Determination of Sixteen Kinds of Free Amino Acids in Citrus by Liquid Liquid Microextraction/Solid Phase Microextraction Coupled with Gas Chromatography
-
摘要: 目的:建立气相色谱氢火焰离子检测法同时测定柑橘果肉中16种游离氨基酸含量。方法:以对氯甲酸异丁酯为衍生剂,先进行衍生条件优化,再采用自制的溶胶-凝胶甲基丙烯酸丁酯二乙烯基苯涂层为萃取头,通过液液微萃取联合固相微萃取对样品进行前处理后上气相色谱仪检测。结果:对氯甲酸异丁酯衍生化柑橘中游离氨基酸采用一次性衍生法,衍生时间1 min。优化后的萃取温度为60 ℃,萃取时间30 min,离子强度为30% Na2SO4,搅拌速度1000 r·min−1。在此条件下,目标化合物具有良好的线性关系,相关系数均大于0.99,检出限为0.85~12.60 μg·kg−1,相对标准偏差为2.51%~12.80%,三种加标浓度下回收率为82.51%~121.75%。结论:该方法操作简单、结果准确、受基质干扰少,适用于柑橘中多种游离氨基酸的检测,并可以游离氨基酸含量来评价柑橘内在品质。
-
关键词:
- 柑橘 /
- 游离氨基酸 /
- 液液微萃取(LLME) /
- 固相微萃取(SPME) /
- 气相色谱(GC)
Abstract: Objective: To evaluate the quality of the citrus, a gas chromatography (GC) coupled with flame ion detector was established for the determination of sixteen kinds of free amino acids in citrus simultaneously. Methods: Firstly, the single factor tests werre used to optimize the derivatization conditions of isobutyl chloroformate. Subsequently, a method was developed by liquid-liquid microextraction (LLME)/solid phase microextraction (SPME) coupled with GC using a home-made sol-gel butylmethacrylate/divinylbenzene (BMA/DVB) fibers. Results: The results showed that the derivatization conditions were the best under one derivation with one min. The best SPME conditions can be obtained as follows: The extraction temperature of 40 ℃, the extraction time of 30 min and the 30% sodium sulfate at the extraction speed of 1000 r·min−1. Under the optimized experimental parameters, there were good linear relationships for 16 analytes with correlation coefficients larger than 0.99. The limit of detection was 0.85~12.60 μg·kg−1 and the standard recoveries of the spiked samples ranged from 82.51% to 121.75%, with relative standard deviations of 2.51%~12.80%. Conclusion: The method was convenient, accurate, and matrix resistant. It could be used as a new model for the determination of the free amino acids in citrus pulp samples and can also provide a valuable reference for the quality evaluation of cirtrus. -
柑橘是世界第一大水果,在中国乃至世界范围内被广泛种植和食用[1]。柑橘果实富含氨基酸、矿物质、维生素、膳食纤维、酚类、萜类等多种人体所需要的营养成分和活性物质,具有重要的营养和保健作用[2]。目前,国内外关于柑橘的研究主要集中在栽培技术、病虫害防治、贮存保鲜、抗氧化成分等方面,对于其果品营养学的研究还不够深入。氨基酸作为人体必需营养成分之一,参与体内众多生命活动,其含量和丰富度已经成为柑橘内在品质和深加工的重要指标[3-4]。
目前果蔬中氨基酸含量测定的方法有很多种,主要包括了高效液相色谱法(high performance liquid chromatography,HPLC)[5-6]、氨基酸自动分析仪[7]、液相色谱-质谱(mass spectrometry,MS)联用法[8]、毛细管电泳法[9]、气相色谱(gas chromatography,GC)法[10]和气相色谱-质谱联用法[11]等。不同的测定方法各有利弊,GB 5009.124-2016《食品安全国家标准 食品中的氨基酸氨基酸的测定》[12]采用的是氨基酸分析仪,该法适用范围广,样品用量小,但分析时间长,受仪器型号和实验条件影响较明显。液相色谱、质谱等仪器设备灵敏度高,易于自动化,但价格昂贵,对分析条件要求严格,难以普及。相比较而言,GC检测法具有衍生物稳定、柱效高、运行成本低等优点也越来越受到关注。氨基酸结构中存在极性较强的氨基、羰基和羟基基团,直接进行GC分析时会导致不出峰、峰形拖尾、灵敏度低等问题,因此一般需要先进行衍生化,降低氨基酸的极性满足检测的需要。受到样品复杂基质干扰,无论采用何种方法进行氨基酸定量检测,都需要经过必要的样品前处理步骤,而且前处理的方式要和检测分析技术相兼容。从文献报道来看,气相色谱法检测样品中氨基酸的前处理方式主要有固相萃取[13]、液液萃取[14]和液相-固相萃取联用[15],这些方法一般需要多次重复操作,使用大量的溶剂。
固相微萃取(solid phase microextraction,SPME)是由Arthur和Pawliszyn发明的一种无溶剂萃取技术,集采样、萃取、浓缩和进样于一体,被广泛应用于各类复杂基质目标物分析[16-18]。该技术具有快速、便携、易于操作等诸多优点,和GC、HPLC等仪器联用灵敏度高、选择性强、重现性好。Deng等[19]报道了一种顶空固相微萃取联合GC-MS检测氨基酸的方法,但是只能分析少量的5种氨基酸,这是因为对于一些挥发性较低或极性较大的分析物,更适合直接固相微萃取(Direct SPME,DI-SPME)。Mudiam等[20]采用DI-SPME/GC-MS法建立了大豆中20种氨基酸的检测方法,此方法检出限仅为0.18~5.62 μg L−1,可能是受到SPME萃取涂层的影响,目前商用的萃取涂层并不多,在复杂基质中直接萃取,涂层容易吸附固体杂质或大分子物质,使得涂层表面变脏,最终对涂层造成不可逆的破坏,影响到萃取效率和使用寿命。近年来,已有学者采用分子印迹[21]、溶胶-凝胶[22]、碳纳米材料[23]、离子液体[24]等新技术、新材料制备了各种抗干扰性强,使用寿命长的新涂层,大大延伸了SPME应用范围。
本研究拟采用课题组自制的溶胶-凝胶BMA-DVB萃取头,该涂层在前期研究中证明具有较好的基质相容性和萃取容量[25]。为降低基质的干扰,提高涂层的使用性能和寿命,提升方法的灵敏度,在DI-SPME前,先通过液液微萃取(liquid-liquid microextraction,LLME)方式对目标物进行富集。基于前期氨基酸衍生化试验基础[10],采用氯甲酸异丁酯一次衍生,建立了一种柑橘果肉中游离氨基酸检测的LLME/DI-SPME-GC法,旨在为柑橘内在品质评价和氨基酸检测提供参考依据。
1. 材料与方法
1.1 材料与仪器
氨基酸标品:谷氨酸(Glu)、赖氨酸(Lys)、丝氨酸(Ser)、天冬氨酸(Asp)、丙氨酸(Ala)、苯丙氨酸(Phe)、酪氨酸(Tyr)、苏氨酸(Thr)、亮氨酸(Leu)、异亮氨酸(Ile)、缬氨酸(Val)、色氨酸(Trp)、甘氨酸(Gly)、半胱氨酸(Cys)、甲硫氨酸(Met)、组氨酸(His)、脯氨酸(Pro)、天冬酰胺(Asn) 纯度均≥98.5%,Biosharp生物科技公司;对氯甲酸异丁酯(isobutyl chloroformate, IBCF) 分析纯,阿拉丁试剂公司;硝酸、浓盐酸、氢氧化钠、二氯甲烷、吡啶、异丁醇、无水硫酸钠 分析纯,国药集团化学试剂有限公司。柑橘(伦晚橙)样品 购自当地农贸市场。
SP-7890型气相色谱仪(配置氢火焰离子化检测器,FID) 山东鲁南瑞虹化工有限公司;N2000色谱数据工作站 浙江大学智达信息工程有限公司;SE-54毛细管柱(30m×0.32 mm i.d.×0.25 μm) 兰州中科安泰分析科技有限公司;CT-1型氮氢空气发生器 武汉科林普丰仪器有限公司;TGL-16型离心机 上海安亭仪器有限公司;KQ-300DE型数控超声波清洗器 昆山市超声仪器有限公司;QL-861型漩涡混合器 江苏海门其林贝尔仪器制造有限公司;DF-101型集热式恒温加热磁力搅拌器 武汉德力祥仪器设备有限公司;JYL-C022型料理机 九阳股份有限公司;微量进样器 上海高鸽工贸有限公司;65 μm溶胶-凝胶BMA-DVB萃取头 实验室自制。
1.2 实验方法
1.2.1 氨基酸标准溶液的制备
分别称取10 mg 氨基酸标准品于10 mL容量瓶中,用0.1 mol·L−1 HCl溶液溶解,配制成1 mg·mL−1的一级内标混合储备液,放置4 ℃冰箱储存。使用时再用上述HCl溶液逐级稀释成所需要的标准工作液。
1.2.2 氨基酸标准溶液衍生处理
一次衍生:先在4 mL离心管中加入1 mL 10 μg·mL−1 混合标准溶液,然后加入7 mol·L−1氢氧化钠溶液调节pH至9,再依次加入20 μL吡啶、120 μL 异丁醇、100 μL IBCF,涡旋1 min后加入1 mL二氯甲烷,继续涡旋1 min,静置3 min,于3000 r·min−1下离心5 min,取下层分层溶液置于-18 ℃下冷冻,待残留水冷冻后快速取出液体状有机相,在50 ℃下氮气吹干,最后用50 μL二氯甲烷复溶。
两次衍生:先在4 mL离心管中加入1 mL 10 μg mL−1 混合标准溶液,然后加入20 μL吡啶,120 μL 异丁醇,50 μL IBCF,超声2 min,再加入500 μL二氯甲烷,涡旋1 min,静置3 min,于3000 r·min−1下离心5 min;然后向上层分层溶液加入7 mol·L−1氢氧化钠溶液调节pH至9,再加入50 μL IBCF,超声2 min后加入500 μL二氯甲烷,涡旋1 min,静置3 min,后续离心、冷冻、吹干、复溶等程序与一次衍生一致。
1.2.3 柑橘中游离氨基酸溶液制备与衍生
将去皮后的柑橘果肉放在匀浆机中高速匀浆两次,称取10 g匀浆液置于100 mL的离心管中,依次加入谷氨酸、甘氨酸、缬氨酸、亮氨酸、异亮氨酸、组氨酸、色氨酸、甲硫氨酸、半胱氨酸、 赖氨酸、酪氨酸标准溶液,添加量均为10 μg·g−1,其余氨基酸含量较高,未添加。涡旋5 min,置于4 ℃冰箱静置12 h后取出,按照1:2料液比加入去离子水涡旋5 min混匀,在240 W、25 ℃条件下超声提取20 min。随后将提取液涡旋混匀,取出1.0 g置于4 mL离心管中,按照1.4.2一次衍生方法衍生,衍生化的待测样品加入2 mL二氧化氯涡旋萃取1 min,5000 r·min−1下离心5 min,取下层溶液置于−18 ℃下保存备用。
1.2.4 色谱条件
以超纯氮气为载体,柱头压0.055 MPa,空气0.095 MPa,氢气0.065 MPa,尾吹气0.075 MPa,分流进样,分流比为20:1。进样口温度280 ℃,检测器温度300 ℃。升温程序:初始温度100 ℃,以8 ℃·min−1升至200 ℃,以6 ℃·min−1升至300 ℃,保留2 min。
1.2.5 LLME和DI-SPME操作步骤
量取600 μL衍生化后的萃取液于4 mL萃取瓶中,加入2 mL去离子水,磁子,放置在70 ℃水浴中磁力搅拌3 min,待溶剂挥发后加入0.6 g无水Na2SO4,加盖密封,将自制SPME装置插入萃取瓶中,推出萃取头,60 ℃,1000 r·min−1磁力搅拌萃取30 min,萃取完成后用无尘纸拭干,随后在GC进样口解吸10 min,解吸后用1:1甲醇/丙醇混合溶剂清洗2 min,拭干后在进样口解吸5 min除去残留溶剂,以待下次使用。
1.2.6 方法评价
在空白柑橘匀浆中加入内标2-氨基丁酸(用于脯氨酸定量,样品中浓度为6 μg·g−1)和一系列浓度的氨基酸混合标准溶液,使样品中氨基酸浓度为0、0.001、0.01、0.1、1、10、20、100 μg·g−1,放在4 ℃冰箱静置12 h,让氨基酸与基质充分结合。按照1.2.3和1.2.5的步骤进行衍生和萃取。
以氨基酸加标浓度为横坐标,峰面积为纵坐标建立标准工作曲线,获得线性方程。检出限LOD根据三倍信噪比(3S/N)计算得来,定量限LOQ为10倍信噪比(10S/N),方法RSD%为0.1 μg·g−1加标浓度下平行测定5次结果。
1.2.7 加标回收试验与真实样品检测
按照1.2.6的加标方法和步骤,选取0.1、1.0和100 μg·g−1三个加标浓度制备的样品,在优化后的条件下,重复测定3次取平均值,根据测得值与加入量计算方法的加标回收率。根据建立的标准曲线可知,曲线与横坐标的交点的绝对值即为真实样品中游离氨基酸的含量。
1.3 数据处理
本文图表中数据表示形式为平均值±标准差或平均值,图采用origin 2018进行绘制,差异性显著分析采用SPSS 22.0进行单因素方差分析。
2. 结果与分析
2.1 氨基酸衍生化条件优化
2.1.1 氨基酸一次衍生与二次衍生物含量比较
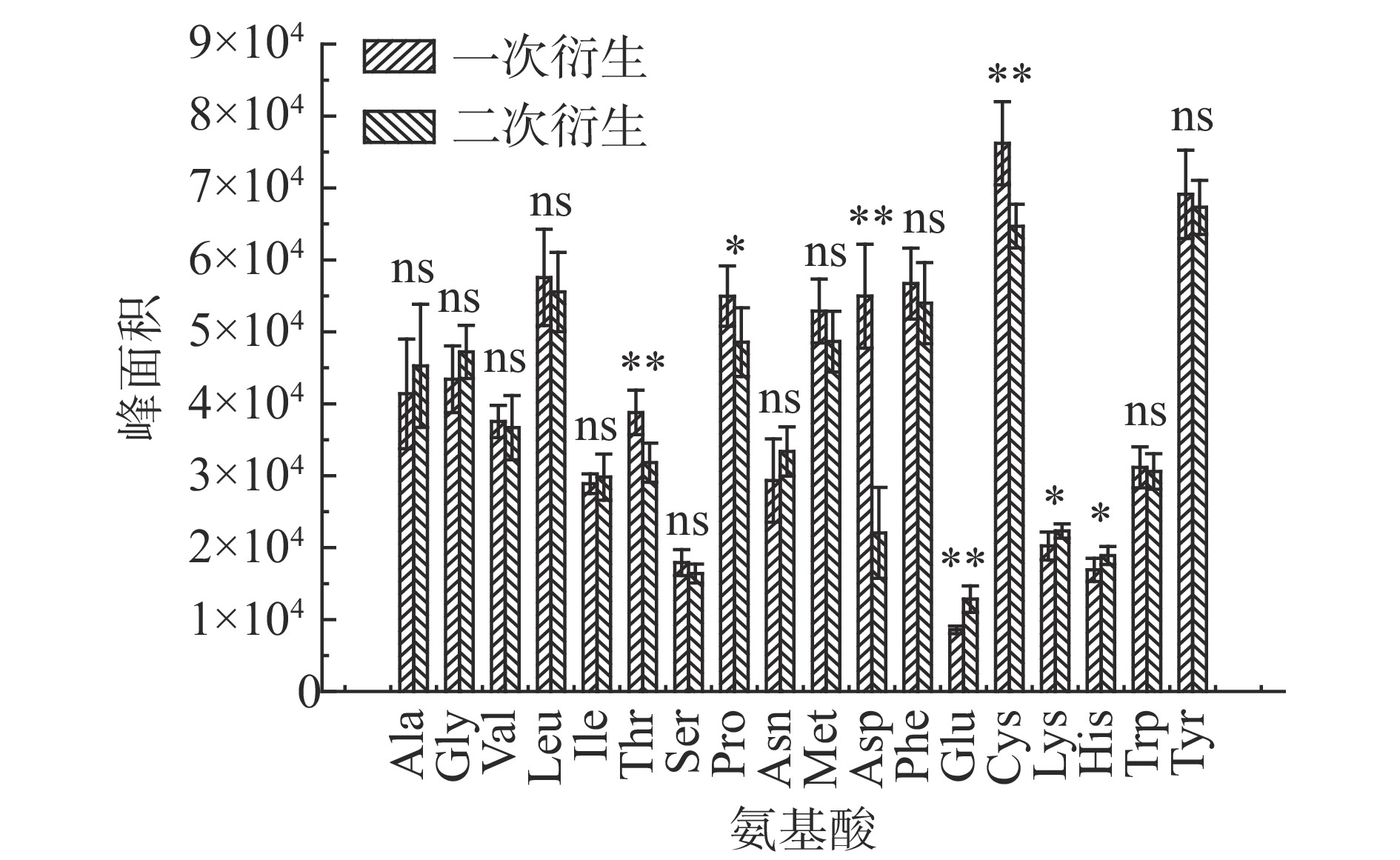
氨基酸结构中含有氨基、羧基和羟基等极性基团,在色谱检测前一般需要进行相应的衍生化处理[26]。为简化衍生程序,本研究在二次衍生基础上,进一步比较一次衍生和两次衍生法氨基酸衍生物含量变化,结果如图1所示。丙氨酸、甘氨酸、异亮氨酸、天冬酰胺、谷氨酸和赖氨酸6种氨基酸两次衍生物含量要高于一次衍生,其他12种氨基酸一次衍生高于两次衍生。通过方差进一步分析苏氨酸、天冬氨酸、谷氨酸和半胱氨酸衍生次数与衍生物含量存在极显著性差异,脯氨酸、赖氨酸和组氨酸存在显著性差异,其余氨基酸衍生次数对衍生物含量的影响不显著。说明衍生物含量跟衍生次数或者调节pH值的先后顺序关联不强因此,后续试验均采取一次衍生。
2.1.2 涡旋衍生时间比较
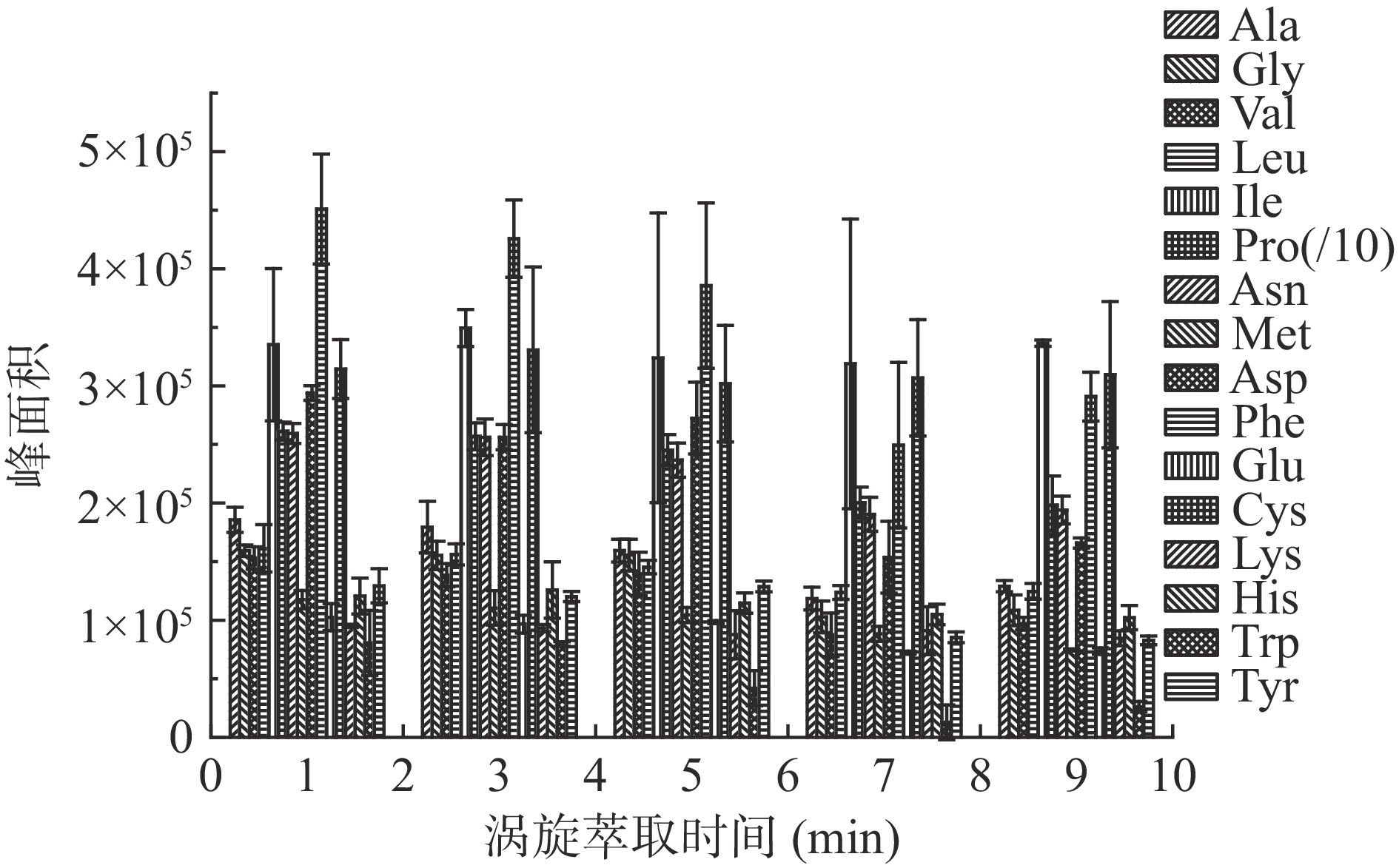
涡旋衍生时间对氨基酸衍生化结果也至关重要,图2比较了不同反应时间下柑橘果肉提取液中游离氨基酸衍生物含量,衍生反应时间1~3 min内,GC检测各氨基酸峰面积无明显变化,随着涡旋时间继续增加,部分氨基酸衍生物峰面积开始降低,尤其到第10 min时,酪氨酸、色氨酸、赖氨酸、半胱氨酸、苯丙氨酸、甲硫氨酸、天冬氨酸、脯氨酸、亮氨酸、甘氨酸等氨基酸衍生物峰面积大幅度降低,故本研究最终选择衍生化时间为1 min。
2.2 萃取条件优化
2.2.1 LLME条件优化
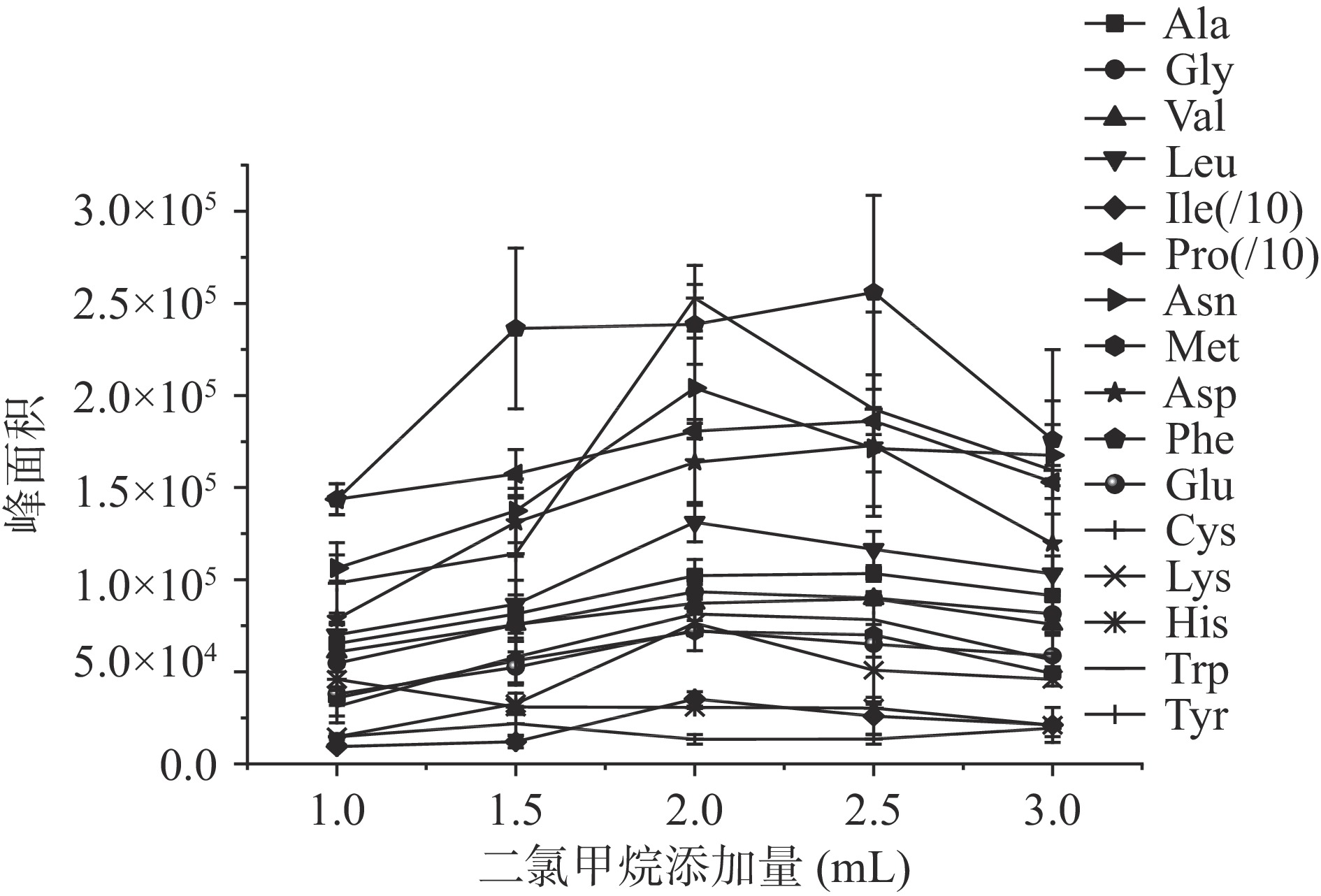
图3和图4分别考察了二氯甲烷添加量和涡旋萃取时间对氨基酸衍生物萃取量的影响。从图3结果可知,色氨酸最大峰面积出现在二氯甲烷添加量为1.5 mL,苯丙氨酸衍生物最大峰值出现在2.5 mL,缬氨酸、脯氨酸和天冬氨酸在加入量2.0和2.5 mL下萃取量差异不大,而其他11种氨基酸衍生物在二氯甲烷添加量为2 mL时萃取量达到最大量。继续加大二氯甲烷添加量,绝大部分衍生物的萃取量开始降低,可能是溶剂量太大后造成样品稀释,影响到衍生物的萃取,最终确定二氯甲烷添加量为2.0 mL。
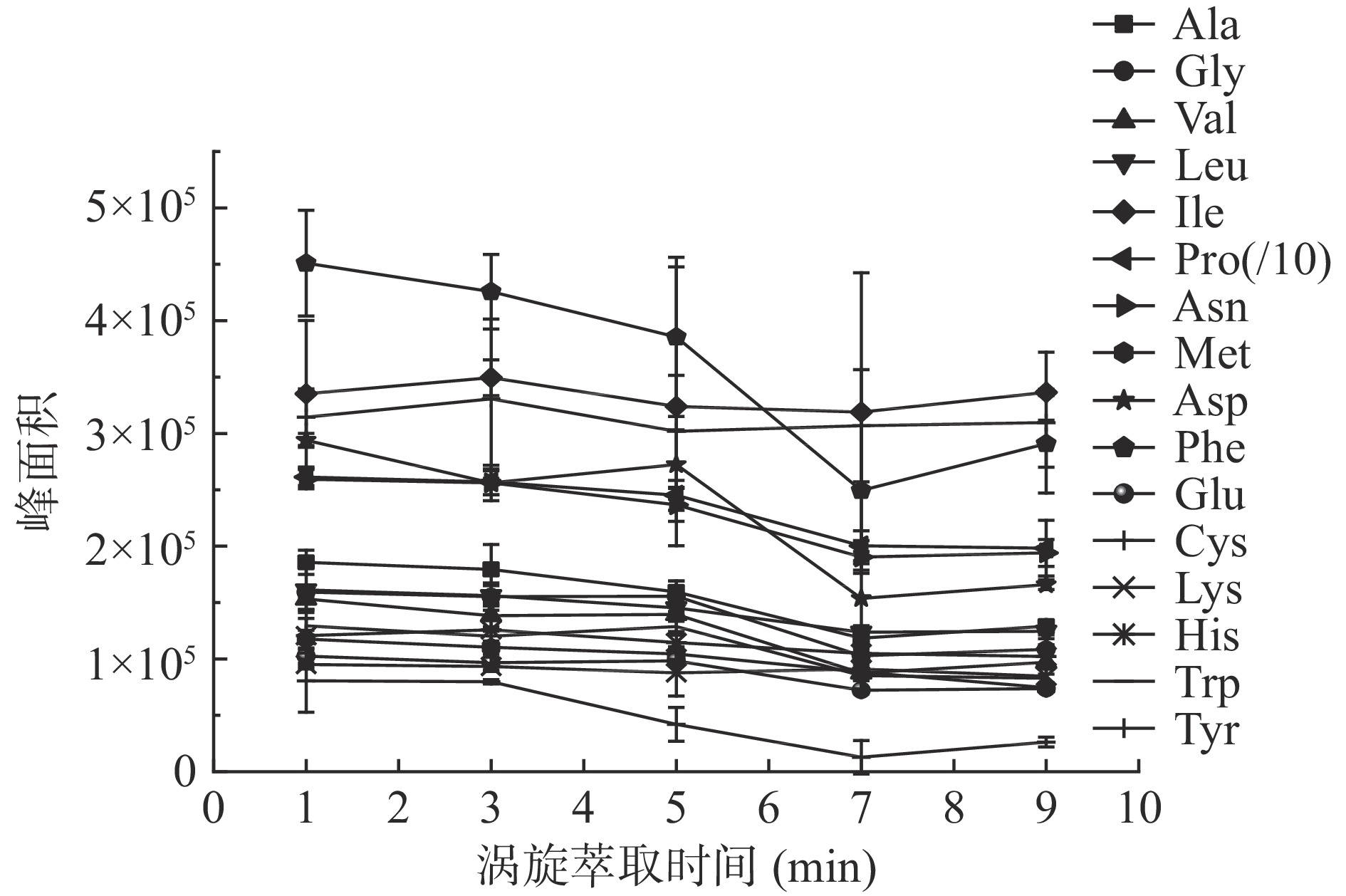
根据图4结果,除异亮氨酸、半胱氨酸和组氨酸在涡旋3 min时萃取峰面积最大,其他氨基酸衍生物在涡旋萃取时间为1 min时,检测到的峰面积出现最大值,综合考虑,涡旋萃取时间为1 min。
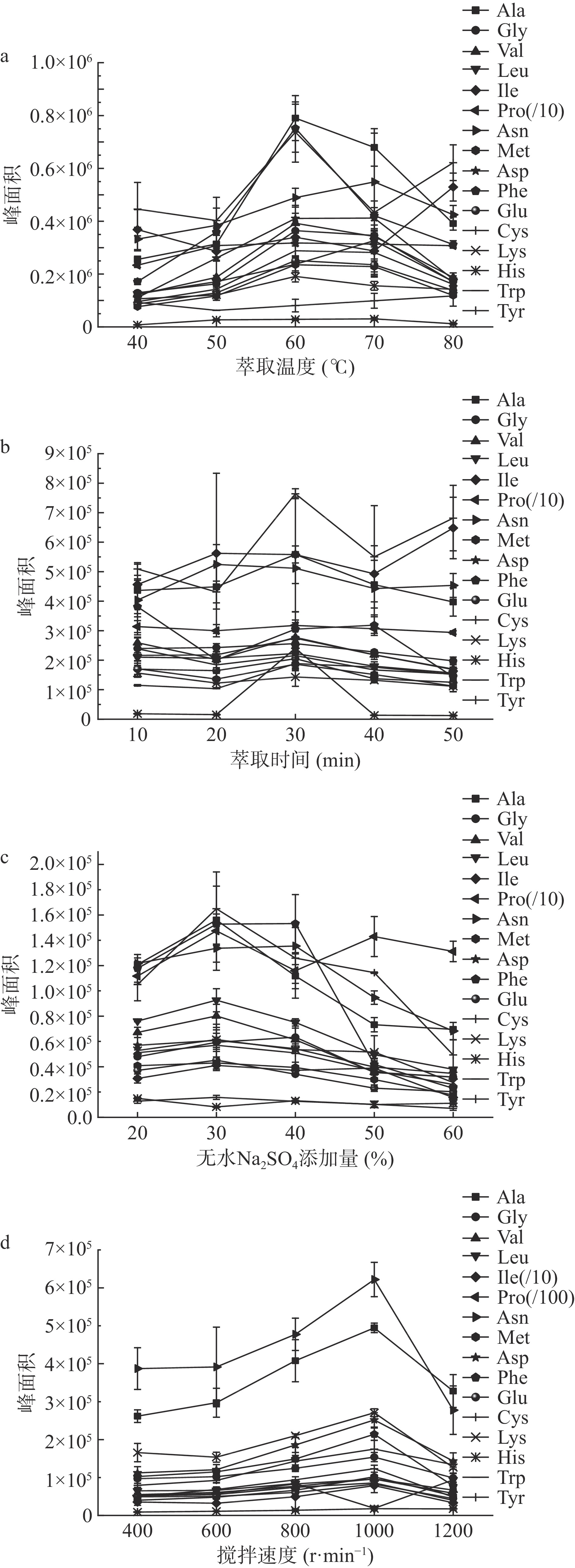
2.2.2 DI-SPME条件优化
图5分别考察了萃取温度、萃取时间、离子强度和搅拌速度等影响SPME萃取效率的因素。
实验选取了40、50、60、70、80 ℃五个梯度,考察萃取温度的影响,如图5a。随着温度升高,待测物含量呈现先升高后降低的趋势,其中亮氨酸和天冬酰胺在70 ℃,异亮氨酸在80 ℃时萃取量最大,其他氨基酸衍生物在60 ℃时达到最大,故将萃取温度确定为60 ℃。这可能是因为萃取温度升高,溶液中分析物的扩散速度会加快,增加涂层对氨基酸衍生物的吸附量,但是萃取也是一个放热过程,温度过高反而会减小分析物在涂层和基质之间的分配系数,导致吸附量下降。
图5b比较了10~50 min萃取时间下目标物萃取量的变化。结果表明,大部分氨基酸衍生物的萃取量随着萃取时间的延长而增大,其中苯丙氨酸、赖氨酸和酪氨酸在10 min时就达到最大值,天冬酰胺在20 min时峰面积最大,异亮氨酸衍生物在50 min时才出现最大萃取量,其他氨基酸衍生物在萃取时间为30 min时,萃取效率最高,因此选择30 min作为后续试验萃取时间。究其原因,SPME实质是一个富集平衡的过程,萃取头对待测物吸附的初始时是一段快速吸附期,随着目标物富集程度增加而进入一个平缓期,最终达到饱和状态。
本研究选用无水Na2SO4来考察溶液离子强度对萃取量的影响,见图5c,在添加量20%~60%范围内,除组氨酸和脯氨酸外,其他氨基酸衍生物萃取量均随着离子浓度增大先增加,添加量为30%时,达到最大,继续加大添加量,萃取量开始减少,可能是因为过量的无水Na2SO4增大了溶液的粘度,影响目标物扩散的速度,加上柑橘匀浆基质本身成分复杂,使得衍生物自由浓度大大降低,故30% Na2SO4是最合适的盐浓度。
图5d比较了400、600、800、1000和1200 r·min−1五种搅拌速度下萃取效率的变化。大部分待测物在转速1000 r·min−1时,萃取量最大,再加大转速后萃取量开始减少,可能是因为适当搅拌有利于溶液中待测物质的扩散,从而提高萃取效率,但速度太快又会导致目标物与涂层的接触时间变短,故选择搅拌速率为1000 r·min−1。
2.3 方法评价
在上述优化的试验条件下,对柑橘匀浆中加标氨基酸样品进行一次衍生,考察了LLME/DI-SPME/GC-FID法的线性范围、相关系数(Correlation Coefficients,R2)、精密度(Radio Science Division,RSD)、检出限(Limits of detection,LOD)和定量限(Limits of quantitation,LOQ),结果如表1所示。
表 1 15种氨基酸的线性关系、检出限、定量限和重复性Table 1. Linearity, LOD, LOQ and precision of 15 amino acids氨基酸 线性范围
(μg·g−1)线性方程 R2 检出限
(μg·kg−1)定量限
(μg·kg−1)精密度
(%, n=5)Ala 0.01~100 y=5906.1x+362684 0.9960 1.13 3.77 6.38 Gly 0.01~100 y=6231.3x+65829 0.9982 0.85 2.82 5.03 Val 0.001~100 y=3264.6x+40275 0.9994 1.13 3.78 11.93 Leu 0.001~100 y=5363.6x+43811 0.9986 1.01 3.36 3.93 Ile 0.001~100 y=1878.2x+8723.2 0.9981 1.08 3.60 9.12 Asn 0.1~100 y=4498.1x+509690 0.9989 2.48 8.26 6.14 Met 0.001~100 y=3540.7x+14159 0.9987 1.09 3.65 3.53 Asp 0.1~100 y=523.37x+202610 0.9999 12.60 41.99 6.93 Phe 0.001-100 y=5422.5x+144034 0.9991 0.92 3.05 11.40 Glu 0.1~100 y=643.45x+37042 0.9984 4.12 13.73 8.00 Cys 0.001~100 y=668.51x+55466 0.9962 3.16 10.52 7.56 Lys 0.01~100 y=1210.5x+122596 0.9999 2.56 8.53 12.80 His 0.1~100 y=473.23x+14353 0.9999 4.76 15.97 2.51 Trp 0.01~100 y=2131.4x+28292 0.9998 1.58 5.28 8.74 Tyr 0.01~100 y=2470.9x+16296 0.9998 1.27 4.24 12.10 该方法对柑橘中15种氨基酸所得回归方程均具有良好的线性关系,R2均大于0.99,其中天冬酰胺、天冬氨酸、谷氨酸和组氨酸线性范围为0.1~100 μg·g−1,丙氨酸、甘氨酸、赖氨酸、色氨酸和酪氨酸线性范围为0.01~100 μg·g−1,缬氨酸、亮氨酸、异亮氨酸、甲硫氨酸、苯丙氨酸和半胱氨酸线性范围为0.001~100 μg·g−1。以3倍信噪比计算,方法的检出限LOD为0.85~12.60 μg·kg−1,以10倍信噪比计算,方法的定量限LOQ为2.82~41.99 μg·kg−1。重复5次平行试验,RSD介于2.51%~12.80%,说明方法的重复性也较好。
2.4 LLME/DI-SPME-GC法与相关文献方法比较
表2比较了LLME/DI-SPME-GC法与相关文献报道的食品中氨基酸检测的各指标关系。结果表明,在同样采用气相色谱氢火焰离子检测法(GC-FID)进行检测时,本方法检出限为2.82~41.99 μg·kg−1,比Nozal等[27]建立的方法检测限更低;与Omar等[28-29]采用的毛细管电泳紫外检测法和师子豪等[30]采用的反相高效液相色谱法相比,衍生化时间更短。总体说来,本方法具有操作简单、衍生快速、较低的检出限,较好的重现性,较高的精密度等优点,适合柑橘果肉中氨基酸检测和内在品质评价。
表 2 LLME/DI-SPME-GC与相关文献食品中氨基酸检测比较Table 2. Comparison of LLME/DI-SPME -GC in this paper with other methods reported in literatures for determination of amino acids in food samples2.5 加标回收率和真实样品检测
按高(100 μg·g−1)、中(1 μg·g−1)、低(0.1 μg·g−1)3个浓度水平进行加标回收试验,结果见表3所示。除加标浓度0.1 μg·g−1时,个别氨基酸回收率低于80%或超过125%,绝大部分氨基酸加标回收率在82.51%~121.75%,RSD%为0.75%~15.51%(除谷氨酸加标1 μg·g−1时RSD为17.93%),该结果与课题组前期数据结果相近[10]。将本方法应用于市场采购的新鲜柑橘(伦晚橙)样品进行检测,共检测出了16种氨基酸,包括丙氨酸、甘氨酸、缬氨酸、亮氨酸、异亮氨酸、脯氨酸、天冬酰胺、甲硫氨酸、天冬氨酸、苯丙氨酸、谷氨酸、半胱氨酸、赖氨酸、组氨酸、色氨酸和酪氨酸,含量范围为1.20~232.39 mg·100 g−1,其中脯氨酸含量最高232.8 mg·100 g−1,甲硫氨酸含量最低1.20 mg·100 g−1。
表 3 柑橘样品加标回收率和真实样品检测Table 3. Recoveries and precision of free amino acids in spiked orange samples氨基酸 加标浓度0.1 μg·g−1 加标浓度1.0 μg·g−1 加标浓度100 μg·g−1 真实样品检测
(mg·100 g−1)回收率(%) RSD(%) 回收率(%) RSD(%) 回收率(%) RSD(%) Ala 100.29 6.55 100.00 13.27 100.13 9.92 18.42 Gly 82.51 5.03 100.16 10.19 99.38 14.82 3.17 Val 100.77 11.93 100.01 0.33 99.67 12.66 3.70 Leu 124.62 3.93 99.54 2.97 99.76 7.07 2.45 Ile 109.41 9.12 99.91 2.53 99.33 10.19 1.39 Asn 121.75 6.14 97.73 13.11 99.78 11.84 33.99 Met 70.86 3.53 100.28 9.05 99.50 11.52 1.20 Asp 84.56 6.93 101.67 3.05 100.06 5.99 116.14 Phe 126.90 11.40 99.75 11.07 99.55 14.00 7.97 Glu 127.75 8.00 96.97 17.93 101.33 10.79 17.27 Cys 107.32 7.56 99.94 12.75 100.32 6.39 24.89 Lys 106.89 12.80 113.60 15.51 99.83 8.58 30.38 His 106.53 2.51 99.33 0.75 100.05 12.18 9.09 Trp 87.07 8.74 127.98 11.47 99.83 3.67 3.98 Tyr 78.74 12.10 100.69 8.80 99.88 3.24 1.97 3. 结论
本研究建立了同时测定柑橘果肉中16种游离氨基酸的LLME/DI-SPME-GC法。通过LLME对样品进行净化提取,减少了基质对萃取涂层的破坏,拓展了DI-SPME在复杂基质样品中的应用范围。所建立的方法稳定,检出限为0.85~12.60 μg·kg−1,RSD为2.5%~12.8%,回收率为82.51%~121.75%。本方法具有操作简单,灵敏度较低,衍生化时间短等优点,应用于真实柑橘样品中检测,共检出16种游离氨基酸氨基,可为柑橘内在质量评价和氨基酸检测提供参考依据。
-
表 1 15种氨基酸的线性关系、检出限、定量限和重复性
Table 1 Linearity, LOD, LOQ and precision of 15 amino acids
氨基酸 线性范围
(μg·g−1)线性方程 R2 检出限
(μg·kg−1)定量限
(μg·kg−1)精密度
(%, n=5)Ala 0.01~100 y=5906.1x+362684 0.9960 1.13 3.77 6.38 Gly 0.01~100 y=6231.3x+65829 0.9982 0.85 2.82 5.03 Val 0.001~100 y=3264.6x+40275 0.9994 1.13 3.78 11.93 Leu 0.001~100 y=5363.6x+43811 0.9986 1.01 3.36 3.93 Ile 0.001~100 y=1878.2x+8723.2 0.9981 1.08 3.60 9.12 Asn 0.1~100 y=4498.1x+509690 0.9989 2.48 8.26 6.14 Met 0.001~100 y=3540.7x+14159 0.9987 1.09 3.65 3.53 Asp 0.1~100 y=523.37x+202610 0.9999 12.60 41.99 6.93 Phe 0.001-100 y=5422.5x+144034 0.9991 0.92 3.05 11.40 Glu 0.1~100 y=643.45x+37042 0.9984 4.12 13.73 8.00 Cys 0.001~100 y=668.51x+55466 0.9962 3.16 10.52 7.56 Lys 0.01~100 y=1210.5x+122596 0.9999 2.56 8.53 12.80 His 0.1~100 y=473.23x+14353 0.9999 4.76 15.97 2.51 Trp 0.01~100 y=2131.4x+28292 0.9998 1.58 5.28 8.74 Tyr 0.01~100 y=2470.9x+16296 0.9998 1.27 4.24 12.10 表 2 LLME/DI-SPME-GC与相关文献食品中氨基酸检测比较
Table 2 Comparison of LLME/DI-SPME -GC in this paper with other methods reported in literatures for determination of amino acids in food samples
表 3 柑橘样品加标回收率和真实样品检测
Table 3 Recoveries and precision of free amino acids in spiked orange samples
氨基酸 加标浓度0.1 μg·g−1 加标浓度1.0 μg·g−1 加标浓度100 μg·g−1 真实样品检测
(mg·100 g−1)回收率(%) RSD(%) 回收率(%) RSD(%) 回收率(%) RSD(%) Ala 100.29 6.55 100.00 13.27 100.13 9.92 18.42 Gly 82.51 5.03 100.16 10.19 99.38 14.82 3.17 Val 100.77 11.93 100.01 0.33 99.67 12.66 3.70 Leu 124.62 3.93 99.54 2.97 99.76 7.07 2.45 Ile 109.41 9.12 99.91 2.53 99.33 10.19 1.39 Asn 121.75 6.14 97.73 13.11 99.78 11.84 33.99 Met 70.86 3.53 100.28 9.05 99.50 11.52 1.20 Asp 84.56 6.93 101.67 3.05 100.06 5.99 116.14 Phe 126.90 11.40 99.75 11.07 99.55 14.00 7.97 Glu 127.75 8.00 96.97 17.93 101.33 10.79 17.27 Cys 107.32 7.56 99.94 12.75 100.32 6.39 24.89 Lys 106.89 12.80 113.60 15.51 99.83 8.58 30.38 His 106.53 2.51 99.33 0.75 100.05 12.18 9.09 Trp 87.07 8.74 127.98 11.47 99.83 3.67 3.98 Tyr 78.74 12.10 100.69 8.80 99.88 3.24 1.97 -
[1] 祁春节, 顾雨檬, 曾彦. 我国柑橘产业经济研究进展[J]. 华中农业大学学报,2021,40(1):58−69. [QI Chunjie, GU Yumeng, ZENG Yan. Progress of citrus industry economy in China[J]. Journal of Huazhong Agricultural University,2021,40(1):58−69. QI Chunjie, GU Yumeng, ZENG Yan. Progress of citrus industry economy in China [J]. Journal of Huazhong Agricultural University, 2021, 40(1): 58-69.
[2] 李勋兰, 洪林, 杨蕾, 等. 11个柑橘品种果实营养成分分析与品质综合评价[J]. 食品科学,2020,41(8):228−233. [LI Xunlan, HONG Lin, YANG Lei, et al. Analysis of nutritional components and comprehensive quality evaluation of citrus fruit from eleven varieties[J]. Food Science,2020,41(8):228−233. LI Xunlan, HONG Lin, YANG Lei, et al. Analysis of nutritional components and comprehensive quality evaluation of citrus fruit from eleven varieties [J]. Food Science, 2020, 41(8): 228-233.
[3] 何莎莎. 不同类型柑橘果实氨基酸组成分析及“三度”法营养价值评价[D]. 重庆: 西南大学, 2018. HE S S. Analysis of amino acid composition of different types of citrus fruits and evaluation of nutritional value by "three degree" method [D]. Chongqing: Southwest University, 2018.
[4] 陈山乔, 李丹丹, 贾丽娜, 等. 柑橘内在品质评价及保鲜技术研究进展[J]. 包装工程,2021,42(7):45−53. [CHEN Shanqiao, LI Dandan, JIA Lina, et al. Research progress in evaluation methods and maintenance techniques of inner citrus quality[J]. Packaging Engineering,2021,42(7):45−53. CHEN Shanqiao, LI Dandan, JIA Lina, et al. Research progress in evaluation methods and maintenance techniques of inner citrus quality [J]. Packaging Engineering, 2021, 42(7): 45-53.
[5] 赫欣睿, 武中庸, 叶永丽, 等. 高效液相色谱法测定氨基酸的研究进展[J]. 分析测试学报,2016,35(7):922−928. [HE Xinrui, WU Zhongyong, YE Yongli, et al. Research progress on detection of amino acids by high performance liquid chromatography[J]. Journal of Instrumental Analysis,2016,35(7):922−928. HE Xinrui, WU Zhongyong, YE Yongli, et al. Research progress on detection of amino acids by high performance liquid chromatography [J]. Journal of Instrumental Analysis, 2016, 35(7): 922-928.
[6] 陈荣珠, 王小平, 高伟城, 等. 柱前衍生-HPLC法测定五指毛桃中16种游离氨基酸含量[J]. 食品工业科技,2022,43(17):306−315. [CHEN Rongzhu, WANG Xiaoping, GAO Weicheng, et al. Determination of 16 free amino acids of Ficus hirta by pre-column derivatization high performance liquid chromatography[J]. Science and Technology and Food Industry,2022,43(17):306−315. doi: 10.13386/j.issn1002-0306.2021110244 CHEN Rongzhu, WANG Xiaoping, GAO Weicheng, et al. Determination of 16 free amino acids of Ficus hirta by pre-column derivatization high performance liquid chromatography [J]. Science and Technology and Food Industry, 2022, 43(17): 306-315. doi: 10.13386/j.issn1002-0306.2021110244
[7] 段静怡, 李自燕, 李建, 等. 基于游离氨基酸的组分及特征比较四种食用菌与四种果蔬的营养与风味特征[J]. 菌物学报,2020,39(6):1077−1089. [DUAN Jingyi, LI Ziyan LI Jian, et al. Comparison of nutritional and flavor characteristics between four edible fungi and four fruits and vegetables based on components and characteristics of free amino acids[J]. Mycosystema,2020,39(6):1077−1089. DUAN Jingyi, LI Ziyan LI Jian, et al. Comparison of nutritional and flavor characteristics between four edible fungi and four fruits and vegetables based on components and characteristics of free amino acids [J]. Mycosystema, 2020, 39(6): 1077-1089
[8] 袁光蔚, 吴毅, 王海波, 等. 基于超高效液相色谱-四级杆-静电场轨道阱高分辨质谱快速测定水果中18种游离氨基酸[J]. 食品工业科技,2021,42(5):243−249. [YUAN Guangwei, WU Yi, WANG Haibo, et al. Determination of eighteen kinds of free amino acids in fruits by ultra performance liquid chromatography-quadrupole-extractive orbitrap mass spectrometry[J]. Science and Technology of Food Industry,2021,42(5):243−249. YUAN Guangwei, WU Yi, WANG Haibo, et al. Determination of eighteen kinds of free amino acids in fruits by ultra performance liquid chromatography-quadrupole-extractive orbitrap mass spectrometry [J]. Science and Technology of Food Industry, 2021, 42(5): 243-249.
[9] HAI Y T, COLLIN F, PERQUIS L, et al. Twenty kinds of amino acid determination using capillary electrophoresis: A review[J]. Analytica Chimica Acta,2021,1174:338233. doi: 10.1016/j.aca.2021.338233
[10] 李晓庆, 蔡颖, 潘思轶, 等. 氯甲酸异丁酯二次衍生-气相色谱法检测柑橘中游离氨基酸[J]. 华中农业大学学报,2020,39(2):122−132. [LI Xiaoqing, CAI ying, PAN Siyi, et al. Analysis of free amino acids in citrus by gas chromatography after isobutyl chloroformate second derivatization[J]. Journal of Huazhong Agricultural University,2020,39(2):122−132. LI Xiaoqing, CAI ying, PAN Siyi, et al. Analysis of free amino acids in citrus by gas chromatography after isobutyl chloroformate second derivatization [J]. Journal of Huazhong Agricultural University, 2020, 39(2): 122-132.
[11] SONG J X, BI J F, CHEN Q Q, et al. Assessment of sugar content, fatty acids, free amino acids, and volatile profiles in jujube fruits at different ripening stages[J]. Food Chemistry,2019,270:344−352. doi: 10.1016/j.foodchem.2018.07.102
[12] 国家卫生健康委员会. GB 5009.124-2016 食品安全标准 食品中氨基酸的测定[S]. 北京: 中国标准出版社, 2017. National Health Commission. GB 5009.124-2016 Food Safety Standard - Determination of Amino Acids in Foods [S]. Beijing: China Standards Press, 2017.
[13] LI Z Q, WU J, LI J. Analysis of amino acids in blood by combining zeolitic imidazolate framework-8-based solid phase extraction and capillary electrophoresis[J]. Journal of Pharmaceutical and Biomedical Analysis,2019,168:30−37. doi: 10.1016/j.jpba.2019.02.015
[14] OMAR K A, SADEGHI R. Novel nonaol-based deep eutectic solvents: thermophysical properties and their applications in liquid-liquid extraction and amino acid detection[J]. Journal of Molecular Liquids,2021,336:116359. doi: 10.1016/j.molliq.2021.116359
[15] MOHABBAT T, DREW B. Simultaneous determination of 33 amino acids and dipeptides in spent cell culture media by gas chromatography-flame ionization detection following liquid and solid phase extraction[J]. Journal of Chromatography B,2008,862:86−92. doi: 10.1016/j.jchromb.2007.11.003
[16] NAKHODCHI S, ALIZADEH N. Rapid simultaneous determination of ketamine and midazolam in biological samples using ion mobility spectrometry combined by headspace solid-phase microextraction[J]. Journal of Chromatography A,2021,1658:462609. doi: 10.1016/j.chroma.2021.462609
[17] YU CHEN, WU F S, LUO X G, et al. Porphyrin-based covalent organic framework coated stainless steel fiber for solid-phase microextraction of polycyclic aromatic hydrocarbons in water and soil samples[J]. Microchemical Journal,2021,168:106364. doi: 10.1016/j.microc.2021.106364
[18] 楚耀娟, 张雪娜, 向孝哲, 等. 分子印迹阵列固相微萃取用于检测果蔬中多种有机磷农药残留[J]. 食品科学,2021,42(6):310−315. [CHU Yaojuan, ZHANG Xuena, XIANG Xiaozhe, et al. Determination of multiple organophosphorus pesticide residues in fruits and vegetables by molecularly imprinted array solid phase microextraction[J]. Food Science,2021,42(6):310−315. CHU Yaojuan, ZHANG Xuena, XIANG Xiaozhe, et al. Determination of multiple organophosphorus pesticide residues in fruits and vegetables by molecularly imprinted array solid phase microextraction [J]. Food Science, 2021, 42(6): 310-315.
[19] DENG C, LI N, ZHANG X. Rapid determination of amino acids in neonatal blood samples based on derivatization with isobutyl chloroformate followed by solid-phase microextraction and gas chromatography/mass spectrometry[J]. Rapid Communication in Mass Spectrometry Rcm,2010,18(21):2558−2564.
[20] MUDIAM M. K. R, RATNASEKHAR C, JAIN R, et al. Rapid and simultaneous determination of twenty amino acids in complex biological and food samples by solid-phase microextraction and gas chromatography mass spectrometry with the aid of experimental design after ethyl chloroformate derivatization[J]. Journal of Chromatography B,2021,907:56−64.
[21] XUE W L, LI N, ZHANG Z M, et al. Dummy template based molecularly imprinted solid-phase microextraction coating for analysis of trace disinfection by-product of 2, 6-dichloro-1, 4-benzoquinone using high-performance liquid chromatography[J]. Talanta,2022,239:123065. doi: 10.1016/j.talanta.2021.123065
[22] XIANG X Z, WANG Y L, ZHANG X W, et al. Multifiber solid-phase microextraction using different molecularly coatings for simultaneous selective extraction and sensitive determination of organophosphorus pesticides[J]. Journal of Separation Science,2020,43(4):1−10.
[23] VALENZUELA E F, PAULA F G F, TEIXEIRA A P C, et al. A new carbon nanomaterial solid-phase microextraction to pre-concentrate and extract pesticides in environmental water[J]. Talanta,2020,217:121011. doi: 10.1016/j.talanta.2020.121011
[24] ORAZBAYEVA D, KOZIEL J A, TRUJILLO-RODRIGUEZ M J, et al. Polymeric ionic liquid sorbent coatings in headspace solid-phase microextraction: A green sample preparation technique for the determination of pesticides in soil[J]. Microchemical Journal,2020,157:104996. doi: 10.1016/j.microc.2020.104996
[25] 高远莉. 分子印迹固相微萃取涂层的制备、评价及在农残检测种的应用[D]. 武汉: 华中农业大学, 2013. GAO Y L. Preparation and evaluation of molecularly imprinted solid phase microextraction coating and its application in pesticide residue detection [D]. Wuhan: Huazhong Agricultural University, 2013.
[26] 陈雪, 梁克红, 朱宏, 等. 游离氨基酸检测方法及其应用[J]. 食品安全质量检测学报,2021,12(18):7298−7304. [CHEN Xue, LIANG Kehong, ZHU Hong, et al. Analysis method and application of free amino acids[J]. Journal of Food Safety and Quality,2021,12(18):7298−7304. CHEN Xue, LIANG Kehong, ZHU Hong, et al. Analysis method and application of free amino acids [J]. Journal of Food Safety and Quality, 2021, 12(18): 7298-7304
[27] NOZAL M J, BERNAL J L, TORIBIO M J, et al. Rapid and sensitive method for determining free amino acids in honey by gas chromatography with flame ionization or mass spectrometric detection[J]. Journal of Chromatography A,2004,20:137−146.
[28] OMAR M M A, ELBASHIR A A, SCHMITZ O J. Capillary electrophoresis method with UV-detection for analysis of free amino acids concentrations in food[J]. Food Chemistry,2017,214:300−307. doi: 10.1016/j.foodchem.2016.07.060
[29] 卢彬, 付铭, 范蕊, 等. 柱前衍生反相高效液相色谱法测定新疆特色食品馕中氨基酸含量[J]. 食品安全质量检测学报,2021,12(2):713−720. [LU Bin, FU Ming, FAN Rui, et al. Determination of amino acids in Xingjiang special food Nang by reversed-phase high performance liquid chromatography with pre-column derivatization[J]. Journal of Food Safety and Quality,2021,12(2):713−720. LU Bin, FU Ming, FAN Rui, et al. Determination of amino acids in Xingjiang special food Nang by reversed-phase high performance liquid chromatography with pre-column derivatization [J]. Journal of Food Safety and Quality, 2021, 12(2): 713-720.
[30] 师子豪, 祝伟霞, 魏蔚, 等. 气相色谱氢火焰离子法测定三文鱼中氨基酸的含量[J]. 河南水产,2021(5):32−34. [SHI Zhihao, ZHU Werixia, WEI Wei, et al. Determination of amino acids in Salmon by gas chromatography hydrogen flame ionization method[J]. Henan Fisheries,2021(5):32−34. SHI Zhihao, ZHU Werixia, WEI Wei, et al. Determination of amino acids in Salmon by gas chromatography hydrogen flame ionization method [J]. Henan Fisheries, 2021(5): 32-34.
-
期刊类型引用(0)
其他类型引用(1)





 下载:
下载:
 下载:
下载:



