| [1] |
MIZUMOTO S, YAMADA S, SUGAHARA K. Molecular interactions between chondroitin-dermatan sulfate and growth factors/receptors/matrix proteins[J]. Current Opinion in Structural Biology,2015,34:35−42. doi: 10.1016/j.sbi.2015.06.004
|
| [2] |
PECCHI E, PRIAM S, MLADENOVIC Z, et al. 156 Inhibition of prostaglandin e2 and matrix metalloproteinases synthesis in interleukin-1b-stimulated osteoblasts: A potential role of chondroitin sulfate on bone in osteoarthritis[J]. Osteoarthritis & Cartilage,2010,18:S77−S77.
|
| [3] |
ROMAN-BLAS J A, MEDIERO A, TARDÍO L, et al. The combined therapy with chondroitin sulfate plus glucosamine sulfate or chondroitin sulfate plus glucosamine hydrochloride does not improve joint damage in an experimental model of knee osteoarthritis in rabbits[J]. European Journal of Pharmacology,2017,794:8−14. doi: 10.1016/j.ejphar.2016.11.015
|
| [4] |
MÜLLER A, LETELIER M E, GALLEGUILLOS M A, et al. Comparison of the antioxidant effects of synovial fluid from equine metacarpophalangeal joints with those of hyaluronic acid and chondroitin sulfate[J]. American Journal of Veterinary Research,2010,71(4):399−404. doi: 10.2460/ajvr.71.4.399
|
| [5] |
SAKAI S, AKIYAMA H, SATO Y, et al. Chondroitin sulfate intake inhibits the IgE-mediated allergic response by down-regulating Th2 responses in mice[J]. Journal of Biological Chemistry,2006,281(29):19872−19880. doi: 10.1074/jbc.M509058200
|
| [6] |
HAN L K, SUMIYOSHI M, TAKEDA T, et al. Inhibitory effects of chondroitin sulfate prepared from salmon nasal cartilage on fat storage in mice fed a high-fat diet[J]. International Journal of Obesity,2000,24(9):1131−1138. doi: 10.1038/sj.ijo.0801378
|
| [7] |
WU N, ZHANGY, YE X, et al. Sulfation pattern of fucose branches affects the anti-hyperlipidemic activities of fucosylated chondroitin sulfate[J]. Carbohydrate Polymers,2016,147:1−7. doi: 10.1016/j.carbpol.2016.03.013
|
| [8] |
KARUMBAIAH L, ENAM S F, BROWN A C, et al. Chondroitin sulfate glycosaminoglycan hydrogels create endogenous niches for neural stem cells[J]. Bioconjugate Chemistry,2015,26(12):2336−2349. doi: 10.1021/acs.bioconjchem.5b00397
|
| [9] |
ZHANG Q, LI J, LIU C, et al. Protective effects of low molecular weight chondroitin sulfate on amyloid beta (A β)-induced damage in vitro and in vivo[J]. Neuroscience,2015,305:169−182. doi: 10.1016/j.neuroscience.2015.08.002
|
| [10] |
肖玉良, 李平利, 程艳娜, 等. 硫酸软骨素的药理作用及应用研究进展[J]. 中国药学杂志,2014,49(13):1093−1098. [XIAO Y L, LI P L, CHENG Y N, et al. Progress in the study of pharmacology and application of chondroitin sulfate[J]. Chinese Pharmaceutical Journal,2014,49(13):1093−1098.
|
| [11] |
XIAO Y, LI P, CHENG Y, et al. Enhancing the intestinal absorption of low molecular weight chondroitin sulfate by conjugation with α-linolenic acid and the transport mechanism of the conjugates[J]. International Journal of Pharmaceutics,2014,465(1-2):143−158. doi: 10.1016/j.ijpharm.2014.02.009
|
| [12] |
HENROTIN Y, MATHY M, SANCHEZ C, et al. Chondroitin sulfate in the treatment of osteoarthritis: From in vitro studies to clinical recommendations[J]. Therapeutic Advances in Musculoskeletal Disease,2010,2(6):335−348. doi: 10.1177/1759720X10383076
|
| [13] |
SOMMERS C D, YE H, KOLINSKI R E, et al. Characterization of currently marketed heparin products: Analysis of molecular weight and heparinase-I digest patterns[J]. Analytical & Bioanalytical Chemistry,2011,401(8):2445−2454.
|
| [14] |
GUO X, YE X, SUN Y, et al. Ultrasound effects on the degradation kinetics, structure, and antioxidant activity of sea cucumber fucoidan[J]. Journal of Agricultural and Food Chemistry,2014,62(5):1088−1095. doi: 10.1021/jf404717y
|
| [15] |
MOU J, WANG C, LI Q, et al. Preparation and antioxidant properties of low molecular holothurian glycosaminoglycans by H 2O 2/ascorbic acid degradation[J]. International Journal of Biological Macromolecules,2018,107:1339−1347. doi: 10.1016/j.ijbiomac.2017.10.161
|
| [16] |
KIM J K, SRINIVASAN P, KIM J H, et al. Structural and antioxidant properties of gamma irradiated hyaluronic acid[J]. Food Chemistry,2008,109(4):763−770. doi: 10.1016/j.foodchem.2008.01.038
|
| [17] |
KAPADNIS G, DEY A, DANDEKAR P, et al. Effect of degree of deacetylation on solubility of low molecular weight chitosan produced via enzymatic breakdown of chitosan[J]. Polymer International,2019,68(6):1054−1063. doi: 10.1002/pi.5795
|
| [18] |
RIGOUIN C, LADRAT C D, SINQUIN C, et al. Assessment of biochemical methods to detect enzymatic depolymerization of polysaccharides[J]. Carbohydrate Polymers,2009,76(2):279−284. doi: 10.1016/j.carbpol.2008.10.022
|
| [19] |
KANG Z, ZHOU Z, WANG Y, et al. Bio-based strategies for producing glycosaminoglycans and their oligosaccharides[J]. Trends in Biotechnology,2018,36(8):806−818. doi: 10.1016/j.tibtech.2018.03.010
|
| [20] |
YAMAGATA T, SAITO H, HABUCHI O, et al. Purification and properties of bacterial chondroitinases and chondrosulfatases[J]. Journal of Biological Chemistry,1968,243(7):1523−1535. doi: 10.1016/S0021-9258(18)93574-X
|
| [21] |
李晔, 陈振娅, 袁其朋. 硫酸软骨素裂解酶ABC的研究进展[J]. 生物工程学报,2015,31(5):621−633. [LI Y, CHEN Z Y, YUAN Q P, et al. Research progress in chondroitinase ABC[J]. Chinese Journal of Biotechnology,2015,31(5):621−633. doi: 10.13345/j.cjb.140459
|
| [22] |
CHEN X R, LIAO S J, YE L X, et al. Neuroprotective effect of chondroitinase ABC on primary and sondary brain injury after stroke in hypertensive rats[J]. Brain Research,2014,1543:324−333. doi: 10.1016/j.brainres.2013.12.002
|
| [23] |
MAKRIS E A, MACBARB R F, PASCHOS N K, et al. Combined use of chondroitinase-ABC, TGF- β1, and collagen crosslinking agent lysyl oxidase to engineer functional neotissues for fibrocartilage repair[J]. Biomaterials,2014,35(25):6787−6796. doi: 10.1016/j.biomaterials.2014.04.083
|
| [24] |
YAMADA K S. Potential therapeutic application of chondroitin sulfate/dermatan sulfate[J]. Current Drug Discovery Technologies,2008,5(4):289−301. doi: 10.2174/157016308786733564
|
| [25] |
LAUDER R M. Chondroitin sulphate: A complex molecule with potential impacts on a wide range of biological systems[J]. Complementary Therapies in Medicine,2009,17(1):56−62. doi: 10.1016/j.ctim.2008.08.004
|
| [26] |
刘万顺, 付静芸, 常菁, 等. 硫酸软骨素酶高产菌株的筛选、鉴定和发酵培养条件的研究[J]. 中国海洋大学学报(自然科学版),2012,42(11):39−57. [LIU W S, FU J Y, CHANG J, et al. Screening and identification of two high chondroitinase producing bacterial strains and determination of their fermenting condition[J]. Periodical of Ocean University of China,2012,42(11):39−57.
|
| [27] |
陶科, 王忠彦, 国锦琳, 等. 硫酸软骨素裂解酶ABC产生菌的筛选及发酵工艺研究[J]. 中国抗生素杂志,2004,29(3):138−141. [TAO K, WANG Z Y, GUO J L, et al. Study on the screening of chondroitinase ABC producing strain and their fermentation technology[J]. Chinese Journal of Antibiotics,2004,29(3):138−141. doi: 10.3969/j.issn.1001-8689.2004.03.003
|
| [28] |
FAHNERT B, LILIE H, NEUBAUER P. Inclusion bodies: Formation and utilisation[J]. Advances in Biochemical Engineering/Biotechnology,2004,89:93−142.
|
| [29] |
VALLEJO F L, RINAS U. Strategies for the recovery of active proteins through refolding of bacterial inclusion body proteins[J]. Microbial Cell Factories,2004,3(1):11. doi: 10.1186/1475-2859-3-11
|
| [30] |
CHEN Z, LI Y, YUAN Q, et al. Expression, purification and thermostability of MBP-chondroitinase ABC I from Proteus vulgaris[J]. International Journal of Biological Macromolecules Structure Function & Interactions,2015,72:6−10.
|
| [31] |
CHEN Z, LI Y, YUAN Q. Study the effect of His-tag on chondroitinase ABC I based on characterization of enzyme[J]. International Journal of Biological Macromolecules,2015,78:96−101. doi: 10.1016/j.ijbiomac.2015.03.068
|
| [32] |
PRABHAKAR V, CAPILA I, SOUNDARARAJAN V, et al. Recombinant expression, purification, and biochemical characterization of chondroitinase ABC II from Proteus vulgaris[J]. Journal of Biological Chemistry,2009,284(2):974−982. doi: 10.1074/jbc.M806630200
|
| [33] |
|
| [34] |
MIKE, CARSON, DAVID, et al. His-tag impact on structure[J]. Acta Crystallographica stion D Biological Crystallography,2007,63(3):295−301. doi: 10.1107/S0907444906052024
|
| [35] |
SPRIESTERSBACH A, KUBICEK J, SCHÄFER F, et al. Purification of his-tagged proteins[J]. Methods in Enzymology,2015,559:1−15.
|
| [36] |
吴敬君, 李晔, 张翀, 等. 肝素黄杆菌硫酸软骨素酶AC的高效重组表达体系构建及其酶学性质研究[J]. 食品科学,2013,34(9):127−134. [WU J J, LI Y, ZHANG C, et al. Gene cloning and recombinant expression of chondroitinase AC from Pedobacter heparinus and characterization of recombinant fusion enzyme[J]. Food Science,2013,34(9):127−134. doi: 10.7506/spkx1002-6630-201309027
|
| [37] |
国家药典委员会. 中华人民共和国药典(二部)[S]. 北京: 中国医药科技出版社, 2015: 1340−1341
Chinese Pharmacopoeia Commission. Pharmacopoeia of the People's Republic of China (Volume II)[S]. Beijing: China Medical Science Press, 2015: 1340−1341.
|
| [38] |
CHEN Y, FU Z, YAN G, et al. Optimization of expression conditions and determination the proteolytic activity of codon-optimized SARS-CoV-2 main protease in Escherichia coli[J]. Sheng Wu Gong Cheng Xue Bao = Chinese Journal of Biotechnology,2021,37(4):1334−1345.
|
| [39] |
KAUR J, KUMAR A, KAUR J. Strategies for optimization of heterologous protein expression in E. coli: Roadblocks and reinforcements[J]. International Journal of Biological Macromolecules,2018,106:803−822. doi: 10.1016/j.ijbiomac.2017.08.080
|
| [40] |
杨云彭, 马晓焉, 霍毅欣. 密码子优化策略在异源蛋白表达中的应用[J]. 生物工程学报,2019,35(12):2227−2237. [YANG Y P, MA X Y, HUO Y X, et al. Application of codon optimization strategy in heterologous protein expression[J]. Chinese Journal of Biotechnology,2019,35(12):2227−2237. doi: 10.13345/j.cjb.190280
|
| [41] |
GOUY M, GAUTIER C. Codon usage in bacteria: Correlation with gene expressivity[J]. Nucleic Acids Research,1982,10(22):7055−7074. doi: 10.1093/nar/10.22.7055
|
| [42] |
ROSANO G L, MORALES E S, CECCARELLI E A. New tools for recombinant protein production in Escherichia coli: A 5-year update: Recombinant protein production in Escherichia coli[J]. Protein Science,2019,28:1412−1422. doi: 10.1002/pro.3668
|
| [43] |
GARCIA-FRAGA B, DA SILVA A F, LOPEZ-SEIJAS J, et al. Optimized expression conditions for enhancing production of two recombinant chitinolytic enzymes from different prokaryote domains[J]. Bioprocess & Biosystems Engineering,2015,38(12):2477−2486.
|
| [44] |
HAMAI A, HASHIMOTO N, MOCHIZUKI H, et al. Two distinct chondroitin sulfate ABC lyases. An endoeliminase yielding tetrasaccharides and an exoeliminase preferentially acting on oligosaccharides[J]. Journal of Biological Chemistry,1997,272(14):9123−9130. doi: 10.1074/jbc.272.14.9123
|





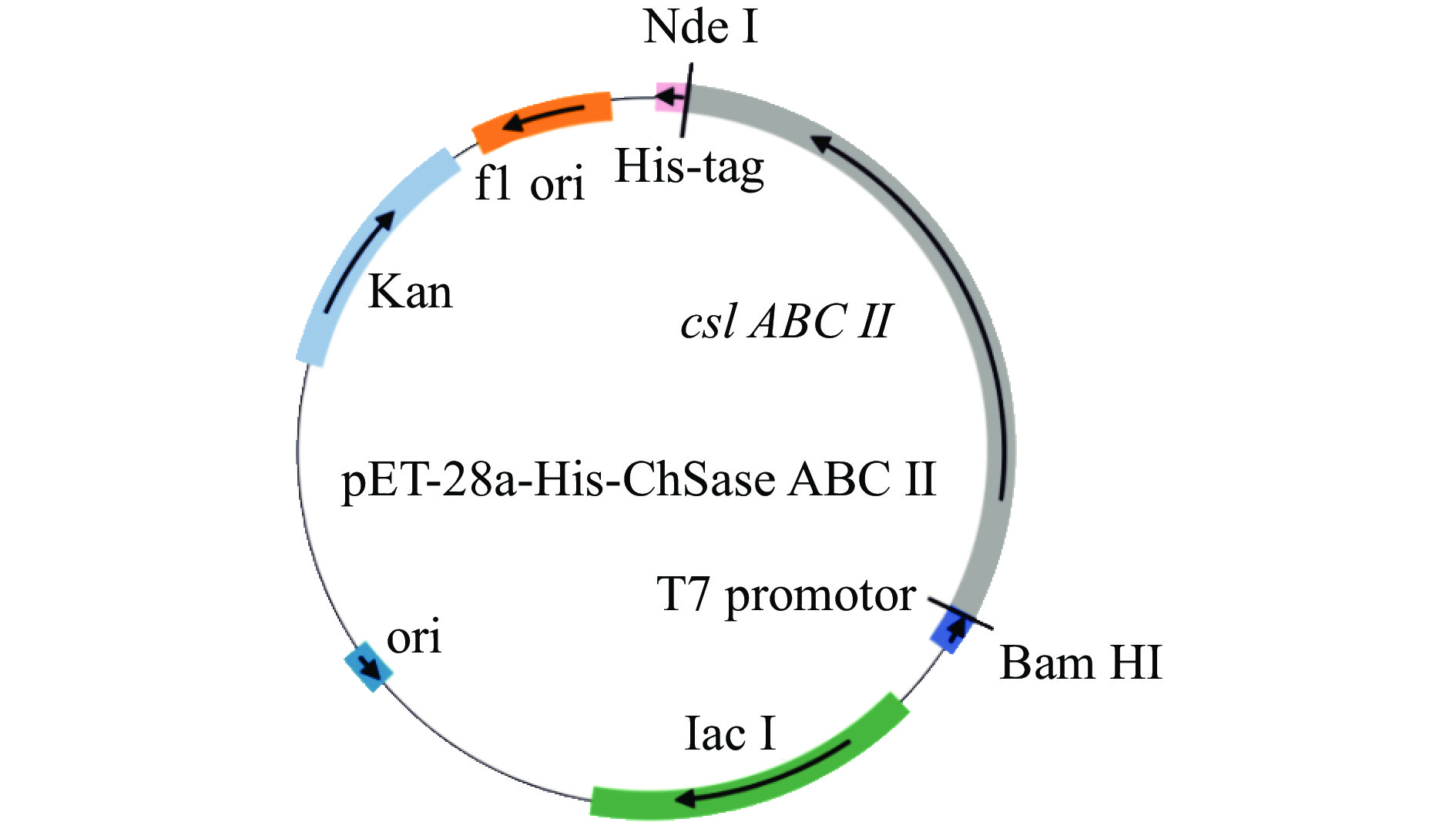




 DownLoad:
DownLoad: