Research Progress on Residue Detection of Methods of Fluoroquinolones and Amphenicols in Animal-derived Foods Based on Liquid Chromatographic and Liquid Chromatographic Mass Spectrometry
-
摘要: 氟喹诺酮类和酰胺醇类药物是畜禽生产中常用的广谱高效抗生素,应用广泛,但药物残留超标现象频出。液相色谱与液质联用法是目前检测氟喹诺酮类和酰胺醇类药物残留的主要方法,当前国内标准规定的检测方法仅能在牛奶中同时检测这两类兽药残留,在其他基质中鲜有报道,因此亟需建立和优化氟喹诺酮类和酰胺醇类药物在不同基质中同时检测的方法。本文概述了液液萃取、固相萃取、QuEChERS方法和加速溶剂萃取等四种常用前处理技术,具体论述了纳米材料吸附剂这一研究热点的应用情况,并从色谱和色谱质谱联用法的角度,总结并论述了不同基质条件、仪器配置、液相条件对方法性能参数的影响,以期为动物源性食品中兽药残留的检测和监管提供参考。Abstract: Fluoroquinolones and amphenicols are widely used as broad-spectrum and high-efficiency antibiotics in livestock and poultry production, but excessive drug residues frequently occur. Liquid chromatographic and liquid chromatographic mass spectrometry are the primary methods to detect the fluoroquinolones and amphenicols. According to the current domestic standard, these two veterinary drug residues can only be detected in milk simultaneously, and there are few reports in other matrices. Therefore, it is urgent to establish and optimize the simultaneous detection methods of fluoroquinolones and amphenicols in different matrices. This paper summarizes four standard pretreatment techniques such as liquid-liquid extraction, solid-phase extraction, QuEChERS method and accelerated solvent extraction, and discusses the application of hotspot nanomaterial adsorbent in detail. From the perspective of chromatography and chromatography mass spectrometry, the effects of different matrix conditions, instrument conditions and liquid phase conditions on the performance parameters of the method are summarized and discussed to provide a reference for the detection and supervision of veterinary drug residues in animal-derived foods.
-
氟喹诺酮类药物(fluoroquinolone,FQs)属于第三、四代喹诺酮类药物(quinolones,QNs)。氟原子的引入增加了QNs的脂溶性,使得组织渗透性增强、半衰期延长,进而拓宽了抗菌谱、增强了抗菌活性。另外,特殊的作用机理确保了FQs与其他抗生素基本不存在交叉耐药性。但研究发现,FQs会造成肌腱断裂以及神经损伤等持久且不可逆转的毒副作用[1-2]。因此,美国食品药品监督管理局和欧洲药品管理局均建议限制FQs的使用[3]。
酰胺醇类药物,又名氯霉素类药物(chloramphenicols,CAPs),是由氯霉素衍生出的一类广谱抗菌药,对革兰氏阳性和阴性菌均有抑制作用。但在长期使用过程中发现,氯霉素会导致再生障碍性贫血等多种毒副作用[4],已被多国禁止作为兽药使用。甲砜霉素(thiamphenicol,TAP)和氟苯尼考(florfenicol,FF)是氯霉素的衍生物,对苯环上的羟基进行了取代,大大减少了氯霉素相关的毒副作用[4-5],但依然具有较低的血液毒性和胚胎毒性。
液相色谱和液质联用法具有灵敏度高、准确性强、重现性好的优点,在药物检测方面应用广泛,是这两类兽药残留检测的常用方法。虽然我国和欧盟均对这两类兽药的最大残留限量(maximum residue limit,MRL)进行了明确规定[6-7],但这两类兽药残留超标现象屡禁不止。本文汇总了国家市场监督管理总局2021年7月至2022年7月发布的国家食品安全监督抽检结果,在畜禽产品中FQs与CAPs不合格的批次数超过不合格总批次数的一半,尤其在禽蛋中这两类药物的不合格批次数占总批次数的69.8%,因此检测和监管中需要格外注意这两类兽药。随着食品安全国家标准的不断更新,要求检测的兽药种类数目也在不断增多,发展快速有效、简便实用且覆盖面广的兽药多残留分析方法尤其重要[8]。
目前,FQs与磺胺类、四环素类、硝基咪唑类等兽药的多残留同时检测已有多个国内和国外标准,但FQs与CAPs的同时检测方法却鲜有报道,因此如何优化并完善这两类兽药在动物源性食品中的同时检测方法是目前亟待解决的问题。本文综述了近十年来FQs与CAPs残留的样品前处理方法以及液相色谱和液质联用法的研究进展,具体论述了纳米材料吸附剂的应用情况,总结了FQs与CAPs各检测方法的检测条件与优缺点,以期为动物源性食品中兽药残留检测新方法的建立提供理论基础。
1. 样品前处理技术
样品前处理是兽药残留检测必不可少的步骤,不当的样品前处理可能导致目标物提取不完全或提取后的样品存在共提物,会对目标物的测定造成干扰,降低检测的准确性,因此如何快速且高效地完成复杂样品中多种兽药残留的提取、净化及富集是液相色谱和质谱法必须解决的问题。常见的兽药残留前处理方法包括液液萃取(liquid-liquid extraction,LLE)、固相萃取(solid-phase extraction,SPE)、QuEChERS(quick,easy,cheap,effective,rugged,safe)方法和加速溶剂萃取(accelerated solvent extraction,ASE)等。
1.1 液液萃取
LLE是利用各组分在不相容溶剂中溶解度不同,而将目标物从样品基质中分离出来的前处理方法。乙腈等有机溶剂常被用作萃取剂,其适用范围广,对大部分兽药残留都有很好的提取效果。除萃取剂外,温度和压力直接影响到各组分在水相和有机相中的溶解度,也是LLE方法所要考虑的因素。Barreto等[9]采用低温净化对萃取液进行处理,只需要经过乙腈萃取、低温净化和离心上清液三个步骤就能有效分离出牛肉、猪肉、禽肉和鱼肉中9种FQs残留,加标回收率为79%~115%,RSD为2.6%~15.4%。Xiao等[10]以0.2%氢氧化铵水溶液作为萃取溶剂,中等压力,200 ℃条件下,对禽组织中的CAP、TAP、FF和氟苯尼考胺(florfenicol amine,FFA)进行亚临界水萃取,所得加标回收率为86.8%~101.5%,RSD小于7.7%,提取效果较好。亚临界水萃取技术是近十年来刚刚兴起的一项前处理技术,暂时没有相应的商业化设备,这不利于标准化方法的建立。
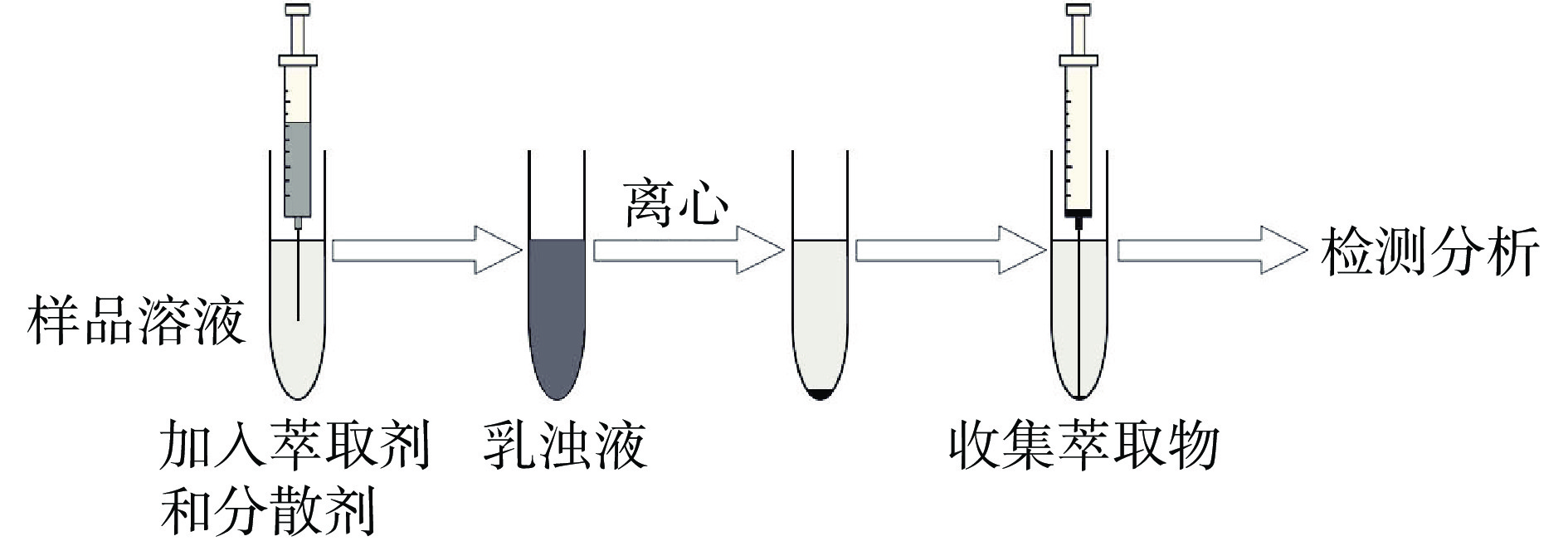
分散液液微萃取(dispersive liquid-liquid microextraction,DLLME)是Rezaee等[11]提出的一种液相微萃取技术。DLLME的操作流程如图1所示,在样品溶液中加入萃取剂和分散剂后,因为分散剂的作用,萃取剂和水相不会分层,而是形成萃取剂、分散剂和样品溶液三者的乳浊液。将乳浊液离心后,利用注射器收集底层萃取物,即可实现对目标物的提取。分散剂的使用,使萃取剂以微小液滴的形式在样品溶液中散开,增加了水相和萃取剂之间的接触表面积,从而提高了萃取效率。孙鹏等[12]以离子液体作为萃取剂采用DLLME提取液态奶中FQs残留,加标回收率在83.9%~98.8%,RSD为4.6%~5.4%。相较于常规有机溶剂,离子液体更加绿色环保,具有广阔的应用前景。Karami-Osbooa等[13]采用DLLME提取牛奶中CAP和FF残留,通过对比发现,当0.4 mL氯仿用作分散剂时,所得回收率最高,为69.1%~79.4%,RSD小于15%。总之,尽量减少提取过程中有机溶剂的消耗,简化实验步骤,是当前LLE方法的发展趋势。
1.2 固相萃取
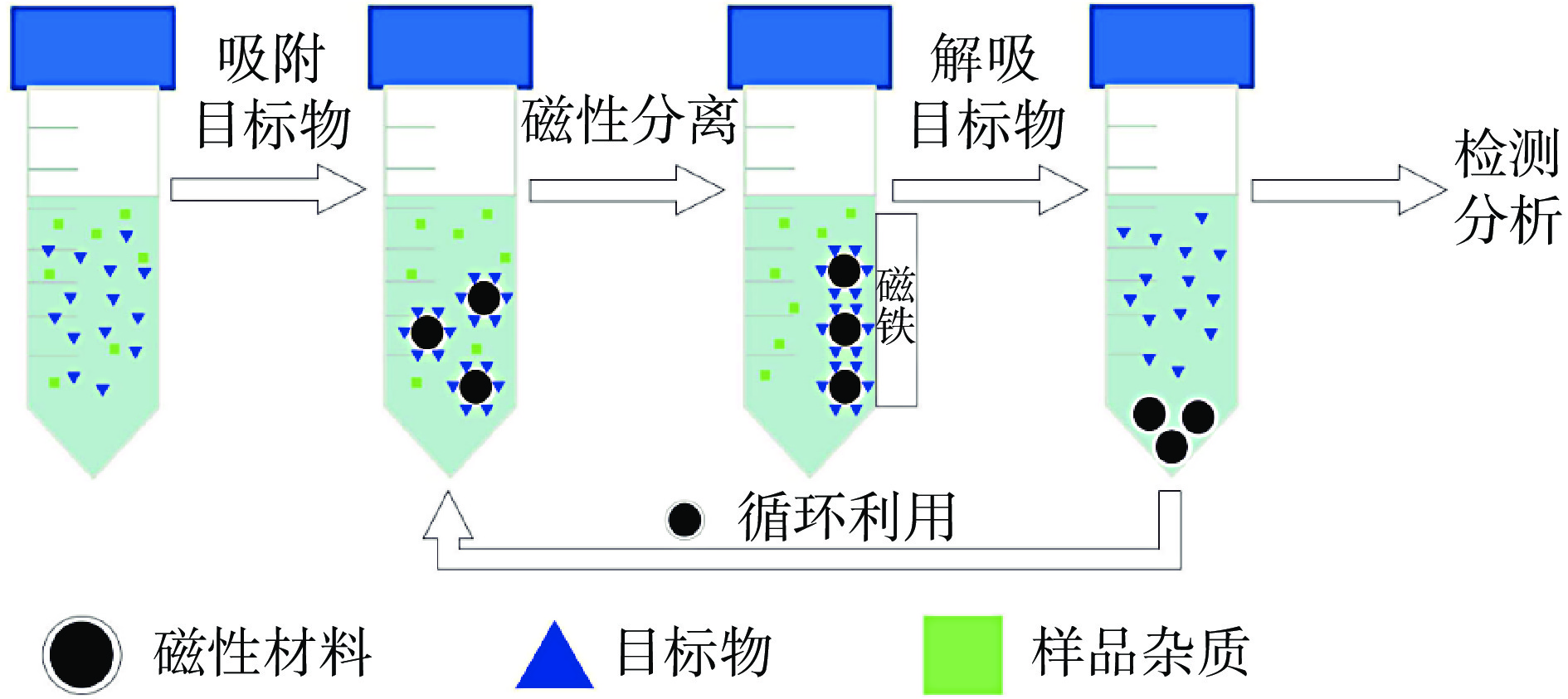
SPE是利用固相吸附剂对不同物质的吸附能力不同,而将目标物从样品基质中分离出来的前处理方法。纳米材料是以处在纳米范围内的物质为基本结构单位所构成的材料,因其高表面积比、独特的结构和理化性质等特点,在被用作固相萃取,尤其是磁性固相萃取(micro-solid phase extraction,MSPE)的吸附剂,较其他材料具有明显的优势,是近年来食品安全检测的研究热点。MSPE的操作流程如图2所示,将磁性吸附剂加入样品溶液,待磁性吸附剂与目标物充分结合后,借助外界磁场来实现磁性吸附剂和样品溶液的分离,然后再用合适的洗脱液将目标物从吸附剂上洗脱下来,以供检测分析[14]。MSPE操作简单,不需要装填SPE柱,只需借助外界磁场,就可以对目标物进行分离,已实现对婴幼儿肉类食品中FQs残留的自动化在线样品处理[15]。
与传统吸附剂相比,纳米材料超高的表面积比,大大提高了它的吸附性能,增强了对目标物的提取能力。Bagheri等[16]将磁性二维金属有机框架用于测定牛奶中FQs残留,经液相色谱串联质谱(liquid chromatography-tandem mass spectrometry,LC-MS/MS)测得OFL、环丙沙星(ciprofloxacin,CIP)、诺氟沙星(norfloxacin,NOR)的检测限(limit of detection,LOD)分别为0.012,0.009,0.016 ng/mL,回收率在95%~105%,RSD小于5%,且并未观察到其他化合物的干扰作用。适当的修饰能使纳米材料和目标物特异性结合,显著提高了检测的选择性和灵敏性。Huang等[17]基于一段能同时特异性识别CAPs残留的DNA序列,制作了相应磁性适配体,并结合HPLC-DAD测得该吸附剂对CAP、TAP和FF的饱和萃取容量分别为2.82、2.56和2.72 μg/g,并且证明了其对两性离子有很弱的非特异性吸附,不会干扰对目标物的检测。所得牛奶中CAP、TAP和FF的定量限(limit of quantification,LOQ)为0.40~0.55 ng/mL。强吸附能力和高选择性使得纳米材料在物质分离的同时,能够完成对目标物的富集作用。此外,纳米材料通常能被重复利用,有利于减少实验成本。He等[18]开发了一种以磁性石墨烯作为吸附剂结合HPLC测定牛肉、鸡肌肉、鸡蛋中7种FQs残留的检测方法,并且证明了该材料的高吸收能力(>6800 ng)、高富集因子(68~79倍)和高重复性(>40次),所得回收率为82.4%~108.5%,RSD为1.6%~3.4%。除了上述材料外,磁性共价有机框架[19]、磁性有机聚合物[20]和分子印迹聚合物[21]等纳米材料也均被应用于FQs与CAPs残留检测中,并取得了不错的分离效果。
1.3 QuEChERS
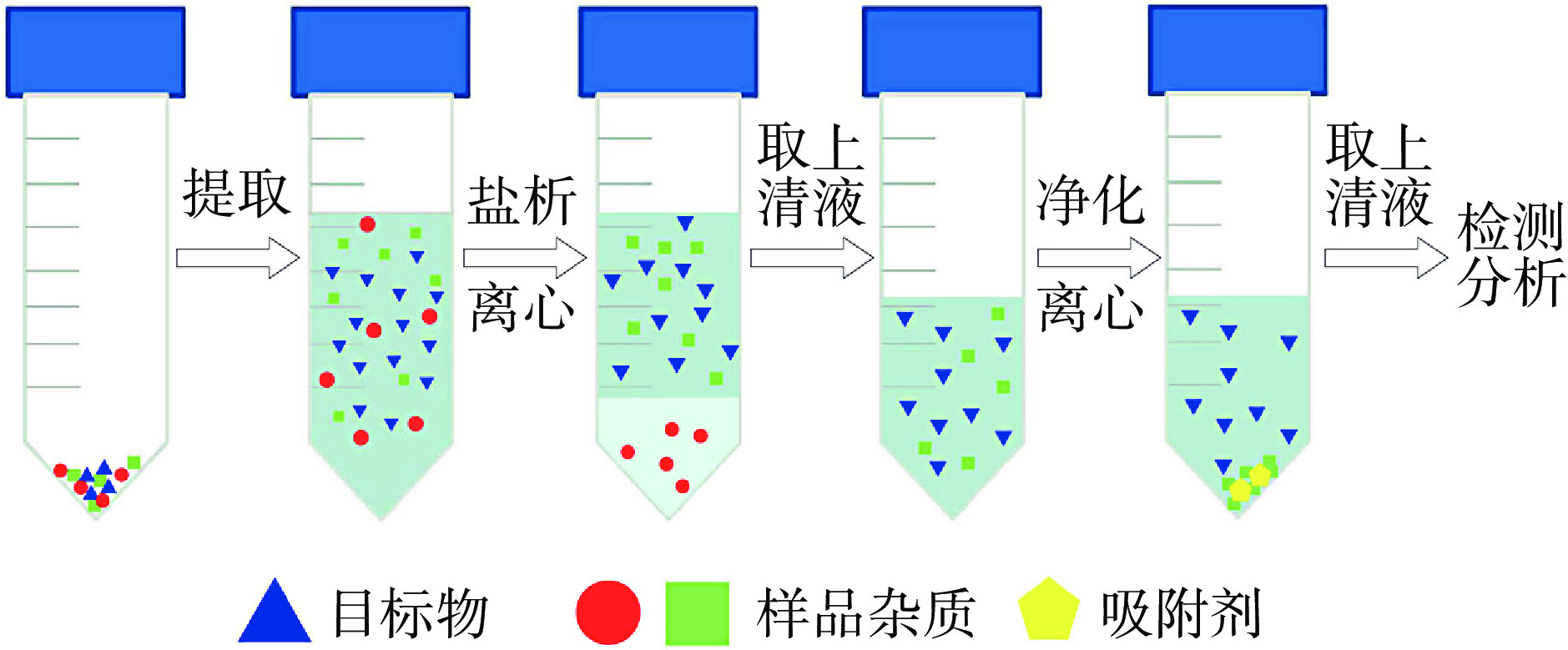
QuEChERS方法于2003年首先由Anastassiades等[22]提出,是一种在分散固相萃取(dispersive solid-phase extraction,DSPE)基础上建立的前处理方法。QuEChERS方法的操作流程如图3所示,粉碎后的样品经提取和盐析过程后,利用分散固相萃取原理,加入吸附剂与绝大多数杂质相互作用,通过离心将杂质与目标物分离,即可达到净化的效果。根据目标物的不同,对应前处理过程中所使用的提取溶剂、盐、吸附剂的种类和用量也会有所差异。在盐析剂的选择上,Lehotay[23]提出的乙酸钠缓冲盐版本和文献[24]提出的柠檬酸钠缓冲盐版本作为美国和欧盟的标准方法,将pH维持在5左右,可以有效防止目标物的电离和降解,已被世界各地的许多实验室作为常规方法采用。
吸附剂组合的选择直接关系到QuEChERS方法的净化效果,常用的吸附剂主要包括N-丙基乙二胺(primary secondary amine,PSA)、十八烷基硅烷(C18)、石墨化炭黑(graphitized carbon black,GCB)等。韩臣波[25]以50 mg PSA和100mg NH2作为吸附剂,用于提取牛奶中6种FQs残留,所得回收率为88.2%~106.2%,RSD小于10%,准确度满足实际检测要求。Liu等[26]探讨了不同吸附剂组合对牛奶和蜂蜜中CAP、TAP、FF残留回收率的影响,最终发现当加入0.35 g C18吸附剂和0.5 g Z-Sep吸附剂时,回收率最高,均高于90%,日内RSD均低于9.1%(n=3),日间RSD均低于8.7%(n=3)。纳米材料作为一种新型吸附剂,也可以用于QuEChERS方法。但目前,该类检测方法多局限于水果蔬菜中农药和真菌毒素的残留检测[27-30],用于动物源性食品中兽药残留检测的报道较为少见[24]。
1.4 加速溶剂萃取
加速溶剂萃取是一种通过较高的温度和压力来增强溶剂溶解目标物能力的自动萃取技术。魏丹等[31]在前处理过程中,先用ASE提取水产品中10种FQs,而后直接使用MSPE进行净化,省去了离心和过滤的过程,大大缩短了试验时间,加标回收率为81.6%~105.8%,RSD为4.2%~13.6%。Wang等[32]采用ASE方法,在80 ℃、1500 psi条件下,使用碱性甲醇溶液萃取禽蛋中的TAP、FF和FFA残留,所得回收率为90.31%~107.79%,RSD为1.46%~3.4%。虽然同样是改变温度和压力来实现目标物的提取,但不同于LLE中所提到亚临界流体萃取技术,ASE方法自动化程度高、操作简便、重现性好,在节约溶剂的同时还能减少人为误差造成的影响。综上所述,动物源性食品中FQs与CAPs残留不同样品前处理方法优缺点比较,见表1。
表 1 动物源性食品中FQs与CAPs残留不同样品前处理方法优缺点比较Table 1. Comparison of advantages and disadvantages of different sample pretreatment methods for FQs and CAPs residues in animal food前处理方法 优点 缺点 液液萃取LLE 操作方便,设备简单 有机溶剂消耗量大,
萃取效率低分散液液萃取DLLME 萃取效率高,环境友好 需要离心分离,易受
基质效应干扰固相萃取SPE 有机溶剂用量少,可自动化
批量处理,回收率高提取时间长,成本高,
易受基质效应干扰磁性固相萃取MSPE 可重复利用,不需要过滤,
应用范围广,富集能力强材料制备困难,成本高 加速溶剂萃取ASE 自动化程度高,有机溶剂用量少 设备成本高 QuEChERS 操作简单,有机溶剂用量少,
应用范围广,提取效果好提取步骤较为繁琐 2. 液相色谱法
2.1 高效液相色谱法
液相色谱法(liquid chromatography,LC)是以液体作为流动相的色谱法,该方法适用于难挥发、热稳定性差、大分子物质的定量定性分析。高效液相色谱(high performance liquid chromatography,HPLC)在液相色谱的基础上加上了高压输液泵,以克服流动相通过固定相时产生的压降,保证流动相以稳定的流速或压力输送到色谱系统,从而提高了分离效率。
高效液相色谱常用的检测器有二极管阵列检测器(diode array detector,DAD)、紫外吸收检测器(ultraviolet detector,UVD)、荧光检测器(fluorescence detector,FLD)、电致化学发光检测器(electrogenerated chemiluminescence,ECL)等。
C18反相色谱柱在进行物质分离时,会将强极性组分洗脱出来,然后依次洗脱极性较弱的组分,常用于FQs与CAPs残留的液相色谱检测中。但不同于其他CAPs,FFA极性较强,在C18色谱柱上的保留时间短,并不能直接与其他CAPs进行同时检测。谢恺舟等[33]在流动相中加入十二烷基硫酸钠与FFA生成弱极性离子对,从而增强样品的疏水性,改善分离效果,将FF和FFA与样品中其他组分完全分开。Granja等[34]采用HPLC-UVD对鱼肉中FF残留进行了测定,将FF全部转化为FFA,以提高测定的准确性,并用LC-MS/MS该方法的准确性进行了验证。结果显示,FF的CCα为840 μg/kg,CCβ为879 μg/kg。
在流动相的选择上,乙腈具有洗脱能力强、吸光度小的特点,常被用作液相色谱检测这两类药物残留的有机相。FQs属于两性化合物,其解离状态、荧光强度和溶解度与pH有直接关系,通常会在水相中加入甲酸、磷酸等酸性物质,以维持样品在流动相中的电离状态,增加FQs的响应值,提高检测方法灵敏度。Choi等[35]以磷酸(pH3)-乙腈作为流动相,采用HPLC-FLD对猪、鸡肌肉中沙拉沙星(sarafloxacin,SAR)残留进行检测,所得LOD分别为1.1和1.7 μg/kg。Moema等[36]以0.1%甲酸水(pH2.74)-乙腈作为流动相采用HPLC-DAD测得鸡肝中4种FQs的LOD为5~19 μg/kg,LOQ为23~62 μg/kg,回收率为83%~102%。Li等[37]设计了一种简单稳定的Ru(bpy)32+ ECL检测器,将Ru(bpy)32+修饰到电极表面,有效避免了传统柱后加入给样品带来稀释,造成色谱峰保留时间增加的缺点,以乙酸-乙酸钠缓冲液(pH5.4)/乙腈作为流动相,测得牛奶中4种FQs的LOD为0.006~0.02 μg/mL。另一方面,采用液相色谱法同时检测FF和FFA残留时,通常也会保证流动相为酸性[38],以改善FFA的出峰情况。
2.2 超高效液相色谱法
超高效液相色谱法(ultra performance liquid chromatography,UPLC)在HPLC基础上,通过减小色谱柱粒径、增加输液泵压力、采用高灵敏度检测器等实验技术,进一步缩短了分析时间,减少了溶剂用量,降低了分析成本[39]。
UPLC的各项性能均优于HPLC,为了进一步提高方法的准确性,对样品前处理的要求往往更高,以减少样品基质对检测结果的影响。Aufartová等[40]将微波辅助萃取与固相萃取结合,采用UPLC-FLD对鱼肉样品中5种FQs残留进行检测,所得LOD为0.5~6 μg/kg,回收率为87.9%~111.5%,并将该方法成功应用于实际样品的检测中。刁志祥等[41]建立了一种ASE-UPLC-FLD方法对猪肉中FF和FFA残留进行检测,所得LOD分别为3.3和1.2 μg/kg,LOD分别为4.7和10.6 μg/kg,所有目标物均能在5 min内被检测到,大大提高了检测效率。
不同基质中FQs与CAPs残留的液相色谱检测方法性能参数的比较见表2和表3。由于基质、前处理方法、液相条件、药物种类的不同,各方法的LOD、LOQ和回收率存在部分差异。由表2和表3可知,DAD和FLD方法的LOD和LOQ普遍高于UVD方法。DAD检测器较传统UVD检测器相比,能够给出各组分的紫外吸收曲线,在定性方面更加准确。除CAP需要衍生外,其他药物都具有荧光性质,可以直接进行荧光检测,所以FLD检测器常被应用于这两类兽药的残留检测当中。目前,还没有FLD方法同时检测FQs与CAPs残留的相关报道。
表 2 不同基质中FQs残留的液相色谱检测方法性能参数的比较Table 2. Comparison of performance parameters of liquid chromatography detection methods for FQs in different matrices基质 药物 色谱柱 流动相 检测限LODs(μg/kg) 定量限LOQs(μg/kg) 回收率(%) 方法 鸡肝 6FQs XTerra MS C18(3.0 mm×
150 mm,3.5 μm)乙腈-0.1%甲酸水(pH2.74) 5~19 23~62 83~102 LC-DAD[38] 牛奶和水 7FQs Kromasil C18(250 mm×
4.6 mm,5 μm)0.5%甲酸水-乙腈 0.10~0.26 0.32~0.85 68.8~120 HPLC-DAD[42] 牛奶 OFL,LOM,CIP Pack Pro C18(4.6 mm×
250 mm,5 μm)乙腈-水(1:9,v/v) 1.5~2.3 5.0~7.5 87.2~106.1 LC-UVD[43] 鱼 NOR,OFL,CIP Venusil XBP C18
(150 mm×4.6 mm,5 μm)乙腈-13 mmol/L四丁基溴化铵溶液(6:94,v/v) 2.65~3.65 8.82~12.16 69.3~102.8 HPLC-UVD[44] 猪、牛、鸡、鱼组织 15FQs Zorbax SB-Aq-C18
(250 mm×4.6 mm,5 μm)甲醇-乙腈-0.02 mol/L柠檬酸和0.03 mol/L醋酸铵 3 10 70.6~111.1 HPLC-UVD[45] 猪肝和湖水 NOR Waters Symmery C18
(4.6 mm×150 mm,5 μm)乙腈-0.4%磷酸和0.4%三乙胺水溶液(15:85,v/v) 6 77.4~97.1 UPLC-UVD[46] 牛奶 OFL,PEF,ENR,DIF Atlantis T3 100Å
(250 mm×4.6 mm,5 μm)乙酸-醋酸钠缓冲液(pH5.4)/乙腈(75:25,v/v) 6~60 94.5~100.8 HPLC-ECL[37] 鸡蛋 7FQs and 3QNs Phenomenex Luna C8
(150 mm×4.6 mm,5 μm)乙腈-0.02 mol/L草酸(pH4.0) 3~10 10~30 92~99 LC-FLD[47] 猪、牛、鸡、鱼的
肌肉组织5FQs Zorbax Eclipse XDB-C18
(150 mm×4.6 mm,5 μm)甲醇-乙腈-磷酸/三乙胺水溶液(15:5:80,v/v) 0.056~0.64 0.187~2.134 87.2~110.6 HPLC-FLD[48] 蜂蜜和水 5FQs Thermo C18(250 mm×
4.6 mm,5 μm)0.5%甲酸水-乙腈 0.14~0.61 0.39~1.1 80.1~119 HPLC-FLD[49] 注:恩诺沙星(enrofloxacin,ENR)、氧氟沙星(ofloxacin,OFL)、洛美沙星(lomefloxacin,LOM)、培氟沙星(pefloxacin,PEF),达氟沙星(danofloxacin,DFLX)、二氟沙星(difloxacin,DIF)。 表 3 不同基质中CAPs残留的液相色谱检测方法性能参数的比较Table 3. Comparison of performance parameters of liquid chromatography detection methods for CAPs in different matrices基质 兽药 色谱柱 流动相 检测限LODs(μg/kg) 定量限LOQs(μg/kg) 回收率
(%)方法 鸡肉 FF,FFA,TAP Hedera C18(250 mm×
4.6 mm,5 μm)0.01 mol/L NaH2PO4,0.005 mol/L十二烷基硫酸钠和0.1%三乙胺-乙腈(68:32,v/v) 0.5~1.5 2~5 78.5~88.7 HPLC-FLD[50] 禽蛋 TAP,FF,FFA Acquity UPLC BEH C18(2.1 mm×100 mm,1.7 μm) 0.005 mol/L NaH2PO4,0.003 mol/L十二烷基硫酸钠和0.05%三乙胺(pH5.3±0.1)-乙腈(64:36,v/v) 1.8~4.9 4.3~11.7 77.2~102 UPLC-FLD[51] 鸡蛋 FF,CAP,TAP XTerra MS C18
(250 mm×4.6 mm,5 μm)乙腈-水(1:5,v/v) 20 69.5~99.8 HPLC-DAD[52] 罗非鱼和对虾 FF,CAP,TAP Waters Symmetry C18
(250 mm×4.6 mm, 5 μm)乙腈-水 10~20 81.0~102.9 HPLC-UVD[53] 牛奶 FF, CAP, TAP Venusil XBP C18
(250 mm×4.6 mm,5 μm)甲醇-水(40:60,v/v) 6.8~27.8 22.7~92.7 80.6~96.7 HPLC-UVD[54] 3. 液质联用法
质谱分析法是将物质离子化,通过质量分析器测定离子的质荷比,最终确定待测物质相对分子质量或分子结构的分析方法。液相色谱-串联质谱法结合了色谱和质谱技术,可对复杂基质中的各个成分进行定性和定量分析,在兽药残留检测方面应用尤为广泛。
电喷雾离子源(electrospray ionization,ESI)是LC-MS最常配备的离子源,从色谱柱中分离出来的样品会在离子源中被电离成离子,质谱检测器在多反应监测模式(multiple reaction monitoring,MRM)下对特定离子进行监测,从而达到检测FQs与CAPs残留的目的。目前,使用其他离子源的报道较少。CAP、TAP和FF需在负离子模式下进行全扫描,FFA在负离子模式下不离子化,需在正离子模式下进行全扫描。Alechaga等[55]采用UPLC-MS/MS测定动物源性食品中CAPs残留,详细对比了不同pH值和色谱柱对FFA的峰形和保留时间的影响,最终确定pH为5时,使用苯基-己基柱能实现快速且有效的分离,所得CCα为0.1~121 μg/kg,CCβ为0.2~138 μg/kg。
为避免测定结果受到基质干扰或出现假阳性现象,液质联用方法往往会通过改进前处理方法、使用同位素内标来减少基质效应对定量的影响。Xu等[56]采用磁性吸附剂以降低兽药残留的基质效应,并采用稳定同位素标记技术来校正猪肉基质干扰导致的FQs目标信号增强,最终使基质效应降低到88.4%~94.6%,所得回收率为88.6%~118.3%,RSD小于12.0%。Xie等[57]以d5-CAP为内标物,采用LC-ESI-MS/MS对鸡蛋中的CAP、TAP、FF和FFA残留,测得LOD为0.04~0.5 μg/kg、LOQ为0.1~1.5 μg/kg。但同位素内标法也有其局限性,同位素内标价格昂贵,质谱方法检测的药物越多,实验所付出的成本也就越高。
高分辨率质谱能准确分析出待测样品中各成分的精确质量数,从而实现目标化合物的分析和非目标化合物的筛查,不需要标准品就能完成对兽药残留的测定。近年来,有关FQs与CAPs残留的液相色谱高分辨率质谱方法报道较少,Rocha等[58]比较了相同条件下三重四极杆质谱(triple quadrupole tandem mass spectrometry,QqQ/MS)和飞行时间质谱(time of flight mass spectrometry,TOF/MS)测定家禽肌肉和肾脏样品中FQs残留的检测结果。结果表明,在定量方面,TOF/MS与QqQ/MS相比,无显著差异;在定性方面,TOF/MS可以实现非靶向的高通量定性筛查,这是QqQ/MS所不能相比的。Xu等[56]采用三重四极线性离子阱质谱(quadrupole-linear ion trap tandem mass spectrometry,Qtrap/MS)检测复杂基质中9种FQs残留,测得LODs和LOQs分别为0.5和1.5 μg/kg,使用了MRM-IDA-EPI的扫描模式,在MRM进行定量的同时,增强离子扫描(enhanced production scan,EPI)可提供所选定前体离子的二级质谱图,从而进一步确认FQs的存在,提高复杂样品中多残留检测的定性能力。
不同基质中FQs与CAPs残留的液质联用检测方法性能参数的比较见表4和表5。由表4和表5可知,两类药物都可以使用甲酸水溶液作为流动相的水相,因为甲酸可以用维持样品在流动相中的电离状态,提高电离效果,从而增加响应值。但部分研究[59-60]表明甲酸对负离子模式下的CAP、TAP、FF的信号响应有抑制作用。另外,当乙腈用作CAPs残留检测方法的流动相时,FFA在色谱柱上的保留时间过短,且不能和杂质有效分离,因此常用洗脱能力较弱的甲醇作为有机相。液质联用方法的LOD和LOQ普遍比液相色谱法低一个数量级,且能对更多基质和兽药残留进行同时测定,等度洗脱往往不能获得令人满意的效果,因此洗脱方式通常选用梯度洗脱。目前,已有动物源性食品中FQs与CAPs残留同时检测的串联质谱方法报道[61-63]。
表 4 不同基质中氟喹诺酮类药物残留的液质联用检测方法性能参数的比较Table 4. Comparison of performance parameters of LC-MS/MS detection methods for FQs in different matrices基质 药物 色谱柱 流动相 检测限LODs(μg/kg) 定量限LOQs(μg/kg) 回收率
(%)方法 巴沙鱼和对虾的可食用组织 ENR,CIP Polaris C18 Å (150 mm×
2.0 mm,3 μm)乙腈-甲酸水溶液(pH2.5) 4, 3 14,10 94~106 LC-MS/MS[64] 生牛乳和脱脂牛奶 15FQs,1QN Synergi Hydro-RP C18 80 Å (50 mm×2 mm,4 μm) 0.2%甲酸水-含0.2%甲酸的乙腈溶液 0.01~1.93 0.03~4.23 65~109 LC-MS/MS[65] 鸡胸肉 6FQs Waters Symmetry C18
(150 mm×4.6 mm,5 μm)含0.3%磷酸的乙腈溶液(pH3)-水(70:30,v/v) 2.7~6.7 8.9~22.1 66.0~97.2 LC-MS/MS[66] 罗非鱼和帕库鱼片 NOR,DAN,ENR,CIP XTerra RP 18(2.1 mm×
150 mm,5 μm)0.1%甲酸水-乙腈 63~126 76~152 90~111 LC-MS/MS[67] 河豚肌肉 5FQs Zorbax SB-C18(2.1 mm×
150 mm,3.6 μm)0.2%甲酸水-甲醇/乙腈(40:60,v/v) 0.3~1.5 1.0~4.6 LC-MS/MS[68] 鸡肌肉 CIP,ENR,NOR Agilent XDB C18
(150 mm×4.6 mm,5 μm)含0.1%甲酸的乙腈溶液-0.1%甲酸水 0.02~0.08 0.05~0.25 83.9~98.7 LC-MS/MS[69] 蜂蜜和牛奶 7FQs Phenomenex Kinetex C18
(100 mm×3.0 mm,2.6 μm)0.5%甲酸水-含0.5%甲酸的乙腈溶液 0.0010~0.0028 0.0033~0.059 74.5~116 HPLC-MS/MS[70] 鸡蛋 CIP, DAN, ENR, SAR Acquity UPLC BEH C18
(2.1 mm×50 mm,1.7 μm)0.1%甲酸水-甲醇-乙腈 <25 95~102 UPLC-MS/MS[71] 牛奶,动物肌肉,肝脏,肾脏,鸡蛋 6FQs Acquity UPLC BEH C18
(2.1 mm×50 mm,1.7 μm)0.2%甲酸水-乙腈 0.012~0.06 0.04~0.2 84.5~110.7 UPLC-MS/MS[72] 表 5 不同基质中酰胺醇类残留的液质联用检测方法性能参数的比较Table 5. Comparison of performance parameters of LC-MS/MS detection methods for CAPs in different matrices基质 药物 色谱柱 流动相 检测限LODs(μg/kg) 定量限LOQs(μg/kg) 回收率(%) 方法 液态奶,奶粉和
牛肌肉CAP,FF Phenomenex Luna C18
(50 mm×2.0 mm,5 μm)水/甲醇溶液-甲醇/水溶液(含5 mmol/L甲醇) 0.066~1.646 0.22~5.485 89~107 LC-MS/MS[73] 鲑鱼和罗非鱼
肌肉FF Hypersil Gold PFP
(10 mm×3 mm,3 μm)含0.1%甲酸的甲醇溶液-0.1%甲酸水 0.13,1.64 0.29, 4.13 89~106 LC-MS/MS[74] 尼罗罗非鱼 CAP,FF,TAP Phenomenex Aqua C18
(50 mm×2.0 mm,5.0 μm)0.1%甲酸水-含0.1%甲酸的甲醇溶液 0.15~12.5 79.8~92.0 LC-MS/MS[75] 鸡肌肉 CAP,TAP,FF,FFA Waters XBridge BEH C18-XP (100 mm×2.1 mm,2.5 μm) 乙腈-水 0.015~0.73 0.05~2.45 86.4~108.1 LC-MS/MS[76] 牛组织和鳗鱼 FF及其代谢产物 Inertsil ODS-4(150 mm×
2.1 mm,3 μm )0.1%乙酸-乙腈 0.5 10 93~104 LC-MS/MS[77] 猪肌肉 CAP,TAP,FF CNW Athena C18-WP
(100 mm×2.1 mm,3.0 μm)甲醇-水 0.10~0.14 0.25~0.47 97.5~117.0 HPLC-MS/MS[78] 猪肉、牛肉、
羊肉、鸡肉CAP,TAP,FF,FFA Waters Acquity UPLC HSS C18(50 mm×2.1 mm,1.8 μm) 水-乙腈 0.03~1.50 0.05~5.00 72~120 UPLC-MS/MS[79] 4. 小结与展望
样品前处理是兽药残留检测至关重要的环节,其萃取效果直接影响到检测的准确性和灵敏度。MSPE、DLLME、ASE等方法作为近年来FQs与CAPs残留的热门前处理方法,是通过改变温度、压强、接触表面积等因素,以达到提高萃取效率,简化实验步骤,节约有机溶剂的目的。此外,这些方法可以相互结合,充分利用它们的优势,来进一步提高样品的回收率。色谱和色谱质谱联用检测方法是兽药残留检测的主要手段,较其他方法而言,有着强大的定性和定量能力,正向着自动化、步骤简化和多残留检测的方向发展。HPLC-FLD和UPLC-FLD是氟喹诺酮类与酰胺醇类最为常见的液相色谱检测方法,两种药物本身就具有荧光性质,且FLD方法的LOD和LOQ低于DAD和UVD,因此FLD的相关报道较多。近年来,色谱质谱串联方法已发展较为成熟,能对复杂基质中兽药多残留进行准确测定,其LOD和LOQ通常比色谱法要低一个数量级,但在分析复杂样品时,需要尽量减少基质效应对测量结果的干扰。
近十年来,纳米材料结合固相萃取已取得了一些突破性的进展,所报道的前处理方法较之前明显增加,纳米材料能与目标物选择性结合,进而提高回收率,是液相色谱前处理方法的研究热点。但仍存在着制备较为繁琐、材料合成成本高、难以商业化生产的问题,如何将该方法与快速检测结合也有待后续研究。此外,动物源性食品中兽药多残留及非目标化合物的测定一直是液相色谱串联质谱的研究热点,但因为动物源性食品基质较为复杂、前处理方法各有优劣,研发出标准化的方法较为困难。随着全球对兽药残留的管控愈发严格,将液质联用法应用于兽药多残留检测已成为了一种趋势。虽然目前动物源性食品中FQs与CAPs的液质联用方法报道较多,但当前的研究多集中于三重四极杆质谱对兽药残留的检测。相信在未来具有更高质量精度和灵敏度的高分辨质谱也能被广泛的运用于兽药残留检测中,同时建立起完善的数据库,为检测结果的确定提供数据支撑,这有利于我国提高兽药残留检测效率,制定科学合理的残留标准,打破国际贸易壁垒,保证进出口食品安全。
-
表 1 动物源性食品中FQs与CAPs残留不同样品前处理方法优缺点比较
Table 1 Comparison of advantages and disadvantages of different sample pretreatment methods for FQs and CAPs residues in animal food
前处理方法 优点 缺点 液液萃取LLE 操作方便,设备简单 有机溶剂消耗量大,
萃取效率低分散液液萃取DLLME 萃取效率高,环境友好 需要离心分离,易受
基质效应干扰固相萃取SPE 有机溶剂用量少,可自动化
批量处理,回收率高提取时间长,成本高,
易受基质效应干扰磁性固相萃取MSPE 可重复利用,不需要过滤,
应用范围广,富集能力强材料制备困难,成本高 加速溶剂萃取ASE 自动化程度高,有机溶剂用量少 设备成本高 QuEChERS 操作简单,有机溶剂用量少,
应用范围广,提取效果好提取步骤较为繁琐 表 2 不同基质中FQs残留的液相色谱检测方法性能参数的比较
Table 2 Comparison of performance parameters of liquid chromatography detection methods for FQs in different matrices
基质 药物 色谱柱 流动相 检测限LODs(μg/kg) 定量限LOQs(μg/kg) 回收率(%) 方法 鸡肝 6FQs XTerra MS C18(3.0 mm×
150 mm,3.5 μm)乙腈-0.1%甲酸水(pH2.74) 5~19 23~62 83~102 LC-DAD[38] 牛奶和水 7FQs Kromasil C18(250 mm×
4.6 mm,5 μm)0.5%甲酸水-乙腈 0.10~0.26 0.32~0.85 68.8~120 HPLC-DAD[42] 牛奶 OFL,LOM,CIP Pack Pro C18(4.6 mm×
250 mm,5 μm)乙腈-水(1:9,v/v) 1.5~2.3 5.0~7.5 87.2~106.1 LC-UVD[43] 鱼 NOR,OFL,CIP Venusil XBP C18
(150 mm×4.6 mm,5 μm)乙腈-13 mmol/L四丁基溴化铵溶液(6:94,v/v) 2.65~3.65 8.82~12.16 69.3~102.8 HPLC-UVD[44] 猪、牛、鸡、鱼组织 15FQs Zorbax SB-Aq-C18
(250 mm×4.6 mm,5 μm)甲醇-乙腈-0.02 mol/L柠檬酸和0.03 mol/L醋酸铵 3 10 70.6~111.1 HPLC-UVD[45] 猪肝和湖水 NOR Waters Symmery C18
(4.6 mm×150 mm,5 μm)乙腈-0.4%磷酸和0.4%三乙胺水溶液(15:85,v/v) 6 77.4~97.1 UPLC-UVD[46] 牛奶 OFL,PEF,ENR,DIF Atlantis T3 100Å
(250 mm×4.6 mm,5 μm)乙酸-醋酸钠缓冲液(pH5.4)/乙腈(75:25,v/v) 6~60 94.5~100.8 HPLC-ECL[37] 鸡蛋 7FQs and 3QNs Phenomenex Luna C8
(150 mm×4.6 mm,5 μm)乙腈-0.02 mol/L草酸(pH4.0) 3~10 10~30 92~99 LC-FLD[47] 猪、牛、鸡、鱼的
肌肉组织5FQs Zorbax Eclipse XDB-C18
(150 mm×4.6 mm,5 μm)甲醇-乙腈-磷酸/三乙胺水溶液(15:5:80,v/v) 0.056~0.64 0.187~2.134 87.2~110.6 HPLC-FLD[48] 蜂蜜和水 5FQs Thermo C18(250 mm×
4.6 mm,5 μm)0.5%甲酸水-乙腈 0.14~0.61 0.39~1.1 80.1~119 HPLC-FLD[49] 注:恩诺沙星(enrofloxacin,ENR)、氧氟沙星(ofloxacin,OFL)、洛美沙星(lomefloxacin,LOM)、培氟沙星(pefloxacin,PEF),达氟沙星(danofloxacin,DFLX)、二氟沙星(difloxacin,DIF)。 表 3 不同基质中CAPs残留的液相色谱检测方法性能参数的比较
Table 3 Comparison of performance parameters of liquid chromatography detection methods for CAPs in different matrices
基质 兽药 色谱柱 流动相 检测限LODs(μg/kg) 定量限LOQs(μg/kg) 回收率
(%)方法 鸡肉 FF,FFA,TAP Hedera C18(250 mm×
4.6 mm,5 μm)0.01 mol/L NaH2PO4,0.005 mol/L十二烷基硫酸钠和0.1%三乙胺-乙腈(68:32,v/v) 0.5~1.5 2~5 78.5~88.7 HPLC-FLD[50] 禽蛋 TAP,FF,FFA Acquity UPLC BEH C18(2.1 mm×100 mm,1.7 μm) 0.005 mol/L NaH2PO4,0.003 mol/L十二烷基硫酸钠和0.05%三乙胺(pH5.3±0.1)-乙腈(64:36,v/v) 1.8~4.9 4.3~11.7 77.2~102 UPLC-FLD[51] 鸡蛋 FF,CAP,TAP XTerra MS C18
(250 mm×4.6 mm,5 μm)乙腈-水(1:5,v/v) 20 69.5~99.8 HPLC-DAD[52] 罗非鱼和对虾 FF,CAP,TAP Waters Symmetry C18
(250 mm×4.6 mm, 5 μm)乙腈-水 10~20 81.0~102.9 HPLC-UVD[53] 牛奶 FF, CAP, TAP Venusil XBP C18
(250 mm×4.6 mm,5 μm)甲醇-水(40:60,v/v) 6.8~27.8 22.7~92.7 80.6~96.7 HPLC-UVD[54] 表 4 不同基质中氟喹诺酮类药物残留的液质联用检测方法性能参数的比较
Table 4 Comparison of performance parameters of LC-MS/MS detection methods for FQs in different matrices
基质 药物 色谱柱 流动相 检测限LODs(μg/kg) 定量限LOQs(μg/kg) 回收率
(%)方法 巴沙鱼和对虾的可食用组织 ENR,CIP Polaris C18 Å (150 mm×
2.0 mm,3 μm)乙腈-甲酸水溶液(pH2.5) 4, 3 14,10 94~106 LC-MS/MS[64] 生牛乳和脱脂牛奶 15FQs,1QN Synergi Hydro-RP C18 80 Å (50 mm×2 mm,4 μm) 0.2%甲酸水-含0.2%甲酸的乙腈溶液 0.01~1.93 0.03~4.23 65~109 LC-MS/MS[65] 鸡胸肉 6FQs Waters Symmetry C18
(150 mm×4.6 mm,5 μm)含0.3%磷酸的乙腈溶液(pH3)-水(70:30,v/v) 2.7~6.7 8.9~22.1 66.0~97.2 LC-MS/MS[66] 罗非鱼和帕库鱼片 NOR,DAN,ENR,CIP XTerra RP 18(2.1 mm×
150 mm,5 μm)0.1%甲酸水-乙腈 63~126 76~152 90~111 LC-MS/MS[67] 河豚肌肉 5FQs Zorbax SB-C18(2.1 mm×
150 mm,3.6 μm)0.2%甲酸水-甲醇/乙腈(40:60,v/v) 0.3~1.5 1.0~4.6 LC-MS/MS[68] 鸡肌肉 CIP,ENR,NOR Agilent XDB C18
(150 mm×4.6 mm,5 μm)含0.1%甲酸的乙腈溶液-0.1%甲酸水 0.02~0.08 0.05~0.25 83.9~98.7 LC-MS/MS[69] 蜂蜜和牛奶 7FQs Phenomenex Kinetex C18
(100 mm×3.0 mm,2.6 μm)0.5%甲酸水-含0.5%甲酸的乙腈溶液 0.0010~0.0028 0.0033~0.059 74.5~116 HPLC-MS/MS[70] 鸡蛋 CIP, DAN, ENR, SAR Acquity UPLC BEH C18
(2.1 mm×50 mm,1.7 μm)0.1%甲酸水-甲醇-乙腈 <25 95~102 UPLC-MS/MS[71] 牛奶,动物肌肉,肝脏,肾脏,鸡蛋 6FQs Acquity UPLC BEH C18
(2.1 mm×50 mm,1.7 μm)0.2%甲酸水-乙腈 0.012~0.06 0.04~0.2 84.5~110.7 UPLC-MS/MS[72] 表 5 不同基质中酰胺醇类残留的液质联用检测方法性能参数的比较
Table 5 Comparison of performance parameters of LC-MS/MS detection methods for CAPs in different matrices
基质 药物 色谱柱 流动相 检测限LODs(μg/kg) 定量限LOQs(μg/kg) 回收率(%) 方法 液态奶,奶粉和
牛肌肉CAP,FF Phenomenex Luna C18
(50 mm×2.0 mm,5 μm)水/甲醇溶液-甲醇/水溶液(含5 mmol/L甲醇) 0.066~1.646 0.22~5.485 89~107 LC-MS/MS[73] 鲑鱼和罗非鱼
肌肉FF Hypersil Gold PFP
(10 mm×3 mm,3 μm)含0.1%甲酸的甲醇溶液-0.1%甲酸水 0.13,1.64 0.29, 4.13 89~106 LC-MS/MS[74] 尼罗罗非鱼 CAP,FF,TAP Phenomenex Aqua C18
(50 mm×2.0 mm,5.0 μm)0.1%甲酸水-含0.1%甲酸的甲醇溶液 0.15~12.5 79.8~92.0 LC-MS/MS[75] 鸡肌肉 CAP,TAP,FF,FFA Waters XBridge BEH C18-XP (100 mm×2.1 mm,2.5 μm) 乙腈-水 0.015~0.73 0.05~2.45 86.4~108.1 LC-MS/MS[76] 牛组织和鳗鱼 FF及其代谢产物 Inertsil ODS-4(150 mm×
2.1 mm,3 μm )0.1%乙酸-乙腈 0.5 10 93~104 LC-MS/MS[77] 猪肌肉 CAP,TAP,FF CNW Athena C18-WP
(100 mm×2.1 mm,3.0 μm)甲醇-水 0.10~0.14 0.25~0.47 97.5~117.0 HPLC-MS/MS[78] 猪肉、牛肉、
羊肉、鸡肉CAP,TAP,FF,FFA Waters Acquity UPLC HSS C18(50 mm×2.1 mm,1.8 μm) 水-乙腈 0.03~1.50 0.05~5.00 72~120 UPLC-MS/MS[79] -
[1] XU Z, ZHAO S J, LV Z S, et al. Fluoroquinolone-isatin hybrids and their biological activities. Eur[J]. Medicinal Chemistry,2019,162:396−406.
[2] PAN X, WANG P, HU N, et al. A Physiologically based pharmacokinetic model characterizing theophylline/an tofloxacin interaction in both rats and humans[J]. Drug Metabolism and Pharmacokinetics,2011,26(4):387−398. doi: 10.2133/dmpk.DMPK-10-RG-126
[3] 吴俊珍, 刘笑芬. 欧洲药品管理局建议限制喹诺酮类和氟喹诺酮类抗菌药物的使用[J]. 中国感染与化疗杂志,2019,19(6):589. [WU J Z, LIU X F. The European Drug Administration recommends limiting the use of quinolones and fluoroquinolones[J]. Chinese Journal of Infection and Chemotherapy,2019,19(6):589. [4] HANEKAMP J C, BAST A. Antibiotics exposure and health risks: Chloramphenicol[J]. Environmental Toxicology & Pharmacology,2015,39(1):213−220.
[5] 赵霞, 王波, 谢恺舟, 等. 动物性食品中氯霉素类药物残留色谱和质谱检测技术研究进展[J]. 分析试验室,2018,37(8):978−985. [ZHAO X, WANG B, XIE K Z, et al. An overview on chromatographic and mass spectrometry analytical methods for the detection of chloramphenicols residues in animal food[J]. Chinese Journal of Analysis Laboratory,2018,37(8):978−985. doi: 10.13595/j.cnki.issn1000-0720.2018.0190 [6] GB 1650-2019 食品安全国家标准 食品中兽药最大残留限量[S]. 北京: 中国标准出版社, 2019. GB 1650-2019 National food safety standard. Maximum residue limits for veterinary drugs in foods[S]. Beijing: China Standard Press, 2019.
[7] European Commission. Commission Regulation (EU) No 37/2010 of 22 December 2009 on pharmacologically active sub-stances and their classification regarding maximum residue limits in foodstuffs of animal origin[S].
[8] 周纯洁, 候美玲, 何春兰, 等. LC-MS/MS技术在食品中农药多残留分析的应用进展[J]. 食品工业科技,2019,40(13):283−286,298. [ZHOU C J, HOU M L, HE C L, et al. Progress on application of LC-MS/MS technology in pesticide multiresidue analysis for food[J]. Science and Technology of Food Industry,2019,40(13):283−286,298. doi: 10.13386/j.issn1002-0306.2019.13.047 [9] BARRETO F, RIBEIRO C, HOFF R, et al. Development and validation of a high-throughput method for determination of nine fluoroquinolones residues in muscle of different animal species by liquid chromatography coupled to tandem mass spectrometry with low temperature clean up[J]. Journal of Chromatography A,2017,1521:131−139. doi: 10.1016/j.chroma.2017.09.036
[10] XIAO Z M, SONG R, RAO Z R, et al. Development of a subcritical water extraction approach for trace analysis of chloramphenicol, thiamphenicol, florfenicol, and florfenicol amine in poultry tissues[J]. Journal of Chromatography A,2015,1418:29−35. doi: 10.1016/j.chroma.2015.09.047
[11] REZAIE M, ASSADI Y, AGHAEE E. Determination of organic compounds in water using dispersive liquid-liquid microextarction[J]. Journal of Chromatography A,2006,1116(1−2):1−9. doi: 10.1016/j.chroma.2006.03.007
[12] 孙鹏, 乔石, 郭秀娟, 等. 离子液体分散液相微萃取-HPLC法测定液态奶中恩诺沙星和环丙沙星[J]. 药物分析杂志,2017,37(1):111−116. [SUN P, QIAO S, GUO X J, et al. Determination of enrofloxacin and ciprofloxacin in liquid milk by ionic liquid-based dispersive liquid-phase microextraction coupled with HPLC[J]. Chinese Journal of Pharmaceutical Analysis,2017,37(1):111−116 (in Chinese). doi: 10.16155/j.0254-1793.2017.01.14 [13] KARAMI-OSBOOA R, MIRIA R, JAVIDNIAA K, et al. Simultaneous chloramphenicol and florfenicol determination by a validated DLLME-HPLC-UV method in pasteurized milk[J]. Iranian Journal of Pharmaceutical Research Ijpr,2016,15(3):361−368.
[14] 李燕莹, 周庆琼, 陈羽中, 等. 磁固相萃取在食品分析中的研究进展[J]. 食品工业科技,2019,40(8):323−330,336. [LI Y Y, ZHOU Q Q, CHEN Y Z, et al. Research progress of magnetic solid phase extraction in food analysis[J]. Science and Technology of Food Industry,2019,40(8):323−330,336. doi: 10.13386/j.issn1002-0306.2019.08.054 [15] VAKH C, ALABOUD M, LEBEDINETS S, et al. An automated magnetic dispersive micro-solid phase extraction in a fluidized reactor for the determination of fluoroquinolones in baby food samples[J]. Analytica Chimica Acta,2018,1001:59−69. doi: 10.1016/j.aca.2017.11.065
[16] BAGHERI N, AL LAWATI H, AL SHARJI N, et al. Magnetic zinc based 2D-metal organic framework as an efficient adsorbent for simultaneous determination of fluoroquinolones using 3D printed microchip and liquid chromatography tandem mass spectrometry[J]. Talanta,2021,224:121796. doi: 10.1016/j.talanta.2020.121796
[17] HUANG S, GAN N, LIU H, et al. Simultaneous and specific enrichment of several amphenicol antibiotics residues in food based on novel aptamer functionalized magnetic adsorbents using HPLC-DAD[J]. Journal of Chromatography B,2017,1060:247−254. doi: 10.1016/j.jchromb.2017.05.035
[18] HE X, WANG G, YANG K, et al. Magnetic graphene dispersive solid phase extraction combining high performance liquid chromatography for determination of fluoroquinolones in foods[J]. Food Chemistry,2017,221:1226−1231. doi: 10.1016/j.foodchem.2016.11.035
[19] WEN A, LI G, WU D, et al. Sulphonate functionalized covalent organic framework-based magnetic sorbent for effective solid phase extraction and determination of fluoroquinolones[J]. Journal of Chromatography A,2020,1612:460651. doi: 10.1016/j.chroma.2019.460651
[20] TIAN C, REN X, HE M, et al. Core-shell magnetic porous organic polymer for magnetic solid-phase extraction of fluoroquinolone antibiotics in honey samples followed by HPLC with fluorescence detection[J]. Journal of Separation Science,2021:1−9.
[21] SAMANIDOU V, KEHAGIA M, KABIR A, et al. Matrix molecularly imprinted mesoporous sol-gel sorbent for efficient solid-phase extraction of chloramphenicol from milk[J]. Analytica Chimica Acta,2016:62−74.
[22] ANASTASIADES M, LEHOTAY S J, ŠTAJNBAHER D, et al. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and ''dispersive solid-phase extraction'' for the determination of pesticide residues in produce[J]. Journal of Aoac International,2003,86:412−431. doi: 10.1093/jaoac/86.2.412
[23] LEHOTAY S J. Determination of pesticide residues in foods by acetonitrile extraction and partitioning with magnesium sulfate: collaborative study[J]. Journal of Aoac International,2007,90:485−520. doi: 10.1093/jaoac/90.2.485
[24] European Committee for Standardization (CEN) Standard Method EN 15662. Food of plant origin-determination of pesticide residues using GC-MS and/or LC-MS/MS following acetonitrile extraction/partitioning and clean-up by dispersive SPE-QuEChERS method[S]. 2008.
[25] 韩臣波. QuEChERS-HPLC-MSMS快速测定牛奶中的喹诺酮类兽药残留[J]. 化学工程师,2020(34):32−35. [HAN C B. Rapid determination of six quinolones in milk by HPLC-MS/MS[J]. Chemical Engineer,2020(34):32−35 (in Chinese). doi: 10.16247/j.cnki.23-1171/tq.20201132 [26] LIU H Y, LIN S L, FUH M R. Determination of chloramphenicol, thiamphenicol and florfenicol in milk and honey using modified QuEChERS extraction coupled with polymeric monolith-based capillary liquid chromatography tandem mass spectrometry[J]. Talanta,2016,150:233−239. doi: 10.1016/j.talanta.2015.12.045
[27] ZHENG H B, DING J, ZHENG S J, et al. Magnetic ''one-step'' quick, easy, cheap, effective, rugged and safe method for the fast determination of pesticide residues in freshly squeezed juice[J]. Journal of Chromatography A,2015,1398:1−10. doi: 10.1016/j.chroma.2015.04.021
[28] WU X, ZHANG H, MENG L, et al. Graphene for cleanup in trace analysis of pyrethroid insecticides in cucumber and spinach[J]. Chromatographia,2012,75(19-20):1177−1183. doi: 10.1007/s10337-012-2299-0
[29] WU X, ZHANG R, LIU X, et al. Evaluation of graphene for effective cleanup of fruit and vegetable extracts in pesticide residue analysis[J]. Food Analytical Methods,2015,8(1):243−253. doi: 10.1007/s12161-014-9868-9
[30] LIU H, LIN T, LIN X, et al. QuEChERS with magnetic hydrophilic-lipophilic balanced adsorbent and its application in multi-class veterinary residues in milk by ultra high-performance liquid chromatography-tandem mass spectrometry[J]. Chromatographia,2018,81(2):265−275. doi: 10.1007/s10337-017-3433-9
[31] 魏丹, 国明, 张菊. 加速溶剂萃取-磁固相萃取-高效液相色谱-串联质谱法测定水产品中10种氟喹诺酮类药物残留[J]. 色谱,2020,38(12):1413−1422. [WEI D, GUO M, ZHANG J. Determination of 10 fluoroquinolones residues in aquaticproducts by accelerated solvent extraction, magnetic solid-phase extraction, and high-performance liquid chromatography-tandem mass spectrometry[J]. Chinese Journal of Chromatography,2020,38(12):1413−1422. [32] WANG B, PANG M, ZHAO X, et al. Development and comparison of liquid-liquid extraction and accelerated solvent extraction methods for quantitative analysis of chloramphenicol, thiamphenicol, florfenicol, and florfenicol amine in poultry eggs[J]. Journal of mass spectrometry,2019,54(6):488−494. doi: 10.1002/jms.4355
[33] 谢恺舟, 姚宜林, 徐东, 等. 高效液相色谱荧光检测法同时检测鸡蛋中氟苯尼考及其代谢物氟苯尼考胺残留[J]. 中国兽医杂志,2011,47(8):74−77. [XIE K Z, YAO Y L, XU D, et al. Simultaneous determination of residues of forfenicol and the metabolite forfenicol amine in hen eggs by HPLC with fluorescence detection[J]. Chinese Journal of Veterinary Medicine,2011,47(8):74−77. doi: 10.3969/j.issn.0529-6005.2011.08.031 [34] GRANJA R, DE LIMA A, PATEL R, et al. Monitoring of florfenicol residues in fish muscle by HPLC-UV with confirmation of suspect results by LC-MS/MS[J]. Drug Testing and Analysis,2012:125−129.
[35] CHOI J H, NA T W, MAMUN M L, et al. Bufferized solvent extraction and HPLC fluorometric detection method for sarafloxacin in pig and chicken muscles[J]. Biomedical Chromatography,2011,25(3):405−411. doi: 10.1002/bmc.1463
[36] MOEMA D, NINDI M M, DUBE S. Development of a dispersive liquid-liquid microextraction method for the determination of fluoroquinolones in chicken liver by high performance liquid chromatography[J]. Analytica Chimica Acta,2012,730:80−86. doi: 10.1016/j.aca.2011.11.036
[37] LI Y, ZHANG Z, LI J, et al. Simple, stable and sensitive electrogenerated chemiluminescence detector for high-performance liquid chromatography and its application in direct determination of multiple fluoroquinolone residues in milk[J]. Talanta,2011,84(3):690−695. doi: 10.1016/j.talanta.2011.01.068
[38] 陈玲珍, 杨洪生, 吴光红, 等. 高效液相色谱-荧光法测定水产品中氟苯尼考的残留量[J]. 理化检验(化学分册),2010,46(6):654−656,659. [CHEN Z L, YANG H S, WU G H, et al. HPLC Determination of residual amount of florfenicol in aquatic products with fluorimetric detection[J]. Physical Testing and Chemical Analysis (Part B:Chemical Analysis),2010,46(6):654−656,659. [39] 周晖, 陈燕, 迟秋池, 等. 动物源性食品中多种兽药残留检测的研究进展[J]. 食品安全质量检测学报,2019,10(10):2889−2895. [ZHOU H, CHEN Y, CHI Q C, et al. Research progress on detection of various veterinary drug residue[J]. Journal of Food Safety & Quality,2019,10(10):2889−2895. doi: 10.3969/j.issn.2095-0381.2019.10.011 [40] AUFARTOVÁ J, BRABCOVÁ I, TORRES-PADRÓN M, et al. Determination of fluoroquinolones in fishes using microwave-assisted extraction combined with ultra-high performance liquid chromatography and fluorescence detection[J]. Journal of Food Composition & Analysis,2017,56:140−146.
[41] 刁志祥, 王波, 张培杨, 等. 加速溶剂萃取/超高效液相色谱-荧光法检测鸽蛋和鹌鹑蛋中氟苯尼考和氟苯尼考胺残留[J]. 福建农业学报,2019,34(11):1315−1322. [DIAO Z X, WANG B, ZHANG P Y, et al. ASE/UHPLC-FLD determination of florfenicol and florfenicol amine residues in pigeon and quail eggs[J]. Fujian Journal of Agricultural Sciences,2019,34(11):1315−1322. [42] MEI M, HUANG X J. Determination of fluoroquinolones in environmental water and milk samples treated with stir cake sorptive extraction based on a boron-rich monolith[J]. Journal of Separation Science,2016,39(10):1908−1918. doi: 10.1002/jssc.201600232
[43] WANG H, WANG R, HAN Y, et al. Preparation of molecular imprinted microspheres based on inorganic-organic co-functional monomer for miniaturized solid-phase extraction of fluoroquinolones in milk[J]. Journal of Chromatography B,2014,949-950:24−29. doi: 10.1016/j.jchromb.2013.10.042
[44] LÜ Y K, MA Y, ZHAO X B, et al. Grafting of norfloxacin imprinted polymeric membranes on silica surface for the selective solid-phase extraction of fluoroquinolones in fish samples[J]. Talanta,2012,89:270−275. doi: 10.1016/j.talanta.2011.12.026
[45] YU H, TAO Y F, CHEN D M, et al. Simultaneous determination of fluoroquinolones in foods of animal origin by a high performance liquid chromatography and a liquid chromatography tandem mass spectrometry with accelerated solvent extraction[J]. Journal of Chromatography B,2012,s885-886(5):150−159.
[46] ZHANG Y, XIE Y, ZHANG C, et al. Preparation of porous magnetic molecularly imprinted polymers for fast and specifically extracting trace norfloxacin residue in pork liver[J]. Journal of Separation Science,2020,43(2):478−485. doi: 10.1002/jssc.201900589
[47] WANG B, XIE X, ZHAO X, et al. Development of an accelerated solvent extraction-ultra-performance liquid chromatography-fluorescence detection method for quantitative analysis of thiamphenicol, florfenicol and florfenicol amine in poultry eggs[J]. Molecules,2019,24:1830. doi: 10.3390/molecules24091830
[48] WANG H, GAO M, XU Y, et al. A phase separation method for analyses of fluoroquinones in meats based on ultrasound-assisted salt-induced liquid-liquid microextraction and a new integrated device[J]. Meat Science,2015,106:61−68. doi: 10.1016/j.meatsci.2015.03.023
[49] PANG J, LIAO Y, HUANG X, et al. Metal-organic framework-monolith composite-based in-tube solid phase microextraction on-line coupled to high-performance liquid chromatography-fluorescence detection for the highly sensitive monitoring of fluoroquinolones in water and food samples[J]. Talanta,2019,199:499−506. doi: 10.1016/j.talanta.2019.03.019
[50] 谢恺舟, 徐东, 陈书琴, 等. 高效液相色谱荧光检测法同时检测鸡肉中甲砜霉素、氟苯尼考及氟苯尼考胺残留[J]. 分析试验室,2011,30(7):31−35. [XIE K Z, XU D, CHEN M Q, et al. Simultaneous determination of thiamphenicol, florfenicol and florfenicol amine residues in chicken by high performance liquid chromatography with fluorescence detection[J]. Chinese Journal of Analysis Laboratory,2011,30(7):31−35. doi: 10.3969/j.issn.1000-0720.2011.07.008 [51] GAJDA A, POSYNIAK A, ZMUDZKI J, et al. Determination of (fluoro) quinolones in eggs by liquid chromatography with fluorescence detection and confirmation by liquid chromatography–tandem mass spectrometry[J]. Food Chemistry,2012,135(2):430−439. doi: 10.1016/j.foodchem.2012.04.106
[52] 罗昭军, 陈幸, 单乃荣, 等. 高效液相色谱法检测鸡蛋中氯霉素类药物残留[J]. 中国畜牧兽医文摘,2018,34(3):72−74. [LUO Z J, CHEN X, CHAN N R, et al. Determination of chloramphenicol residues in eggs by HPLC[J]. Chinese Animal Husbandry Veterinary Medicine Abstract,2018,34(3):72−74. [53] 陶昕晨, 黄和, 李志清, 等. 水产品中氯霉素、甲砜霉素和氟苯尼考的HPLC-UVD法测定[J]. 食品工业科技,2012,33(20):80−83. [TAO Y C, HUANG H, LI Z Q, et al. Multi-residue detection of thiamphenicol, florfenicol and florfenicol-amine in aquatic product by ELISA[J]. Science and Technology of Food Industry,2012,33(20):80−83. doi: 10.13386/j.issn1002-0306.2012.20.017 [54] 吕运开, 张婧琦, 王晓虎, 等. 基于分子印迹聚合物微球的在线固相萃取-液相色谱联用技术测定牛奶中酰胺醇类药物残留[J]. 食品科学,2013,34(16):238−242. [LÜ Y K, ZHANG J Q, WANG X H, et al. On-line solid-phase extraction using molecularly imprinted polymer microspheres in combination with HPLC for the determination of amphenicols residues in milk[J]. Food Science,2013,34(16):238−242. doi: 10.7506/spkx1002-6630-201316048 [55] ALECHAGA L, MOYANO E, GALCERAN M T. Ultra-high performance liquid chromatography-tandem mass spectrometry for the analysis of phenicol drugs and florfenicol-amine in foods[J]. Analyst,2012,137(10):2486−2494. doi: 10.1039/c2an16052h
[56] XU F, LIU F, WANG C Z, et al. Use of phenyl/tetrazolyl-functionalized magnetic microspheres and stable isotope labeled internal standards for significant reduction of matrix effect in determination of nine fluoroquinolones by liquid chromatography-quadrupole linear ion trap mass spectrometry[J]. Analytical and bioanalytical chemistry,2018,b,410(6):1709−1724.
[57] XIE X, WANG B, PANG M, et al. Quantitative analysis of chloramphenicol, thiamphenicol, florfenicol and florfenicol amine in eggs via liquid chromatography-electrospray ionization tandem mass spectrometry[J]. Food Chemistry,2018,269:542−548. doi: 10.1016/j.foodchem.2018.07.045
[58] ROCHA D G, SANTOS F A, DA SILVA J C, et al. Multiresidue determination of fluoroquinolones in poultry muscle and kidney according to the regulation 2002/657/EC. A systematic comparison of two different approaches: Liquid chromatography coupled to high-resolution mass spectrometry or tandem mass spectrometry[J]. Journal of Chromatography A,2015,1379:83−91. doi: 10.1016/j.chroma.2014.12.058
[59] 陈蔷, 宋志超, 张崇威, 等. 动物源食品中酰胺醇类药物及其代谢物残留检测超高效液相色谱-串联质谱法研究[J]. 中国兽药杂志,2015,49(8):28−34. [CHEN Q, SONG Z C, ZHANG C W, et al. Determination of amphenicols and metabolite residues in animal derived food by UPLC-MS/MS[J]. Chinese Journal of Veterinary Medicine,2015,49(8):28−34. [60] 施祖灏, 张小燕, 林伊莱, 等. 同位素稀释高效液相色谱串联质谱法同时测定鸡蛋中酰胺醇类药物及其代谢物[J]. 农产品质量与安全,2012(S1):11−16. [SHI Z H, ZHANG X Y, LIN Y L, et al. Simultaneous determination of amidol drugs and their metabolism in eggs by isotope dilution high performance liquid chromatography tandem mass spectrometry[J]. Quality and Safety of Agro-Products,2012(S1):11−16. doi: 10.3969/j.issn.1674-8255.2012.z1.003 [61] TURNIPSEED S B, CLARK S B, STOREY J M, et al. Analysis of veterinary drug residues in frog legs and other aquacultured species using liquid chromatography quadrupole time-of-flight mass spectrometry[J]. Journal of Agricultural and Food Chemistry,2012,60(18):4430−4439. doi: 10.1021/jf2049905
[62] GIBBS R S, MURRAY S L, WATSON L V, et al. Development and validation of a hybrid screening and quantitative method for the analysis of eight classes of therapeutants in aquaculture products by liquid chromatography-tandem mass spectrometry[J]. Journal of Agricultural & Food Chemistry,2018,66(20):4997−5008.
[63] CHEN Y, XIA S, HAN X, et al. Simultaneous determination of malachite green, chloramphenicols, sulfonamides, and fluoroquinolones residues in fish by liquid chromatography-mass spectrometry[J]. Journal of Analytical Methods in Chemistry,2020:1−12.
[64] DANYI S, WIDART J, DOUNY C, et al. Determination and kinetics of enrofloxacin and ciprofloxacin in Tra catfish (Pangasianodon hypophthalmus) and giant freshwater prawn (Macrobrachium rosenbergii) using a liquid chromatography/mass spectrometry method[J]. Journal of Veterinary Pharmacology & Therapeutics,2011,34(2):142−152.
[65] KANTIANI L, MARINELLA F, DAMIÀ B. Rapid residue analysis of fluoroquinolones in raw bovine milk by online solid phase extraction followed by liquid chromatography coupled to tandem mass spectrometry[J]. Journal of Chromatography A,2011,1218(50):9019−9027. doi: 10.1016/j.chroma.2011.09.079
[66] XU H Y, CHEN L G, SUN L, et al. Microwave-assisted extraction and in situ clean-up for the determination of fluoroquinolone antibiotics in chicken breast muscle by LC-MS/MS[J]. Journal of Separation Science,2010,34(2):142−149.
[67] QUESADA S P, PASCHOAL J A, REYES F G. A simple method for the determination of fluoroquinolone residues in tilapia (Oreochromis niloticus) and pacu (Piaractus mesopotamicus) employing LC-MS/MS QToF[J]. Food Additives and Contaminants Part A-Chemistry Analysis Control Exposure & Risk Assessment,2013,30:813.
[68] TANG Y J, XU J Q, CHEN L, et al. Rapid in vivo determination of fluoroquinolones in cultured puffer fish (Takifugu obscurus) muscle by solid-phase microextraction coupled with liquid chromatography-tandem mass spectrometry[J]. Talanta,2017:550−556.
[69] LI X D, CHEN Y H, CHEN S B, et al. Polyethyleneimine-functionalized Fe3O4/attapulgite particles for hydrophilic interaction-based magnetic dispersive solid-phase extraction of fluoroquinolones in chicken muscle[J]. Analytical and Bioanalytical Chemistry,2021,413(13):3529−3540. doi: 10.1007/s00216-021-03304-9
[70] CHEN L, HUANG X J. Sensitive monitoring of fluoroquinolones in milk and honey using multiple monolithic fiber solid-phase microextraction coupled to liquid chromatography tandem mass spectrometry[J]. Journal of Agricultural and Food Chemistry,2016,64(45):8684−8693. doi: 10.1021/acs.jafc.6b03965
[71] RIBEIRO C C, ORLANDO R M, ROHWEDDER J J, et al. Electric field-assisted solid phase extraction and cleanup of ionic compounds in complex food matrices: Fluoroquinolones in eggs[J]. Talanta,2016,152:498−503. doi: 10.1016/j.talanta.2016.02.047
[72] JIAN N G, ZHAO M, LIANG S H, et al. High-throughput and high-efficient micro-solid phase extraction based on sulfonated-polyaniline/polyacrylonitrile nanofiber mats for determination of fluoroquinolones in animal-origin foods[J]. Journal of Agricultural and Food Chemistry,2019,67(24):6892−6901. doi: 10.1021/acs.jafc.9b01312
[73] REZENDE D R, FILHO N F, ROCHA G L. Simultaneous determination of chloramphenicol and florfenicol in liquid milk, milk powder and bovine muscle by LC-MS/MS[J]. Food Additives & Contaminants: Part A,2012,29:559−570.
[74] WAI-MEI S D, CLARE H, YIU-TUNG W. Phenylboronic acid solid phase extraction cleanup and isotope dilution liquid chromatography-tandem mass spectrometry for the determination of florfenicol amine in fish muscles[J]. Journal of Aoac International,2015,98(3):566−574. doi: 10.5740/jaoacint.14-267
[75] GUIDI L R, TETTE P A S, GLORIA M B A, et al. A simple and rapid LC-MS/MS method for the determination of amphenicols in Nile tilapia[J]. Food Chemistry,2018,262:235−241. doi: 10.1016/j.foodchem.2018.04.087
[76] IMRAN M, HABIB F, MAJEED S, et al. LC-MS/MS based determination of chloramphenicol, thiamphenicol, florfenicol and florfenicol amine in poultry meat from the Punjab-Pakistan[J]. Food Additives & Contaminants Part A,2018,35(8):1530−1542.
[77] SAITO-SHIDA S, KOUJI S, NARUSHIMA J, et al. Determination of total florfenicol residues as florfenicol amine in bovine tissues and eel by liquid chromatography-tandem mass spectrometry using external calibration[J]. Journal of Chromatography B,2019,1109:37−44. doi: 10.1016/j.jchromb.2019.01.018
[78] 王东鹏, 叶诚, 李小莎. Captiva EMR-Lipid固相萃取结合高效液相色谱-串联质谱法同时测定猪肉中氯霉素类药物残留量[J]. 食品安全质量检测学报,2021,12(19):7660−7666. [WANG D P, YE C, LI X S. Simultaneous determination of chloramphenicol drugs residues in pork by Captiva EMR-Lipid solid phase extraction combined with high performance liquid chromatography-tandem mass spectrometry[J]. Journal of Food Safety & Quality,2021,12(19):7660−7666. [79] WU X Y, SHEN X X, CAO X Y, et al. Simultaneous determination of amphenicols and metabolites in animal-derived foods using ultrahigh-performance liquid chromatography-tandem mass spectrometry[J]. International Journal of Analytical Chemistry,2021:3613670.
-
期刊类型引用(0)
其他类型引用(1)





 下载:
下载:
 下载:
下载:



