溶剂用量过多会使双水相体系的稳定性受到影响[23],故导致得率和萃取率降低。结果表明,较佳的液料比为30:1 mL/g。
2.1.3 浸提温度对金银花叶总黄酮得率和萃取率的影响
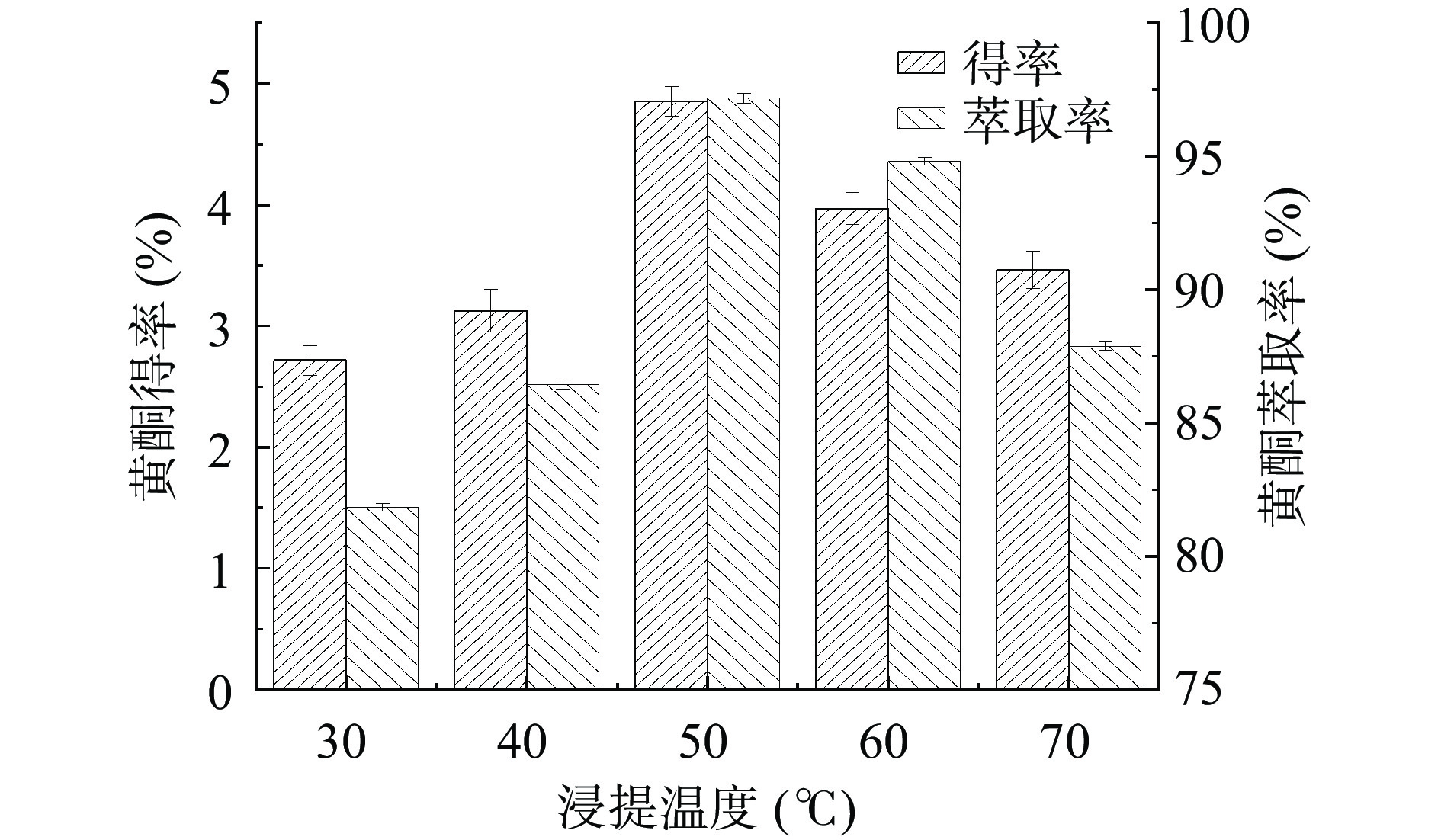
浸提温度对金银花叶总黄酮得率和萃取率的影响如图3所示。随着浸提温度的升高,总黄酮的得率和萃取率先增加后降低。当浸提温度为50 ℃时,总黄酮得率和萃取率最高。随着温度的升高,分子运动速度加快,从而有利于黄酮的溶出,故得率和萃取率增加。然而,当温度进一步增加时,黄酮分子中的酚羟基可能被氧化或破坏[24],故得率和萃取率下降。结果表明,较佳的浸提温度为50 ℃。
2.1.4 浸提时间对金银花叶总黄酮得率和萃取率的影响
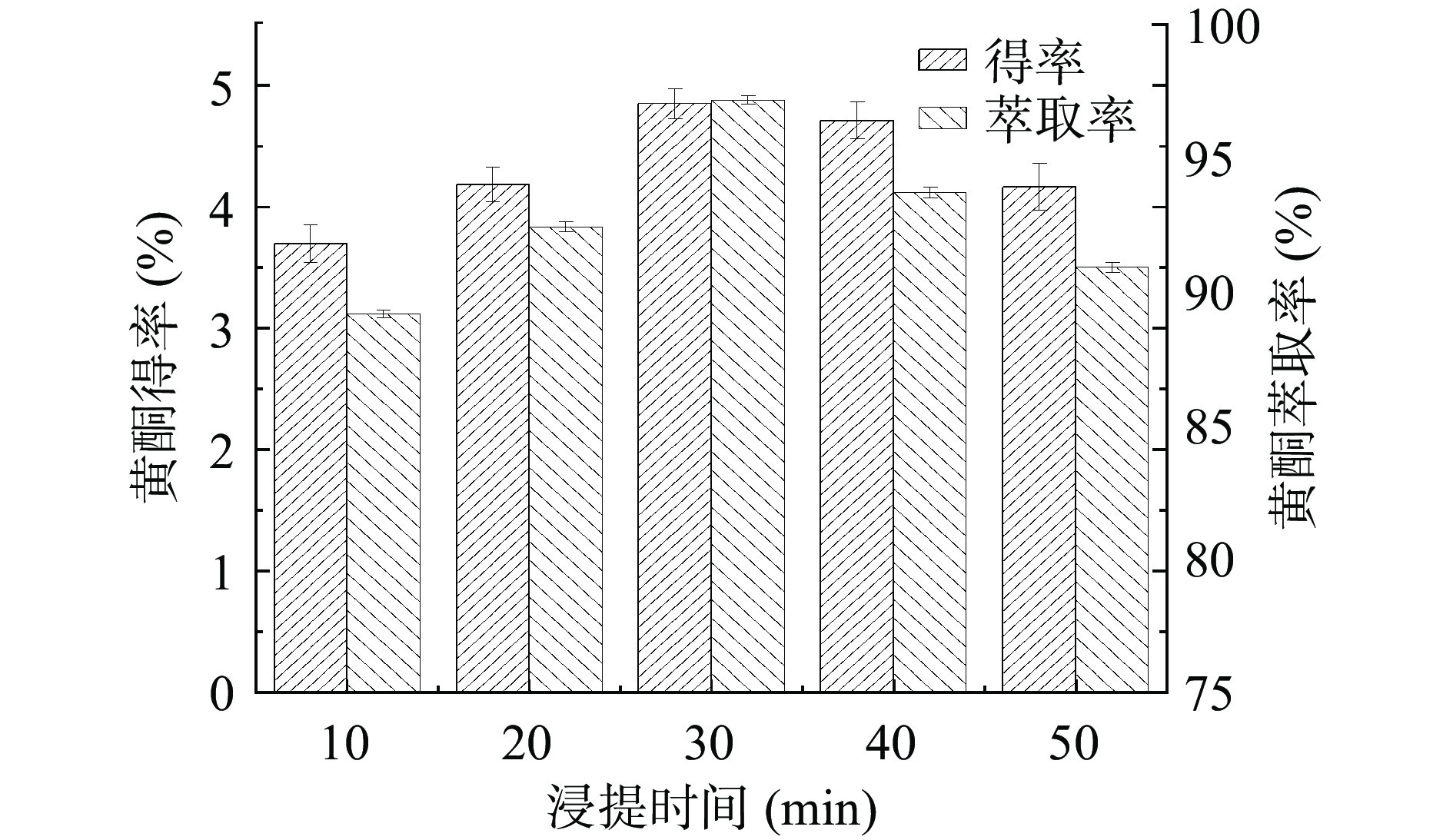
浸提时间对金银花叶总黄酮得率和萃取率的影响如图4所示。总黄酮的得率和萃取率随着浸提时间的增加先增加,然后降低。当浸提时间为30 min时,得率和萃取率最高。可能是由于随着时间的延长,黄酮物质的溶出量逐渐增大,故得率和萃取率增加[24]。当黄酮已基本溶出后,进一步加热则会使其分子结构被破坏,故得率和萃取率下降。结果表明,较佳的浸提时间为30 min。
2.2 正交试验结果
表2为正交试验优化双水相提取金银花叶总黄酮工艺的结果。根据极差R值可判断,不同因素对金银花叶总黄酮得率的影响次序是:A>C>B>D,也就是(NH4)2SO4质量分数>浸提温度>液料比>浸提时间。最佳萃取条件为A3B1C2D2,即(NH4)2SO4质量分数为15%,提液料比为25:1 mL/g,浸提温度为50 ℃,浸提时间为30 min。进行验证性试验,重复试验三次,总黄酮得率分别为4.96%、5.01%、4.98%,平均值为4.98%±0.5053%,试验结果高于实验表中的其它任一组别,相对标准偏差(RSD)为0.5053%,证明该工艺重复性和稳定性都较好。
表 2 正交试验结果Table 2. Results of orthogonal experiments编号 A (NH4)2SO4
质量分数(%)B 液料比
(mL·g−1)C 浸提温度
(℃)D 浸提时间
(min)总黄酮得率
(%)1 13 25:1 40 20 3.21 2 13 30:1 50 30 3.81 3 13 35:1 60 40 2.99 4 14 25:1 50 40 4.91 5 14 30:1 60 20 4.70 6 14 35:1 40 30 4.05 7 15 25:1 60 30 4.92 8 15 30:1 40 40 4.52 9 15 35:1 50 20 4.71 k1 3.337 4.347 3.927 4.207 k2 4.553 4.343 4.477 4.260 k3 4.717 3.917 4.203 4.140 R 1.380 0.430 0.550 0.120 此外,与文献[3, 6-7]相比,本研究中金银花叶总黄酮的得率相对较高,说明采用双水相提取技术对于提高总黄酮的得率具有一定的优势。
2.3 液相色谱分析
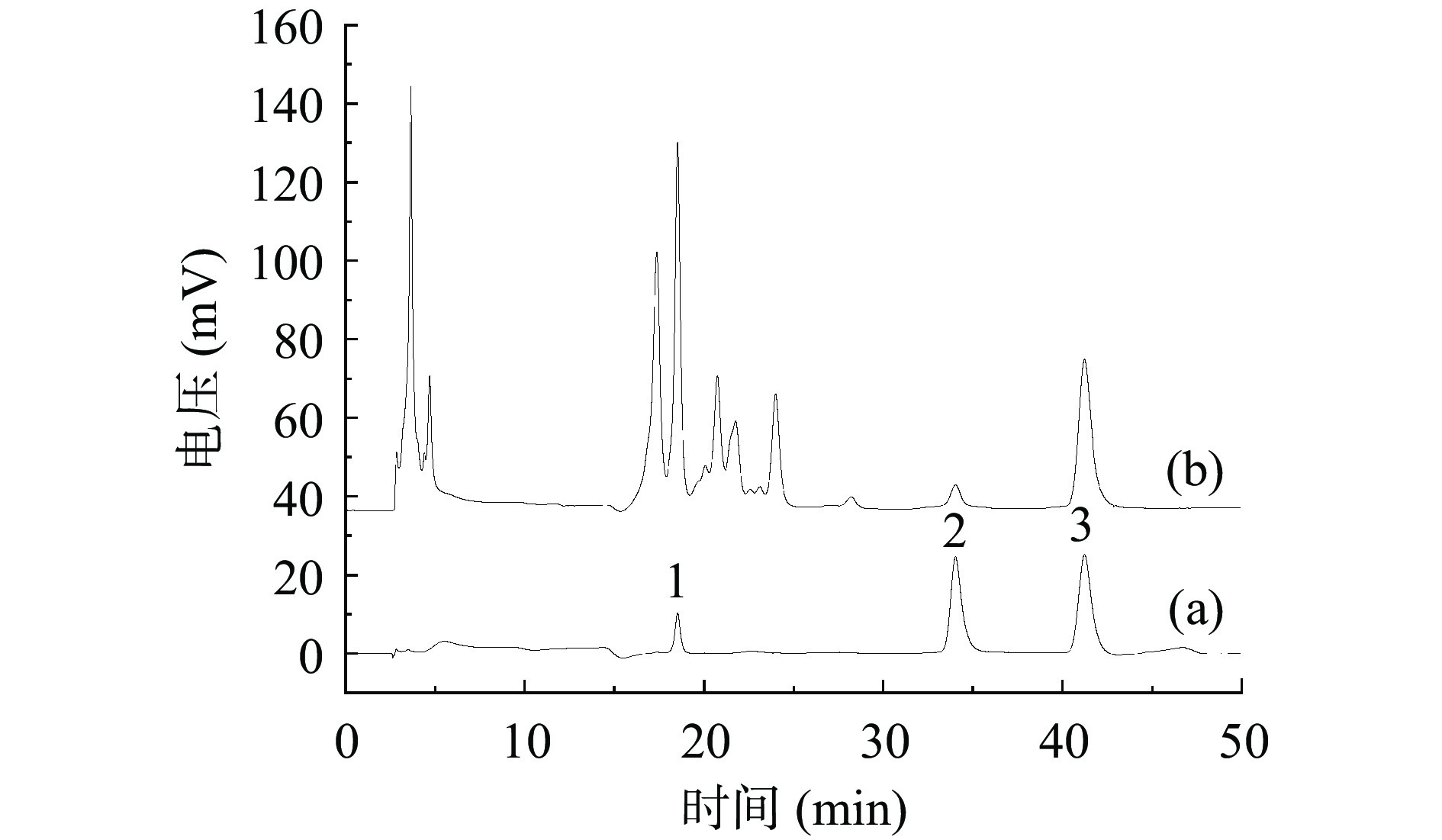
根据文献报道[25-28],木犀草苷、槲皮素和木犀草素为金银花叶黄酮的重要组成成分,其含量是衡量金银花相关产品质量的重要指标。因此,本研究采用HPLC法对金银花叶总黄酮提取物中这三种黄酮化合物进行定性定量分析,如图5所示。标准品木犀草苷、槲皮素和木犀草素的保留时间分别为18.53、34.04和41.23 min。金银花叶总黄酮提取物样品在相应保留时间下也有出峰,且峰型和标准品相似,由此可知,样品中含有这三种黄酮化合物。经定量分析,金银花叶中木犀草苷含量为624.5 μg/g,槲皮素含量为30.8 μg/g,木犀草素含量为247.8 μg/g。
2.4 金银花叶总黄酮的抗氧化性能
2.4.1 DPPH自由基清除效果
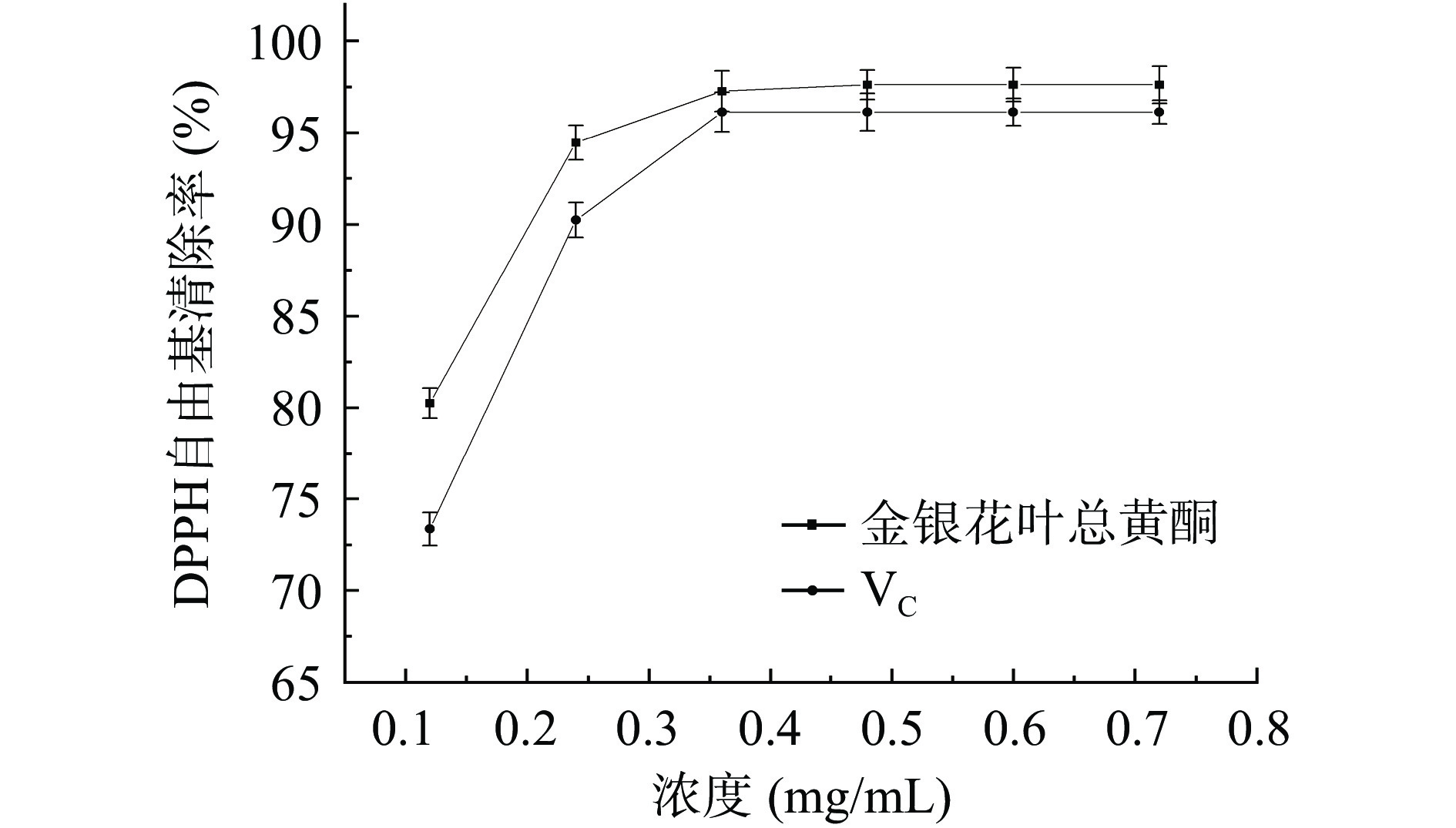
由图6可知,VC对DPPH自由基的清除率随着浓度的增大而增大,当浓度为0.36 mg/mL时,清除率可达96.12%,后进一步增加浓度,清除率未见明显提高。金银花叶总黄酮对DPPH自由基的清除率随样品浓度的增大而增大,在总黄酮浓度为0.48 mg/mL时,清除率为97.61%。采用SPSS 20.0软件分析,VC的IC50值为0.057 mg/mL,金银花叶总黄酮的IC50值为0.052 mg/mL。上述结果表明,金银花叶总黄酮对DPPH自由基具有很强的清除效果,且在相同质量浓度下,金银花叶总黄酮对DPPH自由基的清除率略高于VC,与文献研究黄酮抗氧化的结果相似[29]。
2.4.2 羟基自由基清除效果
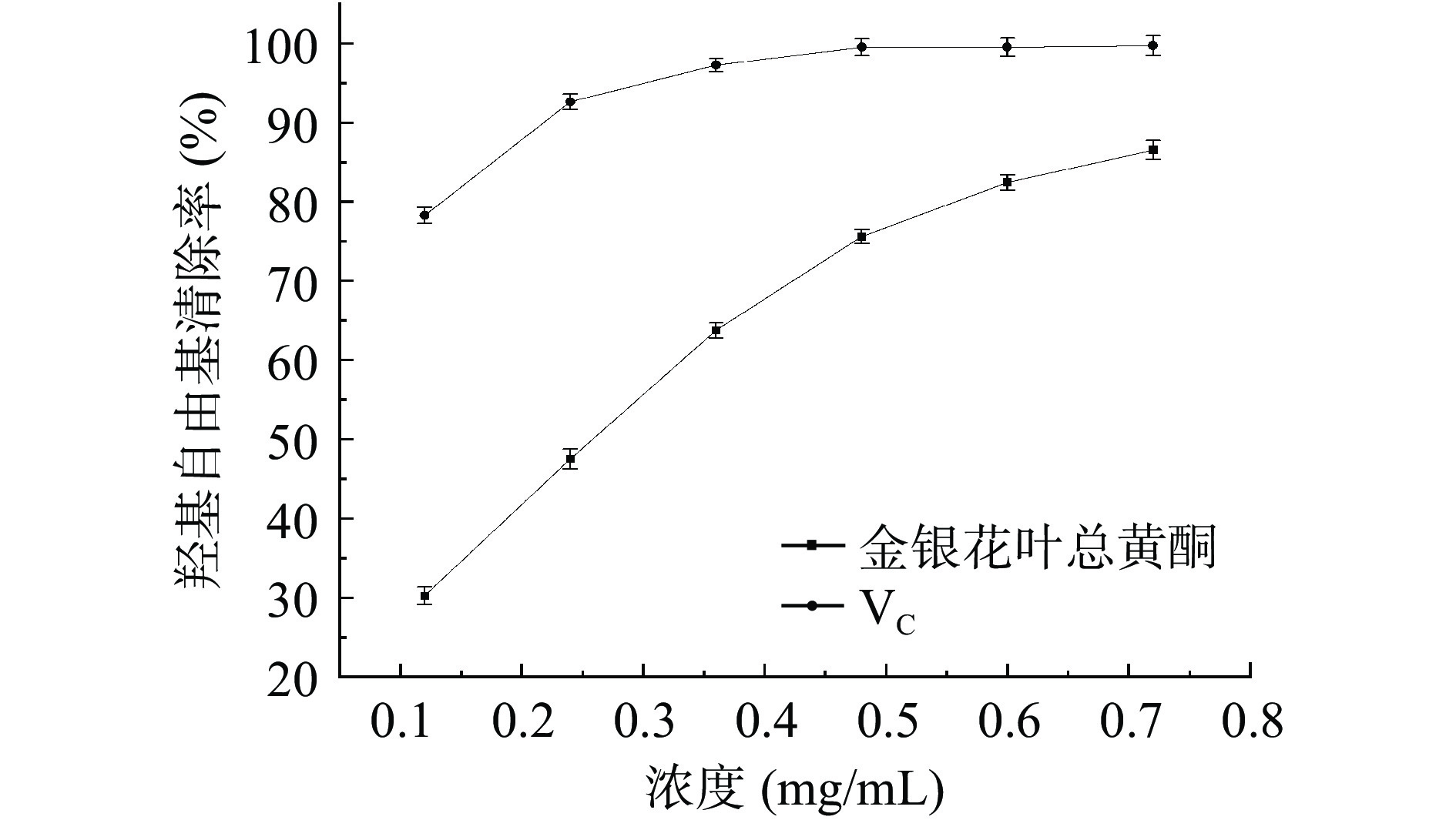
如图7所示,随着浓度的增加,VC对羟基自由基的清除率逐渐增大,当样品质量浓度为0.48 mg/mL时,清除率接近100%。在所测定的浓度范围内,金银花叶总黄酮对羟基自由基的清除率和其浓度呈现出明显的量效关系,在总黄酮浓度为0.72 mg/mL时,清除率为86.53%。采用SPSS 20.0软件分析,VC的IC50值为0.070 mg/mL,金银花叶总黄酮的IC50值为0.231 mg/mL。上述结果表明,金银花叶总黄酮对羟基自由基具有较强的清除作用,与文献研究黄酮抗氧化的结果相似[30]。
2.4.3 超氧阴离子自由基清除效果
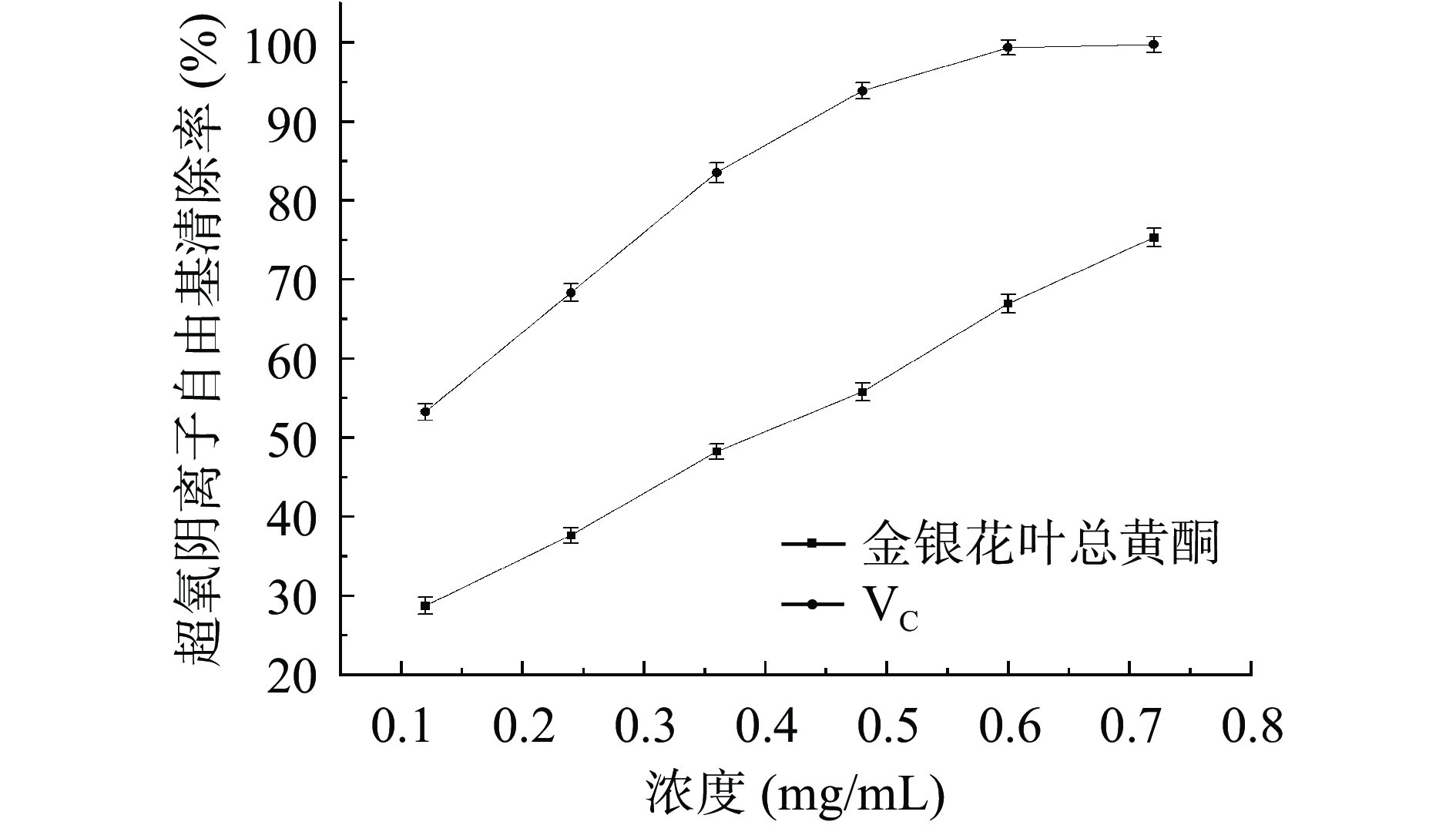
如图8所示,随着浓度的增加,VC对超氧阴离子自由基的清除率逐渐增大,当浓度为0.60 mg/mL时,清除率接近100%。在所测定的浓度范围内,金银花叶总黄酮对超氧阴离子自由基的清除率随着样品浓度的增大而增大,在总黄酮浓度为0.72 mg/mL时,清除率为75.29%。采用SPSS 20.0软件分析,VC的IC50值为0.130 mg/mL,金银花叶总黄酮的IC50值为0.335 mg/mL。上述结果表明,金银花叶黄酮对超氧阴离子自由基具有较好的清除效果, 与文献研究黄酮抗氧化的结论相似[31]。
3. 结论
研究了乙醇-(NH4)2SO4双水相提取金银花叶总黄酮工艺及其抗氧化性能。在单因素实验的基础上,采用正交试验优化了工艺条件。研究结果表明,最佳的工艺参数为(NH4)2SO4质量分数15%,液料比25:1 mL/g,浸提温度50 ℃,浸提时间30 min。在该条件下,重复三次试验,总黄酮得率的平均值为4.98%,相对标准偏差(RSD)为0.5053%,说明该工艺重复性和稳定性都较好。抗氧化实验结果表明,金银花叶总黄酮浓度为0.48 mg/mL时,DPPH自由基的清除率为97.61%;总黄酮浓度为0.72 mg/mL时,羟基自由基的清除率为86.53%,超氧阴离子自由基的清除率为75.29%。双水相提取金银花叶总黄酮具有条件温和、操作简单、效率高等优点,得到的总黄酮表现出良好的抗氧化活性,后续可进一步探索该技术的放大工艺,对黄酮化合物进行分离和纯化,深入研究黄酮组分的抗氧化活性,从而提高金银花叶的综合利用效益。
-
表 1 正交试验因素和水平设计
Table 1 Design of factors and levels of orthogonal experiment
水平 A (NH4)2SO4
质量分数(%)B 液料比
(mL·g−1)C 浸提温度
(℃)D 浸提时间
(min)1 13 25:1 40 20 2 14 30:1 50 30 3 15 35:1 60 40 表 2 正交试验结果
Table 2 Results of orthogonal experiments
编号 A (NH4)2SO4
质量分数(%)B 液料比
(mL·g−1)C 浸提温度
(℃)D 浸提时间
(min)总黄酮得率
(%)1 13 25:1 40 20 3.21 2 13 30:1 50 30 3.81 3 13 35:1 60 40 2.99 4 14 25:1 50 40 4.91 5 14 30:1 60 20 4.70 6 14 35:1 40 30 4.05 7 15 25:1 60 30 4.92 8 15 30:1 40 40 4.52 9 15 35:1 50 20 4.71 k1 3.337 4.347 3.927 4.207 k2 4.553 4.343 4.477 4.260 k3 4.717 3.917 4.203 4.140 R 1.380 0.430 0.550 0.120 -
[1] 杨鹏飞, 熊建华, 杜华英, 等. 金银花叶HPLC指纹图谱及最佳采收期的研究[J]. 江西农业大学学报,2014,36(1):68−75. [YANG P F, XIONG J H, DU H Y, et al. A study on HPLC fingerprint chromatogram and optimum harvest time of Lonicera japonica Thunb. leaves[J]. Acta Agriculturae Universitatis Jiangxiensis,2014,36(1):68−75. doi: 10.3969/j.issn.1000-2286.2014.01.011 YANG P F, XIONG J H, DU H Y, et al. A study on HPLC fingerprint chromatogram and optimum harvest time of Lonicera japonica Thunb. leaves[J]. Acta Agriculturae Universitatis Jiangxiensis, 2014, 36(1): 68-75. doi: 10.3969/j.issn.1000-2286.2014.01.011
[2] 罗磊, 张冰洁, 关宁宁, 等. 金银花叶黄酮对衰老模型小鼠的体内抗氧化作用[J]. 食品科学,2017,38(19):171−176. [LUO L, ZHANG B J, GUAN N N, et al. In vivo antioxidant activity of honeysuckle leaf flavonoids in agingmice[J]. Food Science,2017,38(19):171−176. doi: 10.7506/spkx1002-6630-201719027 LUO L, ZHANG B J, GUAN N N, et al. In vivo antioxidant activity of honeysuckle leaf flavonoids in agingmice[J]. Food Science, 2017, 38(19): 171-176. doi: 10.7506/spkx1002-6630-201719027
[3] 田存章, 贺新平, 曹乐乐, 等. 金银花叶总黄酮不同提取方法比较及其抗氧化活性研究[J]. 生物化工,2021,7(4):53−56. [TIAN C Z, HE X P, CAO L L, et al. Study on extraction methods and antioxidant activity of the total flavonoids from Lonicera japonica[J]. Biological Chemical Engineering,2021,7(4):53−56. doi: 10.3969/j.issn.2096-0387.2021.04.011 TIAN C Z, HE X P, CAO L L, et al. Study on extraction methods and antioxidant activity of the total flavonoids from Lonicera japonica[J]. Biological Chemical Engineering, 2021, 7(4): 53-56. doi: 10.3969/j.issn.2096-0387.2021.04.011
[4] 崔婷婷. 金银花叶中黄酮类物质的分离纯化及应用研究[D]. 泰安: 山东农业大学, 2016 CUI T T. Purification and application of flavonoids in honeysuckle leaves[D]. Taian: Shandong Agricultural University, 2016.
[5] 南海娟, 王正荣, 葛亚明, 等. 金银花与叶醇提物对油脂的抗氧化活性比较[J]. 食品与发酵工业,2017,43(6):175−180. [NAN H J, WANG Z R, GE Y M, et al. Comparison of antioxidant activity of honeysuckle’s flowers and leaves alcohol extract on edible oils and fats[J]. Food and Fermentation Industries,2017,43(6):175−180. doi: 10.13995/j.cnki.11-1802/ts.201706029 NAN H J, WANG Z R, GE Y M, et al. Comparison of antioxidant activity of honeysuckle’s flowers and leaves alcohol extract on edible oils and fats[J]. Food and Fermentation Industries, 2017, 43(6): 175-180. doi: 10.13995/j.cnki.11-1802/ts.201706029
[6] 李康强, 袁诚, 王龙书. 金银花干叶总黄酮提取工艺研究[J]. 广东化工,2016,43(19):35−36. [LI K Q, YUAN C, WANG L S. Study on the optimum extraction conditions of the flavonoids in dehydrated Lonicera japonica leaves[J]. Guangdong Chemical Industry,2016,43(19):35−36. doi: 10.3969/j.issn.1007-1865.2016.19.015 LI K Q, YUAN C, WANG L S. Study on the optimum extraction conditions of the flavonoids in dehydrated Lonicera japonica leaves[J]. Guangdong Chemical Industry, 2016, 43(19): 35-36. doi: 10.3969/j.issn.1007-1865.2016.19.015
[7] 谢威, 王丹, 廖恒康, 等. 复合酶辅助提取金银花叶中黄酮工艺和动力学研究[J]. 湖北农业科学,2013,52(16):3943−3946. [XIE W, WANG D, LIAO H K, et al. Study on the complex enzymes assisted extraction technology of flavonoids from leaves of Lonicera japonica and its kinetics[J]. Hubei Agricultural Sciences,2013,52(16):3943−3946. doi: 10.3969/j.issn.0439-8114.2013.16.048 XIE W, WANG D, LIAO H K, et al. Study on the complex enzymes assisted extraction technology of flavonoids from leaves of Lonicera japonica and its kinetics[J]. Hubei Agricultural Sciences, 2013, 52(16): 3943-3946. doi: 10.3969/j.issn.0439-8114.2013.16.048
[8] CHONG K Y, BROOKS M S L. Effects of recycling on the aqueous two-phase extraction of bioactives from haskap leaves[J]. Separation and Purification Technology,2021,255:117755. doi: 10.1016/j.seppur.2020.117755
[9] 吴丁丁, 穆小静, 易小琦, 等. 双水相萃取技术的新发展[J]. 食品工业科技,2017,38(8):395−400. [WU D D, MU X J, YI X Q, et al. Progress of aqueous two-phase extraction technique[J]. Science and Technology of Food Industry,2017,38(8):395−400. doi: 10.13386/j.issn1002-0306.2017.08.068 WU D D, MU X J, YI X Q, et al. Progress of aqueous two-phase extraction technique[J]. Science and Technology of Food Industry, 2017, 38(8): 395-400. doi: 10.13386/j.issn1002-0306.2017.08.068
[10] 俞耀文, 戴国庆, 华浩立, 等. 乙醇-硫酸铵双水相体系提取桃花总黄酮及其抗氧化性能[J]. 食品工业科技,2022,43(4):187−195. [YU Y W, DAI G Q, HUA H L, et al. Ethanol-ammonium sulfate aqueous two-phase extraction of total flavonoids from peach blossom and its antioxidant activity[J]. Science and Technology of Food Industry,2022,43(4):187−195. YU Y W, DAI G Q, HUA H L, et al. Ethanol-ammonium sulfate aqueous two-phase extraction of total flavonoids from peach blossom and its antioxidant activity[J]. Science and Technology of Food Industry, 2022, 43(4): 187-195.
[11] YANG S X, LIU B, TANG M, et al. Extraction of flavonoids from Cyclocarya paliurus (Juglandaceae) leaves using ethanol/salt aqueous two-phase system coupled with ultrasonic[J]. Journal of Food Processing and Preservation,2020,44(6):e14469.
[12] WANG L T, YANG Q, CUI Q, et al. Recyclable menthol-based deep eutectic solvent micellar system for extracting phytochemicals from Ginkgo biloba leaves[J]. Journal of Cleaner Production,2020,244:118648. doi: 10.1016/j.jclepro.2019.118648
[13] 张敏娜, 朱军, 王光辉, 等. 月季花总黄酮两种提取方法的比较研究[J]. 济宁医学院学报,2012,35(1):18−19, 23. [ZHANG M N, ZHU J, WANG G H, et al. Comparing two kinds of methods for extraction of the total flavonoids from Rosa chinensis[J]. Journal of Jining Medical University,2012,35(1):18−19, 23. doi: 10.3969/j.issn.1000-9760.2012.01.005 ZHANG M N, ZHU J, WANG G H, et al. Comparing two kinds of methods for extraction of the total flavonoids from Rosa chinensis[J]. Journal of Jining Medical University, 2012, 35(1): 18-19, 23. doi: 10.3969/j.issn.1000-9760.2012.01.005
[14] BI P Y, CHANG L, MU Y L, et al. Separation and concentration of baicalin from Scutellaria baicalensis Georgi extract by aqueous two-phase flotation[J]. Separation and Purification Technology,2013,116:454−457. doi: 10.1016/j.seppur.2013.06.024
[15] 姚琴. 小分子醇双水相体系的构建及提取大黄蒽醌类成分的研究[D]. 雅安: 四川农业大学, 2018 YAO Q. Construction of small molecular alcohol aqueous two phase systems and study on the extraction of anthraquinones in Rheum palmatum L.[D]. Ya’an: Sichuan Agriculture University, 2018.
[16] 陈瑛, 王文渊. 微波辅助提取竹叶黄酮的研究[J]. 应用化工,2011,40(4):661−663. [CHEN Y, WANG W Y. Study on extraction of flavones from bamboo leaf by microwave radiation[J]. Applied Chemical Industry,2011,40(4):661−663. doi: 10.3969/j.issn.1671-3206.2011.04.029 CHEN Y, WANG W Y. Study on extraction of flavones from bamboo leaf by microwave radiation[J]. Applied Chemical Industry, 2011, 40(4): 661-663. doi: 10.3969/j.issn.1671-3206.2011.04.029
[17] 刘芳, 谢妍祎, 吴苏喜, 等. 油茶花醇提物的体外抗氧化活性研究[J]. 粮食与油脂,2021,34(5):132−136. [LIU F, XIE Y Y, WU S X, et al. Study on antioxidant activity of ethanolic extract of Camellia oleifera flower in vitro[J]. Cereals & Oils,2021,34(5):132−136. doi: 10.3969/j.issn.1008-9578.2021.05.034 LIU F, XIE Y Y, WU S X, et al. Study on antioxidant activity of ethanolic extract of Camellia oleifera flower in vitro[J]. Cereals & Oils, 2021, 34(5): 132-136. doi: 10.3969/j.issn.1008-9578.2021.05.034
[18] 石玉平, 卢挺, 王永宁. 油菜蜂花粉中黄酮类物质清除羟基自由基的研究[J]. 食品科学,2004,25(11):300−302. [SHI Y P, LU T, WANG Y N. Studies on the hydroxy-group free radical eliminated of flavonoids of Elaeagnus angustifolia L. flowrse[J]. Food Science,2004,25(11):300−302. doi: 10.3321/j.issn:1002-6630.2004.11.079 SHI Y P, LU T, WANG Y N. Studies on the hydroxy-group free radical eliminated of flavonoids of Elaeagnus angustifolia L. flowrse[J]. Food Science, 2004, 25(11): 300-302. doi: 10.3321/j.issn:1002-6630.2004.11.079
[19] 冯娇, 肖海鸿. 不同产地银柴胡黄酮含量及其抗氧化活性研究[J]. 中国食品添加剂,2021,32(5):8−12. [FENG J, XIAO H H. Study on the flavonoids content and antioxidant activity of Stellaria dichotoma var. lanceolata cultivated from different areas[J]. China Food Additives,2021,32(5):8−12. doi: 10.19804/j.issn1006-2513.2021.05.002 FENG J, XIAO H H. Study on the flavonoids content and antioxidant activity of Stellaria dichotoma var. lanceolata cultivated from different areas[J]. China Food Additives, 2021, 32(5): 8-12. doi: 10.19804/j.issn1006-2513.2021.05.002
[20] 陈建福, 胡泽杰, 潘诗玲, 等. 超声波辅助双水相提取胡萝卜叶总黄酮的工艺研究[J]. 河南工业大学学报(自然科学版),2018,39(1):72−77. [CHEN J F, HU Z J, PAN S L, et al. Optimization of ultrasonic-assisted aqueous two-phase extraction of total flavonoids from carrot leaves[J]. Journal of Henan University of Technology (Natural Science Edition),2018,39(1):72−77. doi: 10.3969/j.issn.1673-2383.2018.01.012 CHEN J F, HU Z J, PAN S L, et al. Optimization of ultrasonic-assisted aqueous two-phase extraction of total flavonoids from carrot leaves[J]. Journal of Henan University of Technology (Natural Science Edition), 2018, 39(1): 72-77. doi: 10.3969/j.issn.1673-2383.2018.01.012
[21] 肖连冬, 王莹, 李慧星. 乙醇/磷酸氢二钾双水相体系提取洋葱黄酮工艺条件研究[J]. 食品研究与开发,2020,41(8):160−165. [XIAO L D, WANG Y, LI H X. Study on the extraction of flavone from onion by ethanol/dipotassium hydrogen phosphate two-phase aqueous system[J]. Food Research and Development,2020,41(8):160−165. doi: 10.12161/j.issn.1005-6521.2020.08.026 XIAO L D, WANG Y, LI H X. Study on the extraction of flavone from onion by ethanol/dipotassium hydrogen phosphate two-phase aqueous system[J]. Food Research and Development, 2020, 41(8): 160-165. doi: 10.12161/j.issn.1005-6521.2020.08.026
[22] 郭莹, 周颖, 毕海丹, 等. 响应面优化超声波-微波协同提取凤眼莲黄酮工艺及其不同部位黄酮抗氧化活性分析[J]. 食品工业科技,2019,40(1):168−174,180. [GUO Y, ZHOU Y, BI H D, et al. Optimization of ultrasonic-microwave synergistic extraction of flavonoids from Eichhornia crassipes by response surface methodology and analysis of antioxidant activity of flavonoids from its different parts[J]. Science and Technology of Food Industry,2019,40(1):168−174,180. doi: 10.13386/j.issn1002-0306.2019.01.031 GUO Y, ZHOU Y, BI H D, et al. Optimization of ultrasonic-microwave synergistic extraction of flavonoids from Eichhornia crassipes by response surface methodology and analysis of antioxidant activity of flavonoids from its different parts[J]. Science and Technology of Food Industry, 2019, 40(1): 168-174, 180. doi: 10.13386/j.issn1002-0306.2019.01.031
[23] 玉澜, 黄宏清, 吴承武, 等. 超声波辅助双水相提取枇杷叶中的黄酮及其抗氧化研究[J]. 饲料研究,2022,45(1):67−71. [YU L, HUANG H Q, WU C W, et al. Ultrasonic-assisted two-phase extraction of flavonoids from loquat leaves and their antioxidant activity[J]. Feed Research,2022,45(1):67−71. YU L, HUANG H Q, WU C W, et al. Ultrasonic-assisted two-phase extraction of flavonoids from loquat leaves and their antioxidant activity[J]. Feed Research, 2022, 45(1): 67-71.
[24] 刘子瑄, 孔馨逸, 陈璐, 等. 汉麻叶黄酮提取工艺及抗氧化活性研究[J]. 中国调味品,2022,47(8):187−190. [LIU Z X, KONG X Y, CHEN L, et al. Study on extraction process and antioxidant activity of flavonoids from Cannabis sativa leaves[J]. China Condiment,2022,47(8):187−190. doi: 10.3969/j.issn.1000-9973.2022.08.037 LIU Z X, KONG X Y, CHEN L, et al. Study on extraction process and antioxidant activity of flavonoids from Cannabis sativa leaves[J]. China Condiment, 2022, 47(8): 187-190. doi: 10.3969/j.issn.1000-9973.2022.08.037
[25] 杨敏丽, 王丽婷. 金银花不同品种及不同部位木犀草素含量的比较[J]. 中华中医药杂志,2007,22(10):666−668. [YANG M L, WANG L T. Compare of luteolin content in different types and different parts of Lonicare japonica[J]. China Journal of Traditional Chinese Medicine and Pharmacy,2007,22(10):666−668. YANG M L, WANG L T. Compare of luteolin content in different types and different parts of Lonicare japonica[J]. China Journal of Traditional Chinese Medicine and Pharmacy, 2007, 22(10): 666-668.
[26] 黄麟杰, 曾爱国. HPLC-DAD法测定“武当二号金银花”、“武当三号金银花”叶中木犀草素的含量[J]. 中南药学,2017,15(1):107−111. [HUANG L J, ZENG A G. Determination of content of luteolin in Wudang Ⅱ Flos lonicerae and Wudang Ⅲ Flos lonicerae leaves by HPLC-DAD[J]. Central South Pharmacy,2017,15(1):107−111. doi: 10.7539/j.issn.1672-2981.2017.01.0027 HUANG L J, ZENG A G. Determination of content of luteolin in Wudang Ⅱ Flos lonicerae and Wudang Ⅲ Flos lonicerae leaves by HPLC-DAD[J]. Central South Pharmacy, 2017, 15(1): 107-111. doi: 10.7539/j.issn.1672-2981.2017.01.0027
[27] 李鹏, 郑芳, 李聪, 等. HPLC-DAD法同时测定“武当二号”忍冬叶中绿原酸和木犀草苷的含量[J]. 医药导报,2015,34(5):660−663. [LI P, ZHENG F, LI C, et al. Simultaneous determination of chlorogenic acid and luteoloside in the leaves of “Wudang No. Ⅱ” Flos lonicerae by HPLC-DAD[J]. Herald of Medicine,2015,34(5):660−663. doi: 10.3870/yydb.2015.05.025 LI P, ZHENG F, LI C, et al. Simultaneous determination of chlorogenic acid and luteoloside in the leaves of “Wudang No. Ⅱ” Flos lonicerae by HPLC-DAD[J]. Herald of Medicine, 2015, 34(5): 660-663. doi: 10.3870/yydb.2015.05.025
[28] 王丽婷, 杨敏丽. 高效液相色谱法同时测定金银花及叶中的黄酮类物质[J]. 时珍国医国药,2007,18(8):1850−1851. [WANG L T, YANG M L. Simultaneous determination of flavonoids compounds in Lonicera japonica and its leaf by high performance liquid chromatography[J]. Lishizhen Medicine and Materia Medica,2007,18(8):1850−1851. doi: 10.3969/j.issn.1008-0805.2007.08.026 WANG L T, YANG M L. Simultaneous determination of flavonoids compounds in Lonicera japonica and its leaf by high performance liquid chromatography[J]. Lishizhen Medicine and Materia Medica, 2007, 18(8): 1850-1851. doi: 10.3969/j.issn.1008-0805.2007.08.026
[29] 秦晶晶, 钱慧琴, 魏婧, 等. 菟丝子总黄酮提取工艺及其抗氧化活性[J]. 食品工业科技,2019,40(23):151−157. [QIN J J, QIAN H Q, WEI J, et al. Extraction process of total flavonoids from Cuscuta chinesis and its antioxidant activity[J]. Science and Technology of Food Industry,2019,40(23):151−157. doi: 10.13386/j.issn1002-0306.2019.23.025 QIN J J, QIAN H Q, WEI J, et al. Extraction process of total flavonoids from Cuscuta chinesis and its antioxidant activity[J]. Science and Technology of Food Industry, 2019, 40(23): 151-157. doi: 10.13386/j.issn1002-0306.2019.23.025
[30] 张小梅, 王聪, 靳晓琳, 等. 百香果皮总黄酮的复合酶辅助超声波提取工艺优化及其抗氧化活性分析[J]. 食品工业科技,2022,43(12):215−222. [ZHANG X M, WANG C, JIN X L, et al. Optimization of extraction technology of total flavonoids from Passiflora edulis peel by ultrasonic assisted with complex enzyme and its antioxidant activity[J]. Science and Technology of Food Industry,2022,43(12):215−222. ZHANG X M, WANG C, JIN X L, et al. Optimization of extraction technology of total flavonoids from Passiflora edulis peel by ultrasonic assisted with complex enzyme and its antioxidant activity[J]. Science and Technology of Food Industry, 2022, 43(12): 215-222.
[31] 杨宗玲, 李晗, 范方宇, 等. 超声辅助酶法提取无籽刺梨果渣中黄酮的工艺优化及其抗氧化活性[J]. 食品工业科技,2021,42(13):184−192. [YANG Z L, LI H, FAN F Y, et al. Ultrasound-assisted enzymatic extraction of flavonoids from Rosa sterilis Pomace and its antioxidant activity[J]. Science and Technology of Food Industry,2021,42(13):184−192. doi: 10.13386/j.issn1002-0306.2020090273 YANG Z L, LI H, FAN F Y, et al. Ultrasound-assisted enzymatic extraction of flavonoids from Rosa sterilis Pomace and its antioxidant activity[J]. Science and Technology of Food Industry, 2021, 42(13): 184-192. doi: 10.13386/j.issn1002-0306.2020090273
-
期刊类型引用(0)
其他类型引用(1)





 下载:
下载:



 下载:
下载:



