Physiological Function of Fermented Modified Dietary Fiber and Its Application in Food
-
摘要: 可溶性膳食纤维(Soluble dietary fiber,SDF)分子量小、结构无序,有良好的理化特性。天然膳食纤维中可溶性膳食纤维含量少,生物利用率低,因此要对膳食纤维进行改性。传统改性方法如物理、化学法存在成本高、能耗高、污染环境的缺点。微生物产生的酶能断裂大分子糖苷键,将不溶性大分子转化为可溶性膳食纤维,因此发酵法是一种低成本、低能耗、绿色无污染的新型改性方法。本文系统阐述了发酵改性原理、影响改性效果的因素如接种量、温度、pH等,并进一步介绍了发酵改性膳食纤维的生理功能及其在面制品、肉制品、乳制品中的应用,为未来膳食纤维进一步开发与利用提供理论依据。Abstract: Soluble dietary fiber has small molecular weight, disordered structure and good physicochemical properties. However, natural dietary fiber possesses low bioavailability due to the low content of soluble dietary fiber, so it is essential to modify it. Traditional modification methods such as physical and chemical methods have the disadvantages of high cost, high energy consumption and environmental pollution. Enzymes produced by microorganisms are able to break the glycosidic bond of macromolecules and convert insoluble macromolecules into soluble dietary fiber. Therefore, fermentation is a low-cost, low energy consumption, green and pollution-free new modification method. This article systematically explains the principle of fermentation modification and the factors that affect the modification effect, such as inoculation amount, temperature, pH and so on, then it introduces the physiological function of dietary fiber by fermentation modification and its application in food products such as flour products, dairy products, meat products, providing theoretical basis for further development and utilization of dietary fiber in the future.
-
Keywords:
- dietary fiber /
- modification /
- fermentation /
- physiological function /
- food applications
-
膳食纤维(Dietary fiber,DF)作为七大营养素之一,是一类有利于人体健康的碳水化合物,存在于谷物、果蔬、豆类以及海藻等植物中[1],它不能被人体胃肠道消化吸收,也不为人体供能,但可以在大肠中被发酵,促进肠道功能[2]。膳食纤维根据溶解性可被分为可溶性膳食纤维(Soluble dietary fiber,SDF)与不溶性膳食纤维(Insoluble dietary fiber,IDF)。膳食纤维的生理活性与分子结构密切相关,与IDF相比,SDF拥有较为无序的结构和大量亲水基团,拥有更优良的理化特性与生理活性[3],如水合性质、吸附特性、抗氧化性与离子交换特性等,在保护人体健康方面有更显著的效果。并且,将SDF添加到食品中可以提升食物的营养价值与感官品质,还能有效降低癌症、心血管疾病、肠道疾病以及肥胖症的发病几率[4],满足人们对健康食品的需求[5]。然而,天然膳食纤维中SDF含量较少,直接添加到食品中可能使食物口感不佳,无法最大程度发挥膳食纤维的生理活性。研究表明,SDF含量在10%上才能被称为优质膳食纤维[6]。因此,增加天然膳食纤维中SDF的含量,提高SDF的生理活性,将其应用于食品加工业变得尤为重要[7]。
物理法、化学法、酶法是较传统的改性方法,物理改性存在安全隐患大、能耗高的缺点;化学改性过程中可能会发生较多副反应,污染环境;酶法改性操作繁琐,价格高昂。微生物发酵法作为膳食纤维改性的新型手段,具有成本低廉、安全环保、产物活性高等优点[8],微生物产生的酶作用于大分子,使其降解为小分子SDF,能有效提高膳食纤维中SDF的比例,并改善膳食纤维的理化性质与功能特性[9]。将发酵改性后的膳食纤维添加到乳制品、肉类、焙烤食品中,可以提高食品的营养价值,改善食品的口感[10]。近年来,许多研究利用不同菌种如绿色木霉、黑曲霉、毛霉、混合菌种等对膳食纤维进行改性,发现微生物能够改变膳食纤维的结构、提高可溶性膳食纤维的含量、增强膳食纤维的生理活性。并且发酵后的膳食纤维可以添加到不同食品中制作功能性食品,提升食品的营养价值与口感。本文详述了膳食纤维发酵改性的原理、发酵改性与其他改性法相比的优势、影响改性效果的各个因素、发酵后膳食纤维生理功能的变化、改性膳食纤维添加到食品中的作用,旨在提高膳食纤维的生物利用率并扩大在其食品行业中的应用,迎合当今人们对绿色食品的需求。
1. 膳食纤维的简介
1.1 膳食纤维的定义
膳食纤维的定义随着人们的研究不断更新完善,早在1953年,膳食纤维这个术语就被Hipsley等[11]首次提出,他将植物细胞壁中不可消化的组分称为膳食纤维,如木质素、纤维素和半纤维素,它们不能被人体消化酶分解。随后,在1972~1976年间,Trowell等[12]建立了“膳食纤维假说”,即膳食纤维与人体健康的关系,并把膳食纤维定义为不能被人体消化吸收的木质素、非淀粉类多糖等物质的总称。为了将膳食纤维的定义统一,美国科学学会、美国谷物化学家协会、中国营养学会等机构开展了长达半个世纪的讨论。2014年,世界卫生组织和粮农组织将膳食纤维定义为含有10个以上单体链节碳水化合物的聚合物,它们不被人体小肠中消化酶水解[13]。虽然现在各个组织定义膳食纤维的方法仍存在差异,但是膳食纤维的定义必须包括以下几点[14]:a.来源是食物原料或其副产物,如柑橘、香菇、米糠、脐橙皮等[15];b.不被人体消化酶所消化分解,不为人体提供能量[16];c.单体数目大于等于3的碳水化合物,对人体健康有益[17]。随着现代科学与社会的发展,膳食纤维逐渐受到重视,人们发现膳食纤维是日常生活中不可或缺的营养物质,因此膳食纤维也被称作“第七营养素”[18]。
1.2 膳食纤维的分类
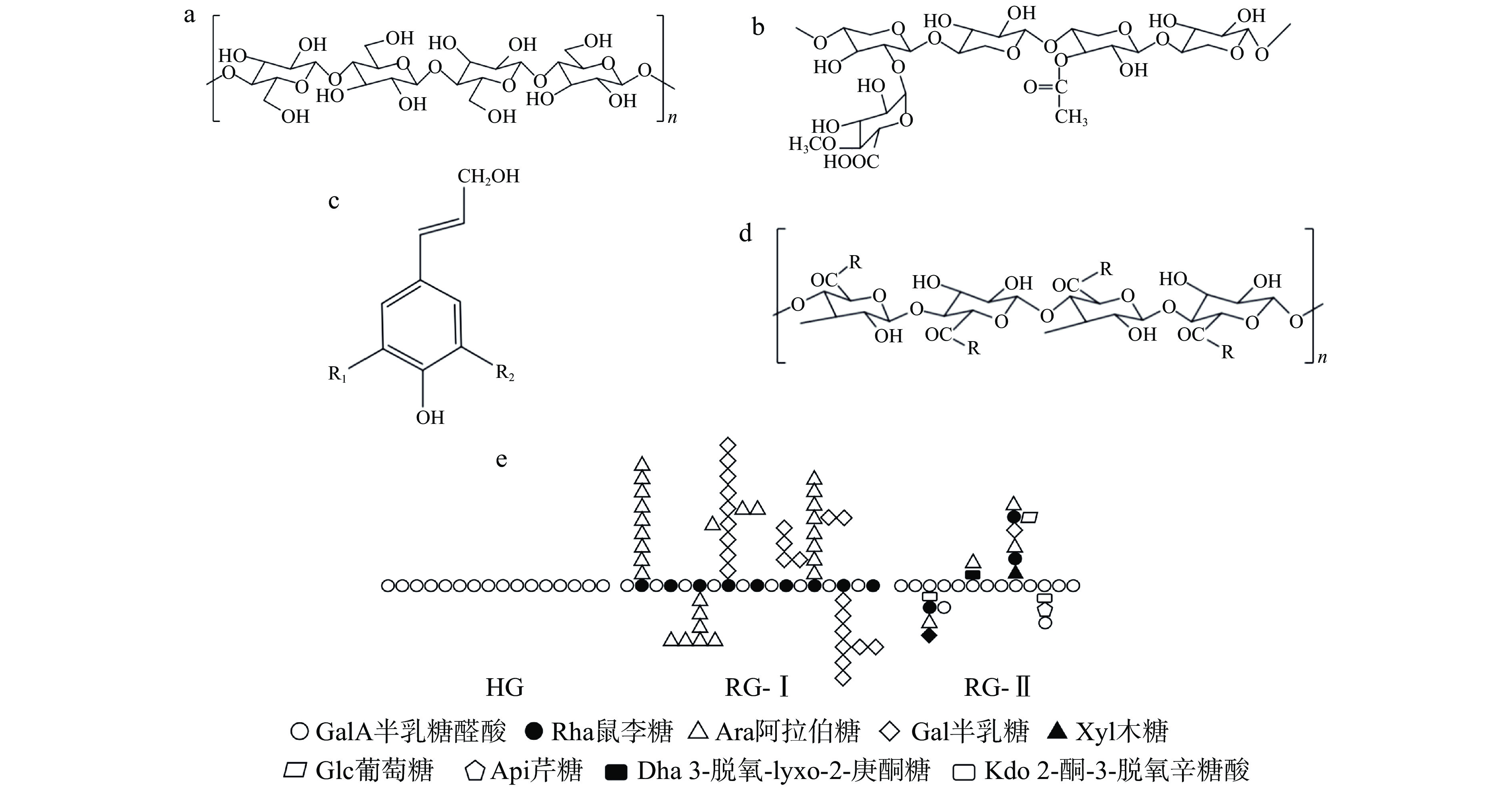
膳食纤维可以根据其来源、理化性质、水溶性以及可发酵性等方面进行分类[19](表1),根据溶解性,膳食纤维可被分为IDF与SDF[20],IDF分子量较大,约为50~250 kDa,是由五碳糖或者六碳糖以β-1,4糖苷键连接而成的天然高聚物,参与植物细胞壁的构成,存在于谷类、薯类、坚果、豆制品中,主要成分有纤维素(A)、半纤维素(B)、木质素等(C)[21](图1)。它们的结构有高度规整性,并且聚合度较高、分子取向性良好、化学稳定性较强,不易溶于水和有机溶剂,可以预防便秘、肥胖等疾病[22]。SDF分子量较小,约为20~400 kDa,广泛存在于果蔬、食用菌中[23],以果胶为例(D),它是以D-半乳糖醛酸由α-1,4-糖苷键连接组成的酸性杂多糖,结构由直链多糖聚半乳糖醛酸区(Homogalacturonan region,HG区)、鼠李糖半乳糖醛酸Ⅰ区(Rhamnogalacturonan Ⅰ region,RGI区)与鼠李糖半乳糖醛酸Ⅱ区(Rhamnogalacturonan Ⅱ region,RGII区)构成[24],分为“毛发区”和“光滑区”(E),较小的分子量和复杂的分子结构赋予其优良的理化特性与生理活性[25],能够影响人体代谢功能,如调节血糖血脂、保护肠道,对维持人体健康起到更大作用[26]。
2. 发酵法改性膳食纤维
当原料中SDF与IDF的比例在1~2.3之间时,才能被称为高品质膳食纤维[27],否则只能作为填充剂使用。然而,在天然植物细胞壁中,SDF的含量远低于IDF,达不到人体平衡膳食的要求[28]。因此,要用简单高效的方法提高SDF的比例[29],使其得以充分开发利用,对人类健康具有重要价值。目前膳食纤维改性方法有物理法、化学法、酶法、微生物法等。
2.1 发酵改性的原理
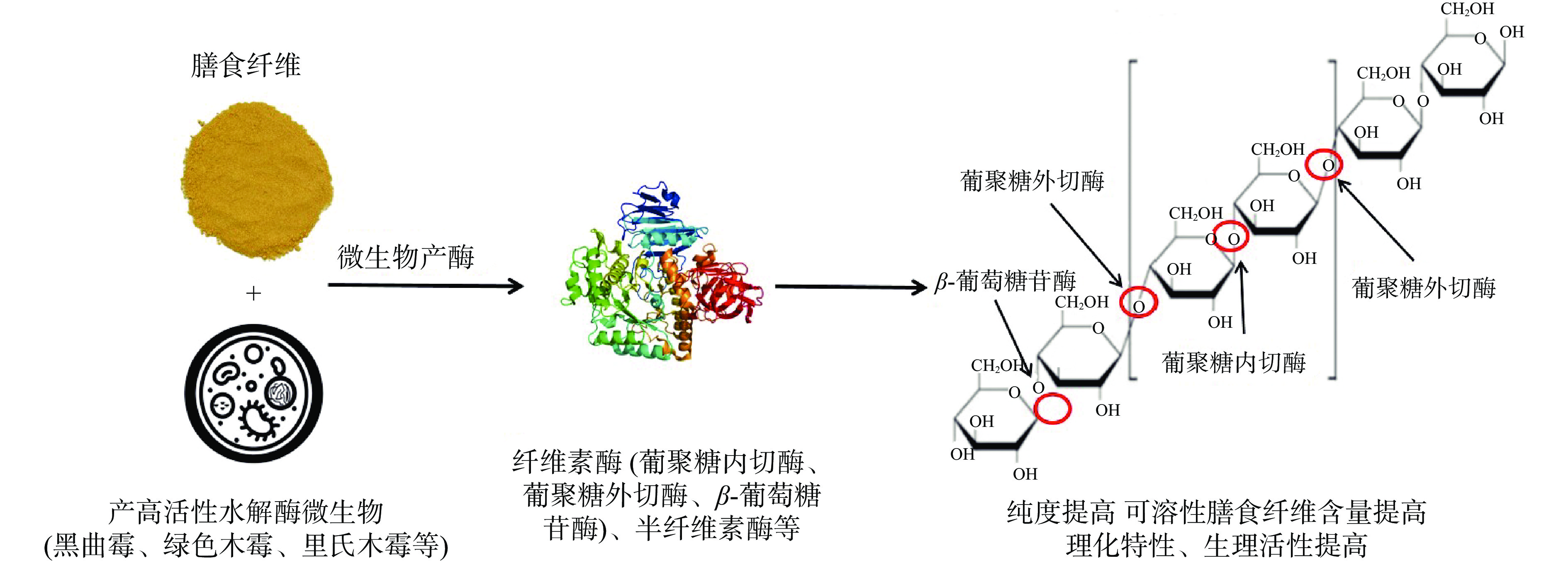
微生物改性法利用菌株发酵过程中分泌的胞外水解酶提高原料中化合物产量以及生理活性[30](图2)。IDF大分子中的纤维素、木质素由大量葡萄糖通过β-1,4糖苷键链接,产酶菌株接种后能将蛋白质、淀粉作为营养物质,并产生大量纤维素酶、半纤维素酶、木聚糖酶等[31]。纤维素酶是一种复合酶系,分为葡聚糖外切酶、葡聚糖内切酶和β-葡萄糖苷酶,首先,葡聚糖外切酶破坏纤维素大分子的晶体结构,暴露出大量化学键,随后葡聚糖内切酶作用于β-1,4-糖苷键,将其转化为纤维二糖、纤维三糖等低聚糖,最后β-葡萄糖苷酶将低聚糖转化为小分子葡萄糖[32],提高可溶性糖类的含量,并且化学键的断裂释放出了更多活性物质,达到改性的目的。蔡泓莹等[33]用黑曲霉、米曲霉发酵半叶马尾藻,发现发酵后的SDF得率增加、结构变得更松散、分子量更小且持水力、持油力、胆固醇吸附能力、胆酸钠吸附能力等功能特性优于未发酵SDF。由于发酵改性的后的膳食纤维能够去除蛋白质、淀粉等杂质,提高产物纯度,并且分子量减小,结构呈碎片状,暴露出更多活性基团,使理化特性与生理活性有一定的提高,色泽、质地、气味和分散程度也比较完善,有望用于食品工业制作功能性食品。
2.2 发酵改性的优势
不同改性方法的比较如表2所示,物理法利用仪器对膳食纤维进行改性,如挤压膨化、高压处理等,工艺简便[34],但设备运行成本较贵,限制其工业化生产[35]。化学法主要使用酸、碱、过氧化氢等化学试剂处理膳食纤维[36],与其他方法相比,能耗低且较高效[37],但是化学反应容易产生副产物,对环境造成污染[38]。用纤维素酶、半纤维素酶处理膳食纤维也可以达到改性的效果,条件温和且专一性强[39],但是酶制剂成本高,较难实现大批量生产[40]。与这几种方法相比,微生物发酵能够实现产酶与酶解过程统一,利用微生物代谢产物将膳食纤维降解为小分子从而达到改性的目的,拥有安全高效、成本低、产物活性高的优点[41]。Chang等[42]比较了蒸汽爆破、米曲霉发酵改性膳食纤维的效果,与爆破组相比,发酵组的纤维素、半纤维素含量更低,为7.35%和38.59%,并且发酵组膳食纤维表面无木质素沉积,表面颗粒更小且粗糙,微生物产生的酶能够更好进入大分子内部,改性效果更加彻底。Ren等[43]用酶法、发酵法改性米糠膳食纤维,发现经过发酵的膳食纤维变成小碎片状,中位粒径减小到79.7 μm,比表面积增加到138.8 m2/kg,吸附葡萄糖能力提高到原来的57.2%。较大的比表面积能够暴露出更多的酶结合位点与活性基团,提高生理活性效果更加显著。
表 2 不同改性方法的比较Table 2. Methods commonly used in the modification of dietary fiber改性名称 采用方法 原理 优势 不足 参考文献 物理法 超高压、超微粉碎、
超声波、挤压等机械处理改变DF的颗粒尺寸、
微观结构简单、安全、作用均匀 操作风险高、改性不彻底 [34−35] 化学法 酸碱试剂、羧甲基化、
羟丙基化等化学试剂破坏糖苷键、改变或
引入官能团效率高、能耗低 副反应多、污染环境 [36−38] 酶法 纤维素酶、木聚糖酶 酶制剂使大分子的分子链断裂 专一性强、易控制、
设备要求小成本过高、效率低 [39−40] 发酵法 绿色木霉、黑曲霉、
乳酸菌、毛霉等微生物代谢产物使大分子降解 环保、条件温和、产物活性高 菌种选育较难、
易受到环境影响[41−43] 联合法 微生物-物理法、微生物-酶法 多种改性法结合,弥补单个方法的不足 效果显著、时间短 工艺复杂,难优化 [44−48] 并且,发酵法能与其他改性方法相结合,如与超高压法、蒸汽爆破法、超声法、酶法联合处理,可以更好发挥各方法优势[44]。Li等[45]用高净水压力法与发酵法联合处理腌菜膳食纤维,膳食纤维先经过高净水压力处理,底物体积减小,缩短了酶反应时间并增强酶活性。改性后SDF产量增加,吸附重金属、胆酸钠和抑制淀粉酶活性能力增强,提高了改性速率。Juodeikiene等[46]用超声波预处理大豆后用乳酸菌发酵,超声波产生的空化气泡崩塌后产生冲击波与剪切力,破坏细胞壁,促进SDF的溶解,大豆中的营养物质蛋白质也大量释放,为发酵提供营养物质。经过预处理后,乳酸菌长势更好,使可溶性物质的含量提升至2.963 g/g。Si等[47]研究了发酵、发酵-酶结合改性海草膳食纤维的效果,研究发现联合法处理显著提高SDF的得率,由3.9%增加到20.76%,且将持水力提高到8.73 g/g、吸附胆固醇特性提高到38.03 mg/g。这些研究表明,联合改性法作为一种多方法集成的改性过程,能够提高改性效率、使改性效果更加彻底、减少能量的消耗与资源的浪费,比单一改性法更有优势,但操作比较复杂,优化工艺有较大难度[48]。
2.3 发酵改性膳食纤维的影响因素
发酵改性法比传统改性法有更大优势,但在发酵过程中,SDF的活性与得率与发酵pH、温度、接种量等因素有密切关系[49],探究其最佳发酵条件,有利于改性的进行。
2.3.1 菌种
菌种的选择在改性中起到重要作用,不同菌株产生的酶系、酶活力不同,选用合适的菌种进行发酵,不仅生产效率高,还能有效改善膳食纤维的理化特性,目前改性使用较多的是高产纤维素酶真菌与工业产酸菌[50],表3展示了利用菌种发酵后膳食纤维的变化。例如,木霉属是较好的纤维素酶生产菌,曲霉属、青霉属和镰刀菌属的产酶能力也较强[51]。黑曲霉(Aspergillus niger)是曲霉属中的常见菌种,能够产生纤维素酶、淀粉酶、糖化酶和果胶酶等多种酶。黑曲霉具有培养条件简单、产酶速度快、酶系全面且安全等优点,可用于食品发酵[52]。绿色木霉(Trichoderma viride)能够分泌多种高活性的细胞壁水解酶,如纤维素酶、木聚糖酶等,可以有效降解纤维素、半纤维素大分子。并且绿色木霉生长速度较快、产酶量多、生长条件温和易满足,适合用于发酵工业[53]。因此,黑曲霉与绿色木霉这两种菌种都适合用于膳食纤维改性。但单菌株发酵存在一些不足,例如木霉属能够分泌大量纤维素酶,其中β-葡萄糖苷酶活性较低,使发酵液中的纤维二糖、可溶性寡糖大量积累,抑制其他水解酶的催化活性,从而影响发酵效果[54]。Xue等[55]用黑曲霉与里氏木霉混合发酵脐橙皮膳食纤维,黑曲霉是一种产高活性β-葡萄糖苷酶的菌种,能与里氏木霉互补形成多组分复杂纤维素酶体系,首先葡聚糖内切酶、外切酶共同作用于纤维素,其次葡萄糖苷酶通过将过量单糖转化为龙胆二糖、三糖从而解除单糖对其他酶的抑制,使木聚糖酶、半纤维素酶、纤维素酶的活力都有所提高。活性物质与大分子通过醚键、酯键相结合,高活性的复合酶系更彻底的破坏了糖苷键,释放出活性物质,达到提高生理活性的目的。Si等[56]从茶叶渣中筛选出两株能够高效降解纤维素的菌种草野旋腔菌与普氏曲霉,并用这两种菌进行混合发酵。实验表明,混菌发酵的羧甲基纤维素酶、滤纸酶、β-葡萄糖苷酶的活性均高于单菌发酵酶活性。与单菌发酵改性相比,混菌发酵改性茶渣膳食纤维的产率更高,由8.92%提高到26.47%。并且混菌发酵的膳食纤维拥有更高的持水力、持油力、亚硝酸盐吸附特性提升至9.23 g/g、10.64 g/g、69.66 μg/g,克服了单菌发酵酶系单一、酶活性不稳定的问题[57]。因此在改性前,要先筛选出高产纤维素酶、蛋白酶、淀粉酶、脂肪酶等水解酶的菌株,它们可以在利用底物的同时产生大量活性物质。随后,探究多种混合菌株最佳产酶条件并用于膳食纤维改性,可以生产出纯度高、产量更大、功能特性更优良的膳食纤维。
表 3 发酵后膳食纤维的变化Table 3. Changes in dietary fiber after fermentation菌种 原料 改性条件 理化特性的变化 参考文献 黑曲霉 米糠膳食纤维 pH5.0,温度26 ℃,
发酵时间77 hSDF结构更疏松,提取率增加29.58%,膨胀力、持水力、
持油力提高84.44%、79.30%、73.25%[61] 绿色木霉 茶渣膳食纤维 接种量10.34%,pH5.46,
发酵时间43.59 hSDF得率从4.3%增加至31.56%,糖醛酸含量由17.81%增加到34.82%;
金属离子结合能力提高2.4倍[63] 纳豆芽孢杆菌 小米糠膳食纤维 接种量3%,温度37 ℃,
发酵时间47 hSDF含量由2.3%增加到13.2%,葡萄糖吸附能力提高到原来的1.39倍;
胆固醇吸附能力提高到105.6 μmol/g[71] 乳酸菌+粗糙链孢霉 豆渣膳食纤维 接种量5.5%,温度30 ℃,
发酵时间2 dSDF含量增加;α-淀粉酶活性抑制能力由9.55%提高到15.04%,
葡萄糖延滞指数提升一倍[10] 草野旋腔菌+普氏曲霉 茶渣膳食纤维 接种比=1:2.46,初始pH4.58,
发酵时间112.07 h,SDF含量由8.92%提高至26.47%;分子量减小至373.35 kDa;
吸附亚硝酸盐离子能力提高至69.66 μg/g[47] 绿色木霉+黑曲霉 豆渣膳食纤维 接种量10%,接种比=1:2初始pH6,
温度26 ℃,发酵时间45 hSDF含量增加到原来的4.46倍,质地均匀,口感细腻,气味淡香 [64] 2.3.2 接种量
微生物数量对菌体生长、产酶量均有影响,过高或过低的接种量都会抑制菌种生长,导致改性效果不佳,若接种量过小,微生物数量少且存在较长时间的适应期,代谢时间变短,导致产酶量不足,进而影响SDF的得率。虽然高接种量可以使菌株繁殖较快,但是随着接种量的增加,菌株竞争生长,生长密度过高,导致氧气与营养物质供应不足,并且发酵过程中微生物产生大量酸,影响发酵环境的pH与酶活,导致产率下降[58]。Miao等[59]采用发酵工艺对黑木耳多糖进行改性,随着绿色木霉接种量的增加,黑木耳多糖的含量先增大后减小,当接种量为12.4%时,黑木耳多糖的产率最高,由5.2%提高到26.9%,此时多糖拥有较好的抗氧化活性、阳离子交换能力,提高产量的同时增强了生理活性,因此确定12.4%为绿色木霉的最佳接种量。Jia等[60]通过响应面实验确定了绿色木霉发酵改性脱脂米糠的最佳接种量为10%,此时脱脂米糠中SDF的产量由10.5%显著提高到33.4%,改性米糠SDF的溶解性、持水力、持油力由0.84 g/g、1.21 g/g、1.57 g/g增加到0.94 g/g、6.01 g/g、2.36 g/g,有望添加到食物中制作功能性食品,适合高血脂人群食用。合适的接种量促菌体生长并减少杂菌的污染,生成产物速率加快,能够加快改性进程。一般而言,酵母菌的种子液接种量约为5%~10%,细菌接种量约为1%~5%,霉菌接种量约为7%~15%。
2.3.3 pH
pH通过影响微生物细胞膜的生理功能、胞内酶的活性、营养物质的解离程度与利用率来调控微生物的生长繁殖速率。不同微生物的适应能力有差异,生长pH也不同[61]。Xie等[62]确定了绿色木霉发酵改性脱脂米糠IDF的条件,当发酵初始pH为4时,菌株产生的木聚糖酶、羧甲基纤维素酶、β-葡萄糖苷酶活性较高,能够有效破坏IDF大分子化学键并释放出酚类物质,释放出的酚类物质具有良好的α-淀粉酶、α-葡萄糖苷酶抑制活性。Chen等[63]利用绿色木霉发酵茶渣,发现5.46为绿色木霉的发酵的最佳pH,在最佳发酵条件下茶渣SDF的产率由4.3%提高到31.56%,SDF改性后有较好的重金属结合能力,可以作为食品添加剂,防止人体受到食源性重金属的危害。大多数发酵pH都要控制在4.5~8之间,通常情况下,细菌的最适生长pH为6.5~7.5,酵母菌、霉菌则适合于pH为5~6的酸性环境。在发酵前要调节培养基到合适pH,同时可以在培养基中加入缓冲物质,以保证pH在微生物生长繁殖过程中的相对稳定。
2.3.4 时间
接种初期,微生物生长速度较慢,产率增加比较缓慢,随着培养时间增加,微生物处于对数生长期,繁殖速度加快,产生的酶系活性高,经过一段时间的稳定期后发酵产物大量积累,达到最大值。若发酵时间过长,培养基中营养物质被耗尽,部分酶开始失活,导致SDF产率变少[64]。Xu等[65]发现,在发酵第九天时,桦褐孔菌菌丝生长量到达峰值,此时菌株产酶量也较高,用于发酵麦秸,可以使纤维素、半纤维素和木质素降解率分别达到46.1%、46.4%和44.1%,此时多糖产量提高142.9%。Chen等[66]研究了红曲霉发酵马铃薯膳食纤维的工艺,当添加50 g/L马铃薯渣作为原料时,SDF的产量在第6 d由3.4 g/L增加到6.3 g/L,在第7 d时,SDF产量又下降到5.7 g/L,可能是由于随着发酵时间增加,部分SDF水解为更小分子的糖,导致产量减少。细菌如乳酸菌、嗜热链球菌生长繁殖速度较快,只需1~2 d便可改性膳食纤维,酵母菌如酿酒酵母、解脂耶氏酵母改性时间约为4~5 d,霉菌如黑曲霉、绿色木霉、里氏木霉改性时间约为2~5 d。
2.3.5 温度
温度在发酵过程中也是十分重要的影响因素,它通过影响细胞内生物大分子的活性影响微生物的生长繁殖[67]。不同种类的微生物有相应的最适生长温度,当温度远低于最适生长温度,微生物的生长繁殖受到抑制,产酶速度较慢;而温度过高时,培养基水分流失,导致部分微生物分生孢子提前形成,无法生成菌丝,且细胞内的生物活性物质发生变性[68],不利于改性。Rodríguez等[69]确定了酵母的最佳发酵温度为29.5 ℃,在最佳温度发酵苹果渣,可以制备出高蛋白、高纤维、不含糖、有高抗氧化性的功能食品原料。此外,微生物的最适生长温度与最适发酵温度不同,最佳发酵温度是发酵速率最高或积累代谢产物量最高时的培养温度,纤维素酶发挥最大活性的温度为45~75 ℃,远高于微生物生长温度范围,曹艳等[70]采用分段控温手段,先用青霉菌最适生长温度28 ℃培养,待菌体大量生长后调节温度至45 ℃继续培养,使纤维素酶展现出高活性,此时控温发酵比固定温度发酵手段的SDF产率高3.7%。在发酵改性时,可将细菌工作温度控制在20~45 ℃,酵母菌工作温度控制在15~30 ℃ ,霉菌温度控制在20~30 ℃,并且可根据发酵进程调节温度,使得率最大化。
3. 发酵法改性膳食纤维的生理功能
膳食纤维的生理功能受到分子结构、活性基团羟基、氨基、羧基的影响,发酵改性后,膳食纤维的结构、组成发生变化,暴露出更多活性基团,提高膳食纤维的吸附力、持水力、持油力、离子交换特性等理化特性,从而赋予膳食纤维优良的生理功能,比如有效预防糖尿病、心血管疾病、调节肠道稳态等。
3.1 预防糖尿病
膳食纤维预防糖尿病的机制有以下几点:a.增加分子间的相互作用并形成物理障碍,抑制葡萄糖在肠道中的扩散[72];亲水基团如羟基、羧基和氨基吸收大量水分,形成凝胶和三维网络,增加食物的黏度,减缓了葡萄糖的扩散与小肠上皮细胞吸收葡萄糖的速率[73]。b.膳食纤维的多孔网状结构可以结合多种化合物及离子,阻碍人体肠道对它们的吸收利用[74],降低肠内葡萄糖浓度。c.膳食纤维有抑制α-葡萄糖苷酶与α-淀粉酶的活性的功效[75],能有效控制小肠上皮细胞对葡萄糖的吸收。d.SDF可以被大肠内的菌群发酵,生成对人体健康有益的短链脂肪酸,如果胶能被微生物完全发酵生成乙酸、丙酸和丁酸,其中丙酸可以降低胰岛素抵抗,促进肝糖原的合成[76]。发酵后,膳食纤维的预防糖尿病能力显著提升,由于大分子结构被微生物所产的酶、酸破坏,比表面积增大、孔洞变多并形成更多SDF,暴露出更多氢键与偶极子,与水、葡萄糖等物质的接触面积变大,水合性质、吸附特性提升。Chu等[71]研究发现发酵后的膳食纤维葡萄糖吸附能力高达3.38 mmol/g,是未发酵膳食纤维的1.39倍,有效延缓了葡萄糖在胃肠道的消化吸收,对降低血糖起着重要作用。Cui等[77]用发酵改性的杏浆SDF给大鼠灌胃,灌胃28 d后,大鼠血糖水平明显低于空白对照组,改性后的SDF的大量网状结构可以将α-葡萄糖苷酶、α-淀粉酶包裹,抑制酶的活性,从而延缓葡萄糖的释放,可以达到预防糖尿病的效果。
3.2 预防心血管疾病
人体内胆固醇、甘油三酯含量过高常诱发心血管疾病[78],膳食纤维经发酵后有更优良的吸附胆固醇、油脂、胆汁酸能力,能够更加有效地预防心血管疾病[79]。Wang等[80]以刺梨果渣为原料,利用纳豆芽孢杆菌进行发酵,在最佳发酵条件下,刺梨果渣中SDF含量由7.68%提高到10.68%,膳食纤维的持油力由1.25 g/g提升至1.40 g/g。因为SDF在肠道中形成黏度较高的凝胶和纤维胶束,这些复合物能够在肠道中吸收大量胆固醇、胆汁酸,影响它们的乳化、扩散过程[81],胆固醇的分解产物为胆汁酸,胆汁酸依靠体内的重吸收作用维持循环,若大量胆汁酸被吸附,无法返回肝脏,肝脏会大量降解胆固醇。随后,血液中胆固醇进入肝脏,降低血液胆固醇含量[82]。并且,SDF有良好的发酵性,SDF含量增多后,在体内发酵产生更多短链脂肪酸,降低肠道内pH,促进肝脏内的脂质分解,下调人体对甘油三脂代谢产物和胆固醇的溶解性[83]。此外,经过发酵的膳食纤维具有表面积较大的多孔结构,Li等[84]发现用乳酸菌发酵的小米膳食纤维结构更加松散,能有效吸附油脂和胰脂肪酶[85],并破坏胰脂肪酶的氢键,影响酶的二级结构,抑制脂肪酶的活性[86],防止油脂分解。在小肠和胃液环境中,改性膳食纤维对胆固醇的吸收能力分别提升至10.21 mg/g和6.93 mg/g。经过发酵后,膳食纤维能吸附更多油脂、胆固醇、胆汁酸,并抑制脂质分解相关酶的活性,减少人体对油脂的吸收,起到预防心血管疾病的作用。
3.3 预防肠道疾病
在结肠内,大多数SDF能作为微生物的底物发酵,产生大量短链脂肪酸[87],酸性条件促进了与炎症性肠病相关的酸化拟杆菌和肠杆菌的生长[88]。微生物改性后,膳食纤维的发酵能力提高,大量SDF被肠道菌群如双歧杆菌发酵,生成乙酸、丁酸和丙酸,增加肠道丰度,改善肠道的屏障功能[89],阻碍肠腔内中内毒素、病原菌等有害物质进入血液,抵抗结肠疾病;并且膳食纤维改性后持水性提高,能够吸收大量水分,增加粪便的含水量与体积,润滑肠道并促进排便[90],加速肠道运动,利于有害物质的排泄,保护了肠粘膜,起到预防便秘、结肠癌等疾病的作用[91]。Zhang等[92]的研究表明,植物乳杆菌发酵改变了芦笋多糖的单糖组成,并提高膳食纤维的免疫调节活性,减少结肠癌、胃癌的发病几率。Sun等[93]利用寡孢根霉发酵提取中药“玉屏风”多糖后加入獭兔饲料中,发现发酵改性多糖可以提高獭兔血清IL-1、IL-2、IL-4等浓度、抗炎因子IL-4和IL-10的mRNA表达量,增强免疫功能并调节肠道能力。改性后的膳食纤维能够润滑肠道,加速有害物质的排出;并在肠道中更好发酵,产生大量短链脂肪酸且能够提升有益菌如厚壁菌门、拟杆菌门等的丰度,改善人体屏障功能,增强免疫力,预防肠道疾病。
3.4 预防肥胖
膳食纤维产能较少,是一种低热量的食物,它可以减少人体对油脂的吸收利用,并增加饱腹感从而达到减肥的目的[94],首先,膳食纤维发酵改性后,产生大量多孔结构和亲水基团,赋予了膳食纤维优良的膨胀性与持水性,膳食纤维吸收水分后体积增大,形成凝胶,使食物在胃中的排空速度降低,使人产生饱腹感,减少食物摄入[95];SDF发酵后产生的短链脂肪酸可以参与肠道激素、多肽物质分泌调节,对降低食欲、减少脂肪生成量有一定效果[96],同时还能改善胰岛素敏感性,通过抑制脂肪细胞胰岛素信号转导,降低脂肪组织中脂肪的蓄积[97],促进全身脂质代谢,减少小肠对脂肪的吸收率。Rodríguez等[98]用苹果酒酵母发酵苹果渣,发酵增加了苹果渣膳食纤维的含量,大分子细胞壁被破坏后,释放出大量酚类物质,可以作为原料生产针对糖尿病、肥胖患者的功能性食品。赵久毅等[99]用未处理、热风干燥处理、真空干燥处理、发酵处理的豆渣膳食纤维给小鼠灌胃,发现发酵后的IDF:SDF变为2.5:1,并且膳食纤维的溶解性、膨胀力和持油力均显著提高。与其他组相比,摄入发酵膳食纤维组的小鼠14 d后体重最轻,并且体重增长量最少。低热量、饱腹感高的改性膳食纤维能够降低食欲和降低胰岛素敏感性,适合减肥人群食用。
4. 发酵膳食纤维在食品中的应用
改性膳食纤维有预防心血管疾病、肠道疾病、癌症的功效,可以作为营养强化剂或食品添加剂加入到面制品、肉制品、乳制品中[100](表4)。食物的口感受到膳食纤维的持水力、持油力、溶解性等理化特性的影响,改性后的膳食纤维可均匀分布到食物中,赋予食品良好的感官特性、营养价值,还能在食物中增加保健功能,改善人们饮食结构,有利于人体健康。Sun等[101]以红曲霉为发酵菌种处理豆渣膳食纤维,改性之后SDF的含量由4.3%增加到7.7%,持水力、持油力、膨胀力分别由4.17 g/g、3.45 g/g、1.96 mL/g提高到4.35 g/g、3.54 g/g、2.71 mL/g,有效提高了豆渣膳食纤维的感官品质与营养特性,使膳食纤维更容易被人体吸收消化,并将红曲霉发酵过程中产生的有益功能物质色素保留下来,可以应用于功能性食品的开发利用。
表 4 发酵改性膳食纤维在食品中的应用Table 4. Application of fermented dietary fiber in food4.1 面制品
发酵改性膳食纤维添加到面制品如饼干、面包、馒头中可以提升其感官品质与加工特性。面制品口感变硬、难以消化的原因是由于淀粉老化。经过发酵后,膳食纤维的水合性质更良好,可以与淀粉分子竞争水分,在淀粉分子之间形成黏性较大的水合层,阻碍直链淀粉的聚集;膳食纤维中的羟基、羧基能与直链淀粉结合,使水分更容易进入淀粉分子内部,淀粉吸水量增多,防止老化[102]。并且,SDF可以通过非共价作用力形成类似面筋的三维凝胶网络,提高面团吸水性,改善面团筋力,有助于提高面制品的弹性和柔软度[103],在储藏过程中不易变干、变硬。此外,在面制品中添加发酵改性膳食纤维时,大量SDF吸附淀粉颗粒,增加消化液的粘度,使淀粉降解酶的可及性受到限制,延迟淀粉的消化和食物中糖的释放,防止消化过程中释放过多葡萄糖导致的血糖升高[104],还能刺激肠道蠕动,达到排毒保健的效果。Coda等[105]用霍氏假丝酵母和短乳杆菌发酵麸皮,经过改性后的麸皮有较多的可溶性阿拉伯木聚糖,加入面包中可增大面包体积,使面包柔软度、稳定性、口感良好,并且延长面包的货架期。孙聪聪[106]在麦麸中接种红曲霉,发酵后麦麸中SDF含量提高到原来的1.54倍,口感也更良好。与未发酵的膳食纤维相比,发酵后膳食纤维的色泽更加明亮、有较好的风味、制作的面制品外观更加美观,更受大众的欢迎。
4.2 肉制品
肉类营养丰富,但含有大量胆固醇,摄入过多可能会增加患结肠癌的风险[107]。具有高持水性、低热量的改性膳食纤维可作为填充物和粘合剂加入肉制品中,用于改善肉制品的质构、流变特性[108]。改性膳食纤维中SDF含量较高,溶于水后促进亲水基团与水结合,形成凝胶,蛋白质相互交联形成较为稳定的三维网络,使各组分紧密结合,并包含大量水分,显著增加肉制品的流变特性和凝胶强度[109],模拟肉中脂肪口感,使其嫩滑多汁。优良的保油、保水能力也是提高肉质品质量至关重要的因素,它们赋予肉制品良好的蒸煮率、多汁性、乳化稳定性和收缩性。张俊等[110]将发酵后的麸皮用大豆油进行预乳化处理,再添加到火腿肠中,发现火腿肠的硬度和咀嚼性减小,对质构特性有一定改善。IDF在肉制品中起到机械填充的作用,若含量过高会导致食物口感粗糙、硬度较大、难以咀嚼,发酵后可溶性单糖含量增加,SDF优良的水溶性使其更好分布在网络孔隙中,形成紧密稳定的凝胶结构,起到支撑体系和保持水分的作用,改善肉制品的口感和组织状态。
4.3 乳制品
添加适量改性膳食纤维,赋予乳制品良好的口感与风味,有一定的保健作用。在制作与贮藏过程中,酸奶乳清蛋白易在表面堆积,出现乳清分离和脱水的现象,影响酸奶的口感与外观,若加入改性膳食纤维,酪蛋白胶束与SDF能通过疏水相互作用、静电相互作用结合,加速蛋白质网络的形成,防止蛋白质聚集,提高酸奶的凝胶强度[111],凝胶捕获大量游离脂肪酸,降低脂肪含量的同时稳定乳液结构,延长了酸奶货架期。此外,SDF可作为酸奶发酵菌的良好碳源,赋予酸奶独特的口感并促进益生菌的生长,提高酸奶的营养价值,可拥有美容、保护肠胃以及预防便秘、提高免疫力等作用[112]。高天宇[113]用食用型真菌对大豆膳食纤维进行改性,发酵后豆渣中IDF含量减少,IDF和SDF比例更接近与均衡膳食纤维要求,豆渣的豆腥味减弱,加入到酸奶中使其口感细腻,有较高的感官接受度。Wu等[114]在制作酸奶时,先添加米糠再添加乳酸菌,发酵改变了米糠膳食纤维的单糖组成,β-葡聚糖、果胶等SDF含量增加,提高了酸奶的营养价值。由于发酵后膳食纤维结构改变,表面变得粗糙且多孔,较小的孔隙尺寸可以限制蛋白质重新排列和游离水的移动,使酸奶脱水率显著降低。此外,营养物质多酚、有机酸等与大分子细胞壁通过酯键、醚键等连接,酶使这些化学键断裂,细胞壁中的结合酚酸转化为游离态酚酸,有更高的生物利用度,饮用后有预防癌症、抗氧化的功效。
综上所述,向各种食物中添加膳食纤维可以改善食物的不良风味与口感,并且满足人体每日所需。此外,添加了改性膳食纤维的食物营养价值更高,有一定的保健功能,如美容、降血压、提高免疫力、防止便秘等。
5. 结论与展望
膳食纤维生理活性的优劣与其结构密切相关,可溶性膳食纤维的结构松散、较为无序,理化特性、生理活性优于不可溶性膳食纤维。因此,可溶膳食纤维的含量越高,膳食纤维品质越好。发酵改性法利用微生物产酶的原理来改变可溶性膳食纤维与不可溶性膳食纤维的比例,有效提高其理化特性与生理活性。与其他改性方法相比,发酵改性有绿色环保、安全的优势,并且发酵改性膳食纤维的理化性质如持水性持油性、膨胀性、抗氧化性等特性更优良,可作为膳食补充剂加入到各种食品中,使食品拥有较好的感官品质与营养价值。然而,有关发酵改性的研究比较有限,大多数研究主要集中于发酵前后膳食纤维理化性质的变化,对于改性膳食纤维与食物中营养成分的相互作用、发酵前后膳食纤维生理活性变化的机理尚不明确,制约了发酵改性的工业化生产与应用。并且发酵过程的工艺较繁琐且易染杂菌,改性效果取决于发酵菌株与操作手段。因此要筛选高产纤维素酶菌株,开发出更先进、简洁的发酵工艺,并更加深入研究发酵与其他方法进行联合改性,以期生产出更高活性的膳食纤维,应用于食品行业,满足人群对膳食纤维的需要,提升膳食纤维在食品工业中的应用价值。
-
表 1 膳食纤维的分类(按溶解性)
Table 1 Classification of dietary fiber by solubility (According to solubility)
表 2 不同改性方法的比较
Table 2 Methods commonly used in the modification of dietary fiber
改性名称 采用方法 原理 优势 不足 参考文献 物理法 超高压、超微粉碎、
超声波、挤压等机械处理改变DF的颗粒尺寸、
微观结构简单、安全、作用均匀 操作风险高、改性不彻底 [34−35] 化学法 酸碱试剂、羧甲基化、
羟丙基化等化学试剂破坏糖苷键、改变或
引入官能团效率高、能耗低 副反应多、污染环境 [36−38] 酶法 纤维素酶、木聚糖酶 酶制剂使大分子的分子链断裂 专一性强、易控制、
设备要求小成本过高、效率低 [39−40] 发酵法 绿色木霉、黑曲霉、
乳酸菌、毛霉等微生物代谢产物使大分子降解 环保、条件温和、产物活性高 菌种选育较难、
易受到环境影响[41−43] 联合法 微生物-物理法、微生物-酶法 多种改性法结合,弥补单个方法的不足 效果显著、时间短 工艺复杂,难优化 [44−48] 表 3 发酵后膳食纤维的变化
Table 3 Changes in dietary fiber after fermentation
菌种 原料 改性条件 理化特性的变化 参考文献 黑曲霉 米糠膳食纤维 pH5.0,温度26 ℃,
发酵时间77 hSDF结构更疏松,提取率增加29.58%,膨胀力、持水力、
持油力提高84.44%、79.30%、73.25%[61] 绿色木霉 茶渣膳食纤维 接种量10.34%,pH5.46,
发酵时间43.59 hSDF得率从4.3%增加至31.56%,糖醛酸含量由17.81%增加到34.82%;
金属离子结合能力提高2.4倍[63] 纳豆芽孢杆菌 小米糠膳食纤维 接种量3%,温度37 ℃,
发酵时间47 hSDF含量由2.3%增加到13.2%,葡萄糖吸附能力提高到原来的1.39倍;
胆固醇吸附能力提高到105.6 μmol/g[71] 乳酸菌+粗糙链孢霉 豆渣膳食纤维 接种量5.5%,温度30 ℃,
发酵时间2 dSDF含量增加;α-淀粉酶活性抑制能力由9.55%提高到15.04%,
葡萄糖延滞指数提升一倍[10] 草野旋腔菌+普氏曲霉 茶渣膳食纤维 接种比=1:2.46,初始pH4.58,
发酵时间112.07 h,SDF含量由8.92%提高至26.47%;分子量减小至373.35 kDa;
吸附亚硝酸盐离子能力提高至69.66 μg/g[47] 绿色木霉+黑曲霉 豆渣膳食纤维 接种量10%,接种比=1:2初始pH6,
温度26 ℃,发酵时间45 hSDF含量增加到原来的4.46倍,质地均匀,口感细腻,气味淡香 [64] 表 4 发酵改性膳食纤维在食品中的应用
Table 4 Application of fermented dietary fiber in food
-
[1] HUANG W Q, TAN H Z, NIE S P. Beneficial effects of seaweed-derived dietary fiber:Highlights of the sulfated polysaccharides[J]. Food Chemistry,2022,373:131608. doi: 10.1016/j.foodchem.2021.131608
[2] LIU H F, ZENG X Y, HUANG J Y, et al. Dietary fiber extracted from pomelo fruitlets promotes intestinal functions, both in vitro and in vivo[J]. Carbohydrate Polymers,2021,252(1):117186.
[3] DONG W J, WANG D D, HU R, et al. Chemical composition, structural and functional properties of soluble dietary fiber obtained from coffee peel using different extraction methods[J]. Food Research International,2020,136:109497. doi: 10.1016/j.foodres.2020.109497
[4] QIN W Y, SUN L J, MIAO M, et al. Plant-sourced intrinsic dietary fiber:Physical structure and health function[J]. Trends in Food Science & Technology,2021,118:341−355.
[5] PATHANIA S, KAUR N. Utilization of fruits and vegetable by-products for isolation of dietary fibres and its potential application as functional ingredients[J]. Bioactive Carbohydrates and Dietary Fibre,2022,27:100295. doi: 10.1016/j.bcdf.2021.100295
[6] YAN L, LI T, LIU C H, et al. Effects of high hydrostatic pressure and superfine grinding treatment on physicochemical/functional properties of pear pomace and chemical composition of its soluble dietary fibre[J]. LWT,2019,107:171−177. doi: 10.1016/j.lwt.2019.03.019
[7] XUE Z H, MA Q Q, GUO Q W, et al. Physicochemical and functional properties of extruded dietary fiber from mushroom Lentinula edodes residues[J]. Food Bioscience,2019,32:100452. doi: 10.1016/j.fbio.2019.100452
[8] TU Z C, CHEN L L, WANG H, et al. Effect of fermentation and dynamic high pressure microfluidization on dietary fibre of soybean residue[J]. Journal of Food Science and Technology,2014,51(11):3285−3292. doi: 10.1007/s13197-012-0838-1
[9] YU C Y, FANG Y, HUANG W W, et al. Effect of surfactants on the production and biofunction of Tremella fuciformis polysaccharide through submerged fermentation[J]. LWT,2022,163:113602. doi: 10.1016/j.lwt.2022.113602
[10] LIN D R, LONG X M, HUANG Y C, et al. Effects of microbial fermentation and microwave treatment on the composition, structural characteristics, and functional properties of modified okara dietary fiber[J]. LWT,2020,123:109059. doi: 10.1016/j.lwt.2020.109059
[11] HIPSLEY E H. Dietary “Fibre” and Pregnancy Toxaemia[J]. BMJ,1953,2(4833):420−422. doi: 10.1136/bmj.2.4833.420
[12] TROWELL H. Ischemic heart disease and dietary fiber[J]. The American Journal of Clinical Nutrition,1972,25(9):926−932. doi: 10.1093/ajcn/25.9.926
[13] JONES J M. CODEX-aligned dietary fiber definitions help to bridge the ‘fiber gap’[J]. Nutrition Journal,2014,13(1):34. doi: 10.1186/1475-2891-13-34
[14] MACAGNAN F T, DA SILVA L P, HECKTHEUER L H. Dietary fibre:The scientific search for an ideal definition and methodology of analysis, and its physiological importance as a carrier of bioactive compounds[J]. Food Research International,2016,85:144−154. doi: 10.1016/j.foodres.2016.04.032
[15] DENG Z Z, WU N, WANG J, et al. Dietary fibers extracted from Saccharina japonica can improve metabolic syndrome and ameliorate gut microbiota dysbiosis induced by high fat diet[J]. Journal of Functional Foods,2021,85(4):104642.
[16] GUO Y T, LIU W, WU B G, et al. Modification of garlic skin dietary fiber with twin-screw extrusion process and in vivo evaluation of Pb binding[J]. Food Chemistry,2018,268:550−557. doi: 10.1016/j.foodchem.2018.06.047
[17] DE MENEZES E W, GIUNTINI E B, DAN M C T, et al. Codex dietary fibre definition- Justification for inclusion of carbohydrates from 3 to 9 degrees of polymerisation[J]. Food Chemistry,2013,140(3):581−585. doi: 10.1016/j.foodchem.2013.02.075
[18] WANG S Q, FANG Y Q, XU Y B, et al. The effects of different extraction methods on physicochemical, functional and physiological properties of soluble and insoluble dietary fiber from Rubus chingii Hu. fruits[J]. Journal of Functional Foods,2022,93:105081. doi: 10.1016/j.jff.2022.105081
[19] LIU X, SUO K K, WANG P, et al. Modification of wheat bran insoluble and soluble dietary fibers with snail enzyme[J]. Food Science and Human Wellness,2021,10(3):356−361. doi: 10.1016/j.fshw.2021.02.027
[20] ZHU F. Dietary fiber polysaccharides of amaranth, buckwheat and quinoa grains:A review of chemical structure, biological functions and food uses[J]. Carbohydrate Polymers,2020,248:116819. doi: 10.1016/j.carbpol.2020.116819
[21] LI S Y, LI J S, ZHU Z Z, et al. Soluble dietary fiber and polyphenol complex in lotus root:Preparation, interaction and identification[J]. Food Chemistry,2020,314:126219. doi: 10.1016/j.foodchem.2020.126219
[22] DAOU C, ZHANG H. Functional and physiological properties of total, soluble, and insoluble dietary fibres derived from defatted rice bran[J]. Journal of Food Science and Technology,2014,51(12):3878−3885. doi: 10.1007/s13197-013-0925-y
[23] WIDANINGRUM, FLANAGAN B M, WILLIAMS B A, et al. Fruit and vegetable insoluble dietary fibre in vitro fermentation characteristics depend on cell wall type[J]. Bioactive Carbohydrates and Dietary Fibre,2020,23:100223. doi: 10.1016/j.bcdf.2020.100223
[24] HUANG J Y, HU Z H, HU L P, et al. Pectin-based active packaging:A critical review on preparation, physical properties and novel application in food preservation[J]. Trends in Food Science & Technology,2021,118:167−178.
[25] ZHONG L Z, FANG Z X, WAHLQVIST M L, et al. Multi-response surface optimisation of extrusion cooking to increase soluble dietary fibre and polyphenols in lupin seed coat[J]. LWT,2021,140(1):110767.
[26] XIONG M, ZHENG S Y, BAI T M, et al. The difference among structure, physicochemical and functional properties of dietary fiber extracted from triticale and hull-less barley[J]. LWT,2022,154:112771. doi: 10.1016/j.lwt.2021.112771
[27] MRABET A, RODRÍGUEZ-GUTIÉRREZ G, RUBIO-SENENT F, et al. Enzymatic conversion of date fruit fiber concentrates into a new product enriched in antioxidant soluble fiber[J]. LWT,2017,75:727−734. doi: 10.1016/j.lwt.2016.10.017
[28] CASSIDY Y M, MCSORLEY E M, ALLSOPP P J. Effect of soluble dietary fibre on postprandial blood glucose response and its potential as a functional food ingredient[J]. Journal of Functional Foods,2018,46:423−439. doi: 10.1016/j.jff.2018.05.019
[29] LI B, YANG W, NIE Y Y, et al. Effect of steam explosion on dietary fiber, polysaccharide, protein and physicochemical properties of okara[J]. Food Hydrocolloids,2019,94:48−56. doi: 10.1016/j.foodhyd.2019.02.042
[30] CODA R, KATINA K, RIZZELLO C G. Bran bioprocessing for enhanced functional properties[J]. Current Opinion in Food Science,2015,1:50−55. doi: 10.1016/j.cofs.2014.11.007
[31] ABD RAZAK D L, ABD RASHID N Y, JAMALUDDIN A, et al. Enhancement of phenolic acid content and antioxidant activity of rice bran fermented with Rhizopus oligosporus and Monascus purpureus[J]. Biocatalysis and Agricultural Biotechnology,2015,4(1):33−38. doi: 10.1016/j.bcab.2014.11.003
[32] SINGH R, SAATI A A, FAIDAH H, et al. Prospects of microbial cellulase production using banana peels wastes for antimicrobial applications[J]. International Journal of Food Microbiology,2023,388:110069. doi: 10.1016/j.ijfoodmicro.2022.110069
[33] 蔡泓滢, 李瑞, 汪卓, 等. 真菌发酵对马尾藻可溶性膳食纤维结构、功能特性和抗氧化活性的影响[J/OL]. 食品科学:1−13 [2023-12-06]. http://kns.cnki.net/kcms/detail/11.2206.TS.20230427.1132.002.html. [CAI H Y, L R, W Z, et al. Effects of fungi fermentation on the structure, functional properties, and antioxidant activity of soluble dietary fiber of Sargassum[J/OL]. 食品科学:1−13 [2023-12-06]. http://kns.cnki.net/kcms/detail/11.2206.TS.20230427.1132.002.html.] CAI H Y, L R, W Z, et al. Effects of fungi fermentation on the structure, functional properties, and antioxidant activity of soluble dietary fiber of Sargassum[J/OL]. 食品科学: 1−13 [2023-12-06]. http://kns.cnki.net/kcms/detail/11.2206.TS.20230427.1132.002.html.
[34] SUI W J, XIE X, LIU R, et al. Effect of wheat bran modification by steam explosion on structural characteristics and rheological properties of wheat flour dough[J]. Food Hydrocolloids,2018,84:571−580. doi: 10.1016/j.foodhyd.2018.06.027
[35] DHIMAN A, PRABHAKAR P K. Micronization in food processing:A comprehensive review of mechanistic approach, physicochemical, functional properties and self-stability of micronized food materials[J]. Journal of Food Engineering,2021,292:110248. doi: 10.1016/j.jfoodeng.2020.110248
[36] SHAIKH M, HAIDER S, ALI T M, et al. Physical, thermal, mechanical and barrier properties of pearl millet starch films as affected by levels of acetylation and hydroxypropylation[J]. International Journal of Biological Macromolecules,2019,124:209−219. doi: 10.1016/j.ijbiomac.2018.11.135
[37] ZHANG M Y, LIAO A M, THAKUR K, et al. Modification of wheat bran insoluble dietary fiber with carboxymethylation, complex enzymatic hydrolysis and ultrafine comminution[J]. Food Chemistry,2019,297:124983. doi: 10.1016/j.foodchem.2019.124983
[38] MA M M, MU T H. Modification of deoiled cumin dietary fiber with laccase and cellulase under high hydrostatic pressure[J]. Carbohydrate Polymers,2016,136:87−94. doi: 10.1016/j.carbpol.2015.09.030
[39] LIU L, WEN W, ZHANG R F, et al. Complex enzyme hydrolysis releases antioxidative phenolics from rice bran[J]. Food Chemistry,2017,214:1−8. doi: 10.1016/j.foodchem.2016.07.038
[40] XIE F Y, ZHAO T, WAN H C, et al. Structural and physicochemical characteristics of rice bran dietary fiber by cellulase and high-pressure homogenization[J]. Applied Sciences,2019,9(7):1270. doi: 10.3390/app9071270
[41] MA W J, LIANG Y T, LIN H S, et al. Fermentation of grapefruit peel by an efficient cellulose-degrading strain ( Penicillium YZ-1):Modification, structure and functional properties of soluble dietary fiber[J]. Food Chemistry,2023,420:136123. doi: 10.1016/j.foodchem.2023.136123
[42] CHANG J, CHENG W, YIN Q Q, et al. Effect of steam explosion and microbial fermentation on cellulose and lignin degradation of corn stover[J]. Bioresource Technology,2012,104:587−592. doi: 10.1016/j.biortech.2011.10.070
[43] REN F Y, FENG Y L, ZHANG H J, et al. Effects of modification methods on microstructural and physicochemical characteristics of defatted rice bran dietary fiber[J]. LWT,2021,151:112161. doi: 10.1016/j.lwt.2021.112161
[44] WANG L, XU H G, YUAN F, et al. Preparation and physicochemical properties of soluble dietary fiber from orange peel assisted by steam explosion and dilute acid soaking[J]. Food Chemistry,2015,185:90−98. doi: 10.1016/j.foodchem.2015.03.112
[45] LI W H, JIN Q, WU Q Y, et al. Effect of a hybrid process, high hydrostatic pressure treatment combined with mixed-strain fermentation, on the quality of the dietary fibre in pickled vegetables[J]. International Journal of Food Science & Technology,2020,55(6):2650−2659.
[46] JUODEIKIENE G, TRAKSELYTE-RUPSIENE K, NAVICKAITE B, et al. Functionalization of soya press cake (okara) by ultrasonication for enhancement of submerged fermentation with Lactobacillus paracasei LUHS244 for wheat bread production[J]. LWT,2021,152:112337. doi: 10.1016/j.lwt.2021.112337
[47] SI J Y, YANG C R, CHEN Y, et al. Structural properties and adsorption capacities of Mesona chinensis Benth residues dietary fiber prepared by cellulase treatment assisted by Aspergillus niger or Trichoderma reesei[J]. Food Chemistry,2023,407:135149. doi: 10.1016/j.foodchem.2022.135149
[48] GAN J P, XIE L, PENG G Y, et al. Systematic review on modification methods of dietary fiber[J]. Food Hydrocolloids,2021,119:106872. doi: 10.1016/j.foodhyd.2021.106872
[49] GRZELAK-BŁASZCZYK K, CZARNECKI A, KLEWICKI R, et al. Lactic acid fermentation of osmo-dehydrated onion[J]. Food Chemistry,2023,399:133954. doi: 10.1016/j.foodchem.2022.133954
[50] LE B, ANH P T N, KIM J-E, et al. Rice bran fermentation by lactic acid bacteria to enhance antioxidant activities and increase the ferulic acid, ρ-coumaric acid, and γ-oryzanol content[J]. Journal of Applied Biological Chemistry,2019,62(3):257−264. doi: 10.3839/jabc.2019.035
[51] MEINI M R, CABEZUDO I, GALETTO C S, et al. Production of grape pomace extracts with enhanced antioxidant and prebiotic activities through solid-state fermentation by Aspergillus niger and Aspergillus oryzae[J]. Food Bioscience,2021,42:101168. doi: 10.1016/j.fbio.2021.101168
[52] MA X Y, GAO M, WANG N H, et al. Lactic acid production from co-fermentation of food waste and spent mushroom substance with Aspergillus niger cellulase[J]. Bioresource Technology,2021,337:125365. doi: 10.1016/j.biortech.2021.125365
[53] LI X H, YANG H J, ROY B, et al. Enhanced cellulase production of the Trichoderma viride mutated by microwave and ultraviolet[J]. Microbiological Research,2010,165(3):190−198. doi: 10.1016/j.micres.2009.04.001
[54] ZHAO X L, ZHENG Z H, CAI Y F, et al. Accelerated biomethane production from lignocellulosic biomass:Pretreated by mixed enzymes secreted by Trichoderma viride and Aspergillus sp[J]. Bioresource Technology,2020,309:123378. doi: 10.1016/j.biortech.2020.123378
[55] XUE P Y, LIAO W, CHEN Y, et al. Release characteristic and mechanism of bound polyphenols from insoluble dietary fiber of navel orange peel via mixed solid-state fermentation with Trichoderma reesei and Aspergillus niger[J]. LWT,2022,161:113387. doi: 10.1016/j.lwt.2022.113387
[56] SI J Y, YANG C R, MA W J, et al. Screen of high efficiency cellulose degrading strains and effects on tea residues dietary fiber modification:Structural properties and adsorption capacities[J]. International Journal of Biological Macromolecules,2022,220:337−347. doi: 10.1016/j.ijbiomac.2022.08.092
[57] LU H H, HUANG C J, YU K N, et al. Effects of mixed inoculation of Leuconostoc citreum and Lactobacillus plantarum on suansun (Sour bamboo shoot) fermentation[J]. Food Bioscience,2022,47:101688. doi: 10.1016/j.fbio.2022.101688
[58] 贾梦云. 绿色木霉发酵制备脱脂米糠可溶性膳食纤维及其在饼干中的应用[D]. 南昌:南昌大学, 2020:12−13. [JIA M Y. Preparation of soluble dietary fiber from defatted rice bran by Trichoderma viride and its application in biscuits[D]. Nanchang:Nanchang University, 2020:12−13.] JIA M Y. Preparation of soluble dietary fiber from defatted rice bran by Trichoderma viride and its application in biscuits[D]. Nanchang: Nanchang University, 2020: 12−13.
[59] MIAO J N, SHI W, ZHANG J Q, et al. Response surface methodology for the fermentation of polysaccharides from Auricularia auricula using Trichoderma viride and their antioxidant activities[J]. International Journal of Biological Macromolecules,2020,155:393−402. doi: 10.1016/j.ijbiomac.2020.03.183
[60] JIA M Y, CHEN J J, LIU X Z, et al. Structural characteristics and functional properties of soluble dietary fiber from defatted rice bran obtained through Trichoderma viride fermentation[J]. Food Hydrocolloids,2019,94:468−474. doi: 10.1016/j.foodhyd.2019.03.047
[61] 闵钟熳, 高路, 高育哲, 等. 黑曲霉发酵法制备米糠粕可溶性膳食纤维工艺优化及其理化分析[J]. 食品科学,2018,39(2):112−118. [MIN Z M, GAO L, GAO Y Z, et al. Optimization of the preparation process for soluble dietary fiber from rice bran by Aspergillus niger fermentation and its physicochemical properties[J]. Food Science,2018,39(2):112−118.] MIN Z M, GAO L, GAO Y Z, et al . Optimization of the preparation process for soluble dietary fiber from rice bran by Aspergillus niger fermentation and its physicochemical properties[J]. Food Science,2018 ,39 (2 ):112 −118 .[62] XIE J Y, LIU S, DONG R H, et al. Bound polyphenols from insoluble dietary fiber of defatted rice bran by solid-state fermentation with Trichoderma viride:profile, activity, and release mechanism[J].Journal of Agricultural and Food Chemistry,2021,69(17):5026−5039. doi: 10.1021/acs.jafc.1c00752
[63] CHEN J J, HUANG H R, CHEN Y, et al. Effects of fermentation on the structural characteristics and in vitro binding capacity of soluble dietary fiber from tea residues[J]. LWT,2020,131:109818. doi: 10.1016/j.lwt.2020.109818
[64] 何晓哲. 发酵法提高豆渣可溶性膳食纤维和蛋白质含量的研究[D]. 合肥:安徽农业大学, 2013:11−21. [HE X Z. Study on improvement of soluble dietary fiber and protein by fermentation on soybean dregs[D]. Hefei:Anhui Agricultural University, 2013:11−21.] HE X Z. Study on improvement of soluble dietary fiber and protein by fermentation on soybean dregs[D]. Hefei: Anhui Agricultural University, 2013: 11−21.
[65] XU X Q, WU P, WANG T Z, et al. Synergistic effects of surfactant-assisted biodegradation of wheat straw and production of polysaccharides by Inonotus obliquus under submerged fermentation[J]. Bioresource Technology,2019,278:43−50. doi: 10.1016/j.biortech.2019.01.022
[66] CHEN X J, GUI R Y, LI N, et al. Production of soluble dietary fibers and red pigments from potato pomace in submerged fermentation by Monascus purpureus[J]. Process Biochemistry,2021,111:159−166. doi: 10.1016/j.procbio.2021.09.011
[67] DEED R C, FEDRIZZI B, GARDNER R C. Influence of fermentation temperature, yeast strain, and grape juice on the aroma chemistry and sensory profile of sauvignon blanc wines[J]. Journal of Agricultural and Food Chemistry,2017,65(40):8902−8912. doi: 10.1021/acs.jafc.7b03229
[68] VERMA N, KUMAR V. Impact of process parameters and plant polysaccharide hydrolysates in cellulase production by Trichoderma reesei and Neurospora crassa under wheat bran based solid state fermentation[J]. Biotechnology Reports,2020,25:e00416. doi: 10.1016/j.btre.2019.e00416
[69] MADRERA R R, BEDRIÑANA R P, VALLES B S. Application of central composite design in the fermentation of apple pomace to optimize its nutritional and functional properties[J]. Acta Alimentaria,2018,47(3):324−332. doi: 10.1556/066.2018.47.3.8
[70] 曹艳, 范铭, 童创, 等. 混菌发酵联合分段控温工艺提高柑橘皮渣可溶性膳食纤维含量[J]. 浙江农业学报,2019,31(3):474−479. [CAO Y, FAN M, TONG C, et al. Improving soluble dietary fiber content of citrus peel and pomace by fermentation with mixed strains combined with two-stage temperature control[J]. Journal of Zhejiang University,2019,31(3):474−479.] CAO Y, FAN M, TONG C, et al . Improving soluble dietary fiber content of citrus peel and pomace by fermentation with mixed strains combined with two-stage temperature control[J]. Journal of Zhejiang University,2019 ,31 (3 ):474 −479 .[71] CHU J X, ZHAO H Z, LU Z X, et al. Improved physicochemical and functional properties of dietary fiber from millet bran fermented by Bacillus natto[J]. Food Chemistry,2019,294:79−86. doi: 10.1016/j.foodchem.2019.05.035
[72] LIU T N, WANG K, XUE W, et al. In vitro starch digestibility, edible quality and microstructure of instant rice noodles enriched with rice bran insoluble dietary fiber[J]. LWT,2021,142:111008. doi: 10.1016/j.lwt.2021.111008
[73] MIEHLE E, HAAS M, BADER-MITTERMAIER S, et al. The role of hydration properties of soluble dietary fibers on glucose diffusion[J]. Food Hydrocolloids,2022,131:107822. doi: 10.1016/j.foodhyd.2022.107822
[74] WANG K L, LI M, HAN Q Y, et al. Inhibition of α-amylase activity by insoluble and soluble dietary fibers from kiwifruit ( Actinidia deliciosa)[J]. Food Bioscience,2021,42:101057. doi: 10.1016/j.fbio.2021.101057
[75] RODRIGUES D, FREITAS A C, SOUSA S, et al. Chemical and structural characterization of Pholiota nameko extracts with biological properties[J]. Food Chemistry,2017,216:176−185. doi: 10.1016/j.foodchem.2016.08.030
[76] LI X X, ZHANG X X, ZHANG R, et al. Gut modulation based anti-diabetic effects of carboxymethylated wheat bran dietary fiber in high-fat diet/streptozotocin-induced diabetic mice and their potential mechanisms[J]. Food and Chemical Toxicology,2021,152:112235. doi: 10.1016/j.fct.2021.112235
[77] CUI J X, GU X, ZHANG Q H, et al. Production and anti-diabetic activity of soluble dietary fiber from apricot pulp by Trichoderma viride fermentation[J]. Food & Function,2015,6(5):1635−1642.
[78] LI B H, HUI F, YUAN Z, et al. Untargeted fecal metabolomics revealed biochemical mechanisms of the blood lipid-lowering effect of koumiss treatment in patients with hyperlipidemia[J]. Journal of Functional Foods,2021,78:104355. doi: 10.1016/j.jff.2021.104355
[79] LI M X, LIU Y X, YANG G, et al. Microstructure, physicochemical properties, and adsorption capacity of deoiled red raspberry pomace and its total dietary fiber[J]. LWT,2022,153:112478. doi: 10.1016/j.lwt.2021.112478
[80] WANG Y Q, WANG J D, CAI Z H, et al. Improved physicochemical and functional properties of dietary fiber from Rosa roxburghii pomace fermented by Bacillus natto[J]. Food Bioscience,2022,50:102030. doi: 10.1016/j.fbio.2022.102030
[81] VILLANUEVA-SUÁREZ M J, MATEOS-APARICIO I, PÉREZ-CÓZAR M L, et al. Hypolipidemic effects of dietary fibre from an artichoke by-product in Syrian hamsters[J]. Journal of Functional Foods,2019,56:156−162. doi: 10.1016/j.jff.2019.03.013
[82] ZHANG T T, ZHAO W Y, XIE B Z, et al. Effects of Auricularia auricula and its polysaccharide on diet-induced hyperlipidemia rats by modulating gut microbiota[J]. Journal of Functional Foods,2020,72:104038. doi: 10.1016/j.jff.2020.104038
[83] HUANG Y L, MA Y S, TSAI Y H, et al. In vitro hypoglycemic, cholesterol-lowering and fermentation capacities of fiber-rich orange pomace as affected by extrusion[J]. International Journal of Biological Macromolecules,2019,124:796−801. doi: 10.1016/j.ijbiomac.2018.11.249
[84] LI Y X, NIU L, GUO Q Q, et al. Effects of fermentation with lactic bacteria on the structural characteristics and physicochemical and functional properties of soluble dietary fiber from prosomillet bran[J]. LWT,2022,154:112609. doi: 10.1016/j.lwt.2021.112609
[85] LIU T T, WANG N, XU X L, et al. Effect of high quality dietary fiber of Hericium erinaceus on lowering blood lipid in hyperlipidemia mice[J]. Journal of Future Foods,2022,2(1):61−68. doi: 10.1016/j.jfutfo.2022.03.018
[86] CHEN H H, LI J, YAO R X, et al. Mechanism of lipid metabolism regulation by soluble dietary fibre from micronized and non-micronized powders of lotus root nodes as revealed by their adsorption and activity inhibition of pancreatic lipase[J]. Food Chemistry,2020,305:125435. doi: 10.1016/j.foodchem.2019.125435
[87] RIVAS M Á, BENITO M J, MARTÍN A, et al. Improve the functional properties of dietary fibre isolated from broccoli by-products by using different technologies[J]. Innovative Food Science & Emerging Technologies,2022,80:103075.
[88] XU T, WU X Y, LIU J, et al. The regulatory roles of dietary fibers on host health via gut microbiota-derived short chain fatty acids[J]. Current Opinion in Pharmacology,2022,62:36−42. doi: 10.1016/j.coph.2021.11.001
[89] WANG M M, WICHIENCHOT S, HE X, et al. In vitro colonic fermentation of dietary fibers:Fermentation rate, short-chain fatty acid production and changes in microbiota[J]. Trends in Food Science & Technology,2019,88:1−9.
[90] BAI X L, HE Y, QUAN B Y, et al. Physicochemical properties, structure, and ameliorative effects of insoluble dietary fiber from tea on slow transit constipation[J]. Food Chemistry: X,2022,14:100340. doi: 10.1016/j.fochx.2022.100340
[91] JIA F J, YANG S F, MA Y Y, et al. Extraction optimization and constipation-relieving activity of dietary fiber from Auricularia polytricha[J]. Food Bioscience,2020,33:100506. doi: 10.1016/j.fbio.2019.100506
[92] ZHANG Z H, FAN S T, HUANG D F, et al. Effect of Lactobacillus plantarum NCU116 fermentation on Asparagus officinalis polysaccharide:Characterization, antioxidative, and immunoregulatory activities[J]. Journal of Agricultural and Food Chemistry,2018,66(41):10703−10711. doi: 10.1021/acs.jafc.8b03220
[93] SUN H, NI X Q, SONG X, et al. Fermented Yupingfeng polysaccharides enhance immunity by improving the foregut microflora and intestinal barrier in weaning rex rabbits[J]. Applied Microbiology and Biotechnology,2016,100(18):8105−8120. doi: 10.1007/s00253-016-7619-0
[94] HE Y, WANG B X, WEN L K, et al. Effects of dietary fiber on human health[J]. Food Science and Human Wellness,2022,11(1):1−10. doi: 10.1016/j.fshw.2021.07.001
[95] BAENAS N, NUÑEZ-GÓMEZ V, NAVARRO-GONZÁLEZ I, et al. Raspberry dietary fibre:Chemical properties, functional evaluation and prebiotic in vitro effect[J]. LWT,2020,134:110140. doi: 10.1016/j.lwt.2020.110140
[96] BENÍTEZ-PÁEZ A, GÓMEZ DEL PULGAR E M, KJØLBÆK L, et al. Impact of dietary fiber and fat on gut microbiota re-modeling and metabolic health[J]. Trends in Food Science & Technology,2016,57:201−212.
[97] CUI X Y, GOU Z Y, JIANG Z Y, et al. Dietary fiber modulates abdominal fat deposition associated with cecal microbiota and metabolites in yellow chickens[J]. Poultry Science,2022,101(4):101721. doi: 10.1016/j.psj.2022.101721
[98] MADRERA R R, BEDRIÑANA R P, VALLES B S. Enhancement of the nutritional properties of apple pomace by fermentation with autochthonous yeasts[J]. LWT-Food Science and Technology,2017,79:27−33. doi: 10.1016/j.lwt.2017.01.021
[99] 赵久毅. 豆渣膳食纤维不同预处理对小鼠润肠通便功能的对比研究[D]. 重庆:重庆工商大学, 2022:41−58. [ZHAO J Y. A comparative study of different pretreatments of dietary fiber from soybean dregs on the laxative function of mice[J]. Chongqing:Chongqing Business University, 2022:41−58.] ZHAO J Y. A comparative study of different pretreatments of dietary fiber from soybean dregs on the laxative function of mice[J]. Chongqing: Chongqing Business University, 2022: 41−58.
[100] ZHENG J, WU J H, DAI Y Y, et al. Influence of bamboo shoot dietary fiber on the rheological and textural properties of milk pudding[J]. LWT,2017,84:364−369. doi: 10.1016/j.lwt.2017.05.051
[101] SUN C C, WU X F, CHEN X J, et al. Production and characterization of okara dietary fiber produced by fermentation with Monascus anka[J]. Food Chemistry,2020,316:126243. doi: 10.1016/j.foodchem.2020.126243
[102] WU N N, QIAO C C, TIAN X H, et al. Retrogradation inhibition of rice starch with dietary fiber from extruded and unextruded rice bran[J]. Food Hydrocolloids,2021,113:106488. doi: 10.1016/j.foodhyd.2020.106488
[103] ZHAO H, WANG L W, BRENNAN M, et al. How does the addition of mushrooms and their dietary fibre affect starchy foods[J]. Journal of Future Foods,2022,2(1):18−24. doi: 10.1016/j.jfutfo.2022.03.013
[104] MA S, WANG Z, LIU H M, et al. Supplementation of wheat flour products with wheat bran dietary fiber:Purpose, mechanisms, and challenges[J]. Trends in Food Science & Technology,2022,123:281−289.
[105] CODA R, KÄRKI I, NORDLUND E, et al. Influence of particle size on bioprocess induced changes on technological functionality of wheat bran[J]. Food Microbiology,2014,37:69−77. doi: 10.1016/j.fm.2013.05.011
[106] 孙聪聪. 发酵法制备膳食纤维红曲及其在面包中的应用[D]. 合肥:合肥工业大学, 2020:34−51. [SUN C C. Preparation of Monascus dietary fiber by fermentation and its application in bread[D]. Hefei:Hefei Polytechnic University, 2020:34−51.] SUN C C. Preparation of Monascus dietary fiber by fermentation and its application in bread[D]. Hefei: Hefei Polytechnic University, 2020: 34−51.
[107] CARVALHO L T, PIRES M A, BALDIN J C, et al. Partial replacement of meat and fat with hydrated wheat fiber in beef burgers decreases caloric value without reducing the feeling of satiety after consumption[J]. Meat Science,2019,147:53−59. doi: 10.1016/j.meatsci.2018.08.010
[108] YOUNIS K, YOUSUF O, QADRI O S, et al. Incorporation of soluble dietary fiber in comminuted meat products:Special emphasis on changes in textural properties[J]. Bioactive Carbohydrates and Dietary Fibre,2022,27:100288. doi: 10.1016/j.bcdf.2021.100288
[109] XU Y, YAN H M, XU W P, et al. The effect of water-insoluble dietary fiber from star anise on water retention of minced meat gels[J]. Food Research International,2022,157:111425. doi: 10.1016/j.foodres.2022.111425
[110] 张俊, 雷激, 刘江, 等. 发酵麸皮对火腿肠品质的影响[J]. 食品与发酵工业,2019,45(22):165−170. [ZHANG J, LEI J, LIU J, et al. Effect of fermented bran on the quality of ham sausage[J]. Food and Fermentation Industry,2019,45(22):165−170.] doi: 10.13995/j.cnki.11-1802/ts.021670 ZHANG J, LEI J, LIU J, et al . Effect of fermented bran on the quality of ham sausage[J]. Food and Fermentation Industry,2019 ,45 (22 ):165 −170 . doi: 10.13995/j.cnki.11-1802/ts.021670[111] REPIN N, SCANLON M G, GARY FULCHER R. Phase behaviour of casein micelles and barley beta-glucan polymer molecules in dietary fibre-enriched dairy systems[J]. Journal of Colloid and Interface Science,2012,377(1):7−12. doi: 10.1016/j.jcis.2012.02.021
[112] 董瑞红. 胡萝卜膳食纤维中结合多酚的体外消化酵解规律和营养功能特性及其在酸奶中的应用[D]. 南昌:南昌大学, 2021:53−61. [DONG R H. The release and nutritional functional properties of bound polyphenols from carrot dietary fiber during in vitro digestion and colonic fermentation and its application in yogurt[D]. Nanchang:Nanchang University, 2021:53−61.] DONG R H. The release and nutritional functional properties of bound polyphenols from carrot dietary fiber during in vitro digestion and colonic fermentation and its application in yogurt[D]. Nanchang: Nanchang University, 2021: 53−61.
[113] 高天宇. 担子菌门食用型真菌发酵改良大豆膳食纤维风味研究[D]. 无锡:江南大学, 2021:34−44. [GAO T Y. Research on the soybean dietary fiber flavor improvement by fermentation of basidiomycota edible fungi[D]. Wuxi:Jiangnan University, 2021:34−44.] GAO T Y. Research on the soybean dietary fiber flavor improvement by fermentation of basidiomycota edible fungi[D]. Wuxi: Jiangnan University, 2021: 34−44.
[114] WU T F, DENG C, LUO S J, et al. Effect of rice bran on properties of yogurt:Comparison between addition of bran before fermentation and after fermentation[J]. Food Hydrocolloids,2023,135:108122. doi: 10.1016/j.foodhyd.2022.108122





 下载:
下载:
 下载:
下载:



