Effects of Freezing and Thawing Cycles on Quality and Processing Characteristics of Surimi
-
摘要: 为研究不同低温条件下的冻融循环对鱼糜加工品质变化的影响。将制得的鱼糜样品定量分装成3组,分别置于4、−18、−35 ℃冰箱中,并模拟5次冻融循环。期间,分别进行各组冻融鱼糜的流变特性、凝胶特性、水分变化(低场核磁共振、冷冻损失率、解冻损失率、蒸煮损失率和持水性)、质构特性,并结合微观结构观察,以综合判定贮藏温度对鱼糜冻融循环品质变化影响。结果表明,贮藏温度和冻融循环是影响鱼糜品质变化的重要因素,贮藏温度越低,鱼糜品质变化越小;随低温冻融循环次数的增加,鱼糜凝胶黏度下降,G’和G”减小、tanδ增大,硬度、咀嚼性、凝胶强度呈现先下降后增加的趋势,−18 ℃和−35 ℃条件下冻融循环5次后冷冻损失、解冻损失分别为2.18%、22.87%和2.38%、10.20%,鱼糜持水性分别由80.04%降低至73.51%和75.33%。本研究可为鱼糜及其凝胶在加工与贮运过程中品质保持提供理论参考。Abstract: In order to study the effect of freeze-thaw cycle on quality of surimi under different low temperature conditions, the prepared surimi samples were divided into 3 groups and placed in the refrigerator at 4, −18, −35 ℃, respectively. Five repeated freeze-thaw cycles were simulated. Different indexes, such as rheological characteristics, gel properties, water changes (low field nuclear magnetic resonance (LF-NMR), freezing loss, thawing loss, cooking loss rate, water holding capacity (WHC)), texture characteristic, were analyzed, which also combined with the microstructure observation, so as to assess storage temperature influence on the quality of minced fish during freeze-thaw cycles comprehensively. The results showed that the storage temperature and freezing-thawing cycle were important factors affecting the quality of surimi. The lower the storage temperature was, the less the quality of surimi changed. With the increase of the freezing-thawing cycle, the viscosity of surimi gel decreased, G' and G" decreased, and tanδ increased, and the hardness, chewiness, gel strength decreased first and then increased. The freezing loss and thawing loss were 2.18% and 22.87%, 2.38% and 10.20% at −18 ℃ and −35 ℃ for 5 cycles, respectively. The WHC of surimi decreased from 80.04% to 73.51% and 75.33%, respectively. The results of this study can provide theoretical reference for quality maintenance of surimi and its gel during circulation.
-
Keywords:
- surimi /
- gel /
- storage /
- freeze-thaw cycles /
- physical and chemical quality
-
鱼糜制品营养丰富,食用方便,深受消费者的喜爱[1]。冷冻鱼糜是加工鱼糜制品的主要原料[2],鱼糜的凝胶特性是鱼糜的主要功能特性,鱼糜凝胶是蛋白质经热变性后有序聚集形成的三维网状结构[3],具有较好的可塑性和粘弹性[4]。
鱼糜通常在冷冻状态下流通[5]。冻藏与温度波动会使鱼糜发生变性、聚集与功能特性丧失[6-7]。冷冻速度、冷冻时间和贮藏温度等都会对鱼糜及其产品的质量带来影响[8]。研究表明,冷冻和解冻会导致鱼糜品质劣变,而劣变程度取决于温度的变化[9]。温度波动引发的重结晶会导致形成大冰晶[10-11],而大冰晶的形成及其在鱼糜中的分布不均匀会导致鱼糜的感官品质下降和解冻后营养物质的流失[12]。此外,冷冻引起的蛋白质变性会导致水从肌原纤维向肌原纤维间的缝隙移动和造成结合水解离,这都将导致鱼糜含水量下降[13],冷冻食品中的水分含量与其产品的质量以及蛋白质的变性程度紧密相关。其中,LIU等[14]研究发现随着冻藏温度的降低,蛋白质理化性质的变化趋势变得迟缓,冻藏温度的高低会影响蛋白质冷冻变性的程度,进而影响鱼糜品质和加工特性。EGELANDSDAL[15]与ZHANG等[16]研究发现,反复冻融鱼糜中的冰晶大小和分布不断变化,导致蛋白发生聚集、重排和不可逆变性,致使凝胶结构破坏,鱼糜品质下降。目前国内对温度波动条件下淡水鱼糜品质变化规律及贮藏保鲜的研究较少,为此,本研究通过冻融循环的方式模拟草鱼糜贮运过程中的温度波动,旨在探究贮藏温度和低温冻融循环对鱼糜及其凝胶品质的变化规律,为鱼糜类产品的贮藏保鲜以及精深加工提供理论参考。
1. 材料与方法
1.1 材料与仪器
鲜活草鱼(Ctenopharyngodon idella) 购于福州永辉超市,要求个体均一,大小一致,鱼体重量为1250~1500 g,长度为(45±5)cm;氯化钠、无水乙醇 分析纯,西陇科学有限公司;戊二醛 分析纯,麦克林生化科技公司;其他药品与试剂均为分析纯。
T18型数码均质机 艾卡仪器设备有限公司;MCR302旋转流变仪 奥地利安东帕有限公司;M1N1 MR核磁共振分析仪 上海纽迈电子科技有限公司;TA. XT PLUS型质构仪 英国SMS公司;Helios G4 CX型场发射扫描电子显微镜 Thermo Scientific公司。
1.2 实验方法
1.2.1 鱼糜制备
参考GAO等[17]的方法并稍作修改。将新鲜草鱼去除头部尾部内脏和鱼皮,冲洗干净切块,放于绞肉机中绞碎;加入5倍体积的清水慢速搅拌10 min,在10000 r/min、4 ℃条件下脱水10 min,取沉淀重复上述操作,然后取沉淀加入5倍体积0.15% NaCl溶液慢速搅拌10 min,在10000 r/min、4 ℃条件下脱水15 min,其沉淀即为鱼糜样品,将制得的鱼糜样品定量分装,置于4、−18、−35 ℃冰箱中贮藏备用。
1.2.2 鱼糜凝胶制备
将鱼糜样品放入研钵中空斩2 min,然后加入3%(W/W)的NaCl斩拌3 min,即为鱼糜溶胶。将斩拌获得的鱼糜注入直径为10 mm的圆柱形模具中,并用保鲜膜封紧,采用二段加热法,先在40 ℃水浴锅中凝胶化30 min,然后迅速转移入90 ℃水浴锅中凝胶化30 min,最后将加热后的鱼糜立即置于冰水中冷却20 min备用,即为鱼糜凝胶[18]。
1.2.3 试验设计
参考WU等[19]方法并稍作修改。新鲜样品即为制备好的鱼糜不做冷冻处理直接进行试验。将定量分装的鱼糜分别放置不同温度的冰箱中贮藏3 d后,全部取出于25 ℃恒温箱中解冻2 h,每个温度下鱼糜随机取一份进行试验,其余样品均继续放于原冰箱中冷冻为低温冻融循环1次;贮藏至第6 d时,重复之前操作,依次重复,并以温度-冻融循环次数的形式命名(例4-1为4 ℃下低温冻融循环1次)。
1.2.4 流变学特性
鱼糜凝胶的流变性能通过旋转流变仪测量,使用直径为25 mm的探头,设置零间隙为1 mm。将鱼糜溶胶样品置于流变仪的传感系统上,平衡5 min使其达到稳态(25 ℃)。温度扫描采用1 Hz作为测试频率,温度测试范围为20~90 ℃,升温速率为2 ℃/min。测试开始前在待测样品周围涂上一层二甲基硅油防止样品水分蒸发,每个样品测试3次。剪切黏度试验是在旋转流动模式下进行的,剪切速率为0.1~10 1/s。剪切速率-黏度曲线符合幂律模型[20]:
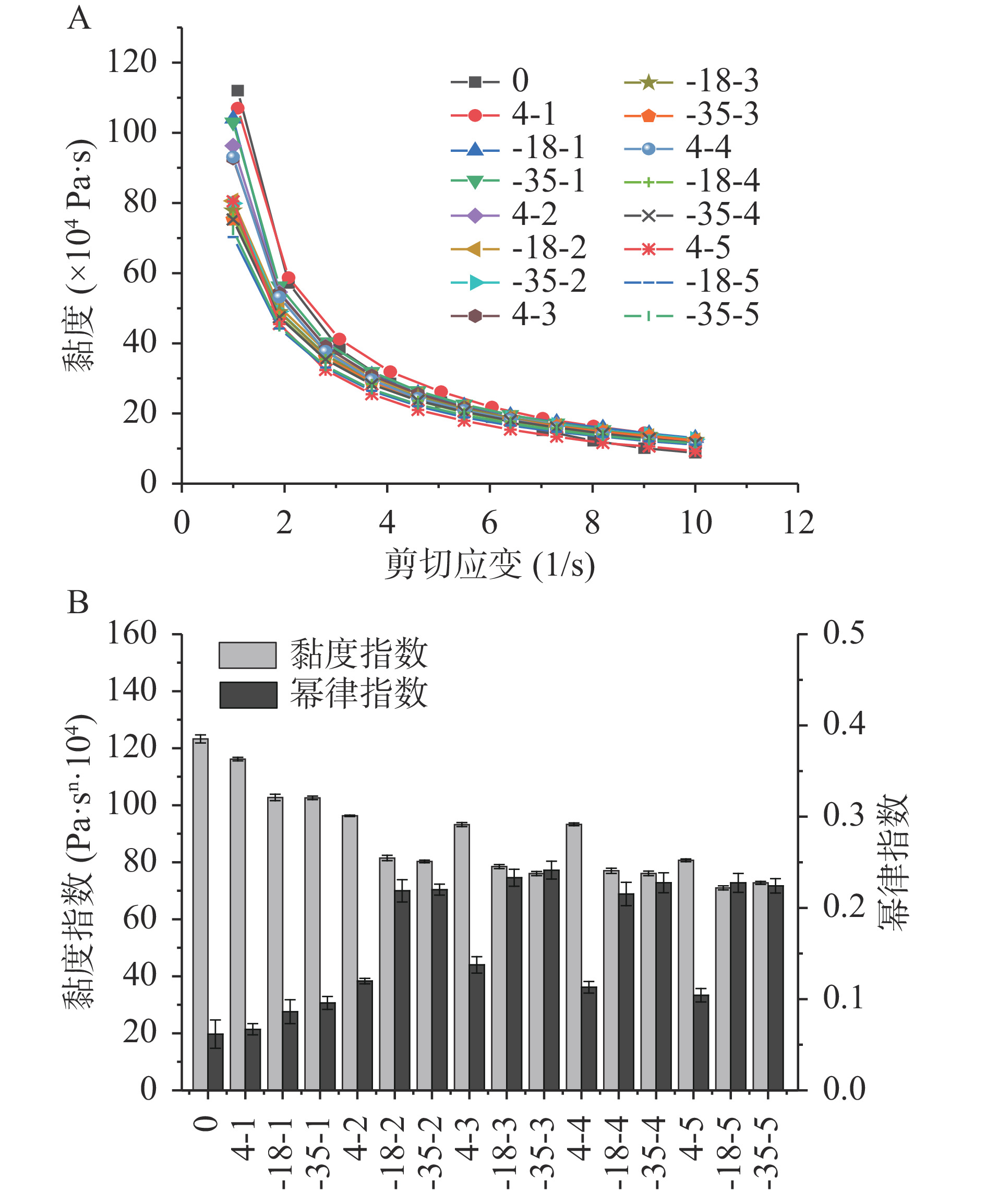
η=K˙γn−1 式中,η为特定剪切速度下的黏度,K为黏度指数,
˙γ 为剪切速率,n为描述材料剪切行为的幂律指数。1.2.5 水分迁移
采用low field nuclear magnetic reso-nance (LF-NMR)分析仪检测横向弛豫时间T2,将高度为20 mm的鱼糜凝胶放入直径为10 mm的LF-NMR玻璃管中,随后将其插入分析仪中。重复采样等待时TW是4000 ms,扫描次数NS是8,回波个数NECH是8000。每个样品重复6次。
1.2.6 冷冻损失率
参考LIU等[21]的方法并稍作修改。样品在冷冻前称重(M2),冷冻结束后立即称重(M1)。每组处理测3次平行,结果为3次测定平均值。
冷冻损失率(%)=M2−M1M2×100 1.2.7 解冻损失率
参考LIU等[21]的方法并稍作修改。样品在解冻前称重(M0),解冻后样品用吸水纸吸干并立即称重(M1)。每组处理测3次平行,结果为3次测定平均值。
解冻损失率(%)=M0−M1M0×100 1.2.8 蒸煮损失率
参考YANG等[22]的方法并稍作修改。准确称取3.0 g不同处理组的凝胶样品,放入自封袋中,封口后在90 ℃水浴锅中加热30 min后,冷却至室温,用滤纸擦去凝胶表面的水分后称量。每组处理测3次平行,结果为3次测定平均值。
蒸煮损失率(%)=生样品质量−熟样品质量生样品质量×100 1.2.9 凝胶持水性
取1 g鱼糜凝胶样品,精确称取质量,用滤纸包好,5000 g离心10 min后,取出样品再次精确称取质量,凝胶持水性为离心前与离心后的样品质量之比[23]。每组处理测3次平行,结果为3次测定平均值。
凝胶持水性(%)= 离心后样品质量 离心前样品质量 ×100 1.2.10 质构特性和凝胶强度分析
参考LIU等[24]的方法并稍作修改。将鱼糜凝胶制备成直径为10 mm,高10 mm的小圆柱,用TA-XT plus质构仪进行质构分析。具体参数为:测试探头36 R,测前速度为2.00 mm·s−1,测中速度为1.00 mm·s−1,测后速度为5.00 mm·s−1,压缩比为40%,压力为5.0 g,探头距离测试台高度为30 mm,每个样品共压缩2次,测定温度为室温,每组测定10个平行样品。利用TA-XT plus质构仪测定凝胶强度,采用P/5S探头,测定速度2.00 mm/s,压缩比为50%,压力为20.0 g。鱼糜凝胶的凝胶强度用破断强度(g)与凹陷深度(cm)的乘积表示,每组测定10个平行样品。
1.2.11 鱼糜凝胶微观形貌
参考YU等[25]的方法并稍作修改。将鱼糜凝胶切成小块,用4%的戊二醛固定24 h,弃取固定液后用体积分数为30%、50%、70%、85%、95%和100%的乙醇梯度脱水一次,25 min/次,再将样品放入通风橱中风干,离子溅射镀金,用扫描电子显微镜观察微观结构。
1.3 数据处理
采用Origin 9.0软件作图,Excel 2010和SPSS 26.0进行图表绘制和统计学分析,数据形式以平均数±标准差表示。数据间差异显著性(P<0.05)分析采用ANOVA方法,利用SIMCA 14.1进行主成分分析。
2. 结果与分析
2.1 鱼糜溶胶的流变特性
2.1.1 剪切黏度
对鱼糜溶胶样品进行剪切应变扫描,结果如图1A所示。由图1A可知,所有样品黏度均随剪切速率的增加而降低,表明样品是具有剪切变稀的假塑性流体。通过拟合剪切速率-黏度曲线幂律模型,得到不同样品的黏度指数(K)和幂律指数(n),如图1B所示。所有样品的n值均小于1,说明鱼糜溶胶具有较强的剪切稀化行为[26],且随着贮藏温度的降低和低温冻融循环次数的增加,其黏度指数呈下降趋势,这与贮藏过程中肌原纤维蛋白的冷冻变性以及鱼糜中水分的流失有关,低温冻融循环次数越多,肌原纤维蛋白的变性越严重,导致蛋白间的交联越弱,黏度越低。在贮藏过程中4 ℃冷藏条件下其K值略高,这是由于冷藏使鱼糜水分大量流失,导致肌原纤维蛋白浓度相对较高,该结果与CHEN等[27]研究一致。
2.1.2 温度扫描
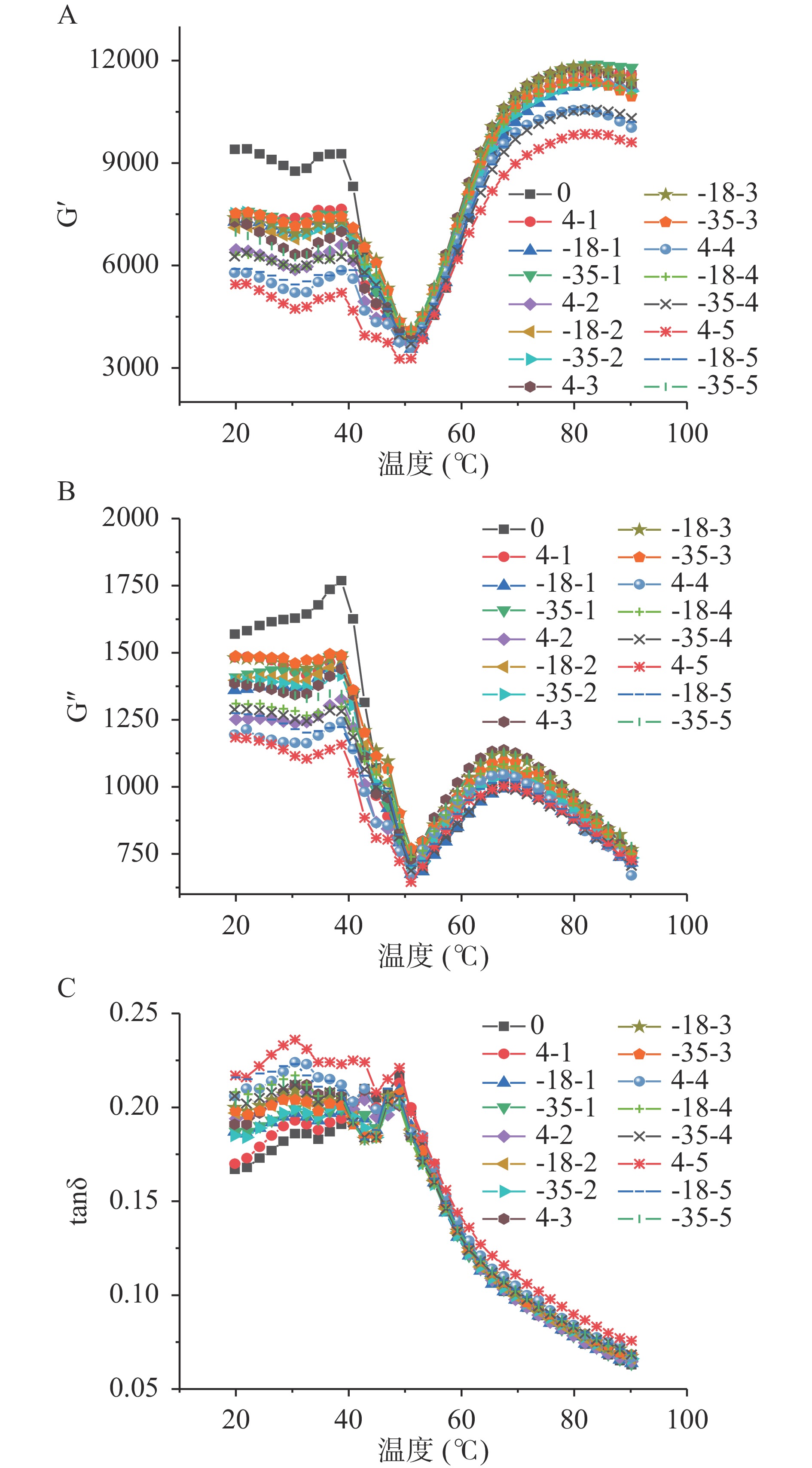
不同样品储能模量G’、损耗模量G”和损耗因子tanδ随温度的变化曲线如图2所示。鱼糜溶胶G’在40 ℃前呈先降后升趋势,可能由于肌球蛋白尾部变性,α-螺旋结构解离,分子间通过化学键形成凝胶网络[28]。当温度达到40 ℃后,内源性水解酶活性增加,凝胶网络结构断裂,蛋白质分子向外延伸,鱼糜凝胶流动性增强[29-30]。当温度到达50 ℃时,随温度增加G’迅速增大,疏水键、二硫键等化学键形成,鱼糜内部形成稳定有序的不可逆凝胶网络[31-32]。图2A~图2C中可以看出,G’和G”随贮藏温度的升高和低温冻融循环次数的增加呈下降趋势,tanδ的变化与G’和G”呈相反趋势,新鲜鱼糜具有较高的G’和较低的tanδ,且4 ℃冷藏条件下低温冻融循环4次后,其tanδ显著高于冻藏组(P<0.05)。可能由于鱼糜经多次冻融后,肌原纤维蛋白发生变性,鱼糜内部蛋白质交联受到影响,凝胶化性能降低[33]。
2.2 水分迁移
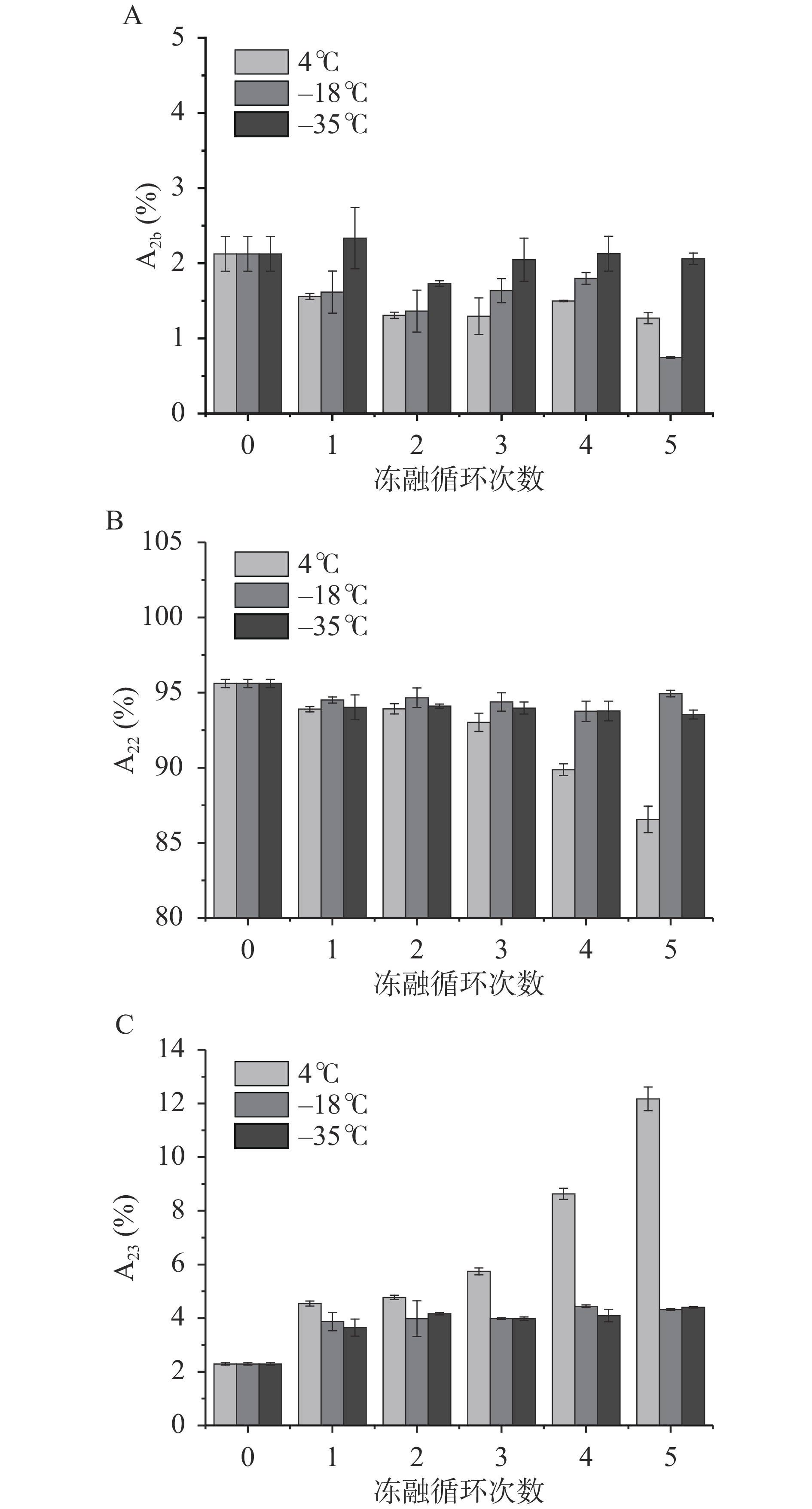
鱼糜凝胶中含有三种状态的水,分别是结合水、不可移动水和自由水,低场核磁可以快速无损的检测这三种状态的水在鱼糜凝胶中的迁移率和分布变化[34],其含量分别用A2b、A22和A23表示,这些变化与蛋白质的变性和聚集有关。从图3可知,鱼糜凝胶中95%以上的水以不可移动水的形式存在,随着冻融循环次数的增加,结合水(图3A)和不可移动水(图3B)含量下降,而自由水(图3C)含量升高,4 ℃贮藏条件下不可移动水含量由95.61%下降至86.64%、自由水含量由2.29%增加至12.18%,其变化最为明显,这表明鱼糜凝胶中不可移动水向自由水方向转化,这是由于在贮藏过程中肌原纤维蛋白发生变性,使得鱼糜凝胶结构变得疏松多孔。LI等[35]也研究认为,冻融循环导致的水分的再分配和细胞外冰晶的形成是凝胶结构中不可移动水向自由水方向转化的主要原因。此外,在−18 ℃冻融循环第5次时,结合水含量由1.79%(第4次)迅速减至0.75%,这可能由于冰晶重分布和蛋白质变性引起的机械损伤使得结合水向不可移动水形式转化,不可移动水含量增加,导致结合水含量相对降低[36]。
2.3 水分特性分析
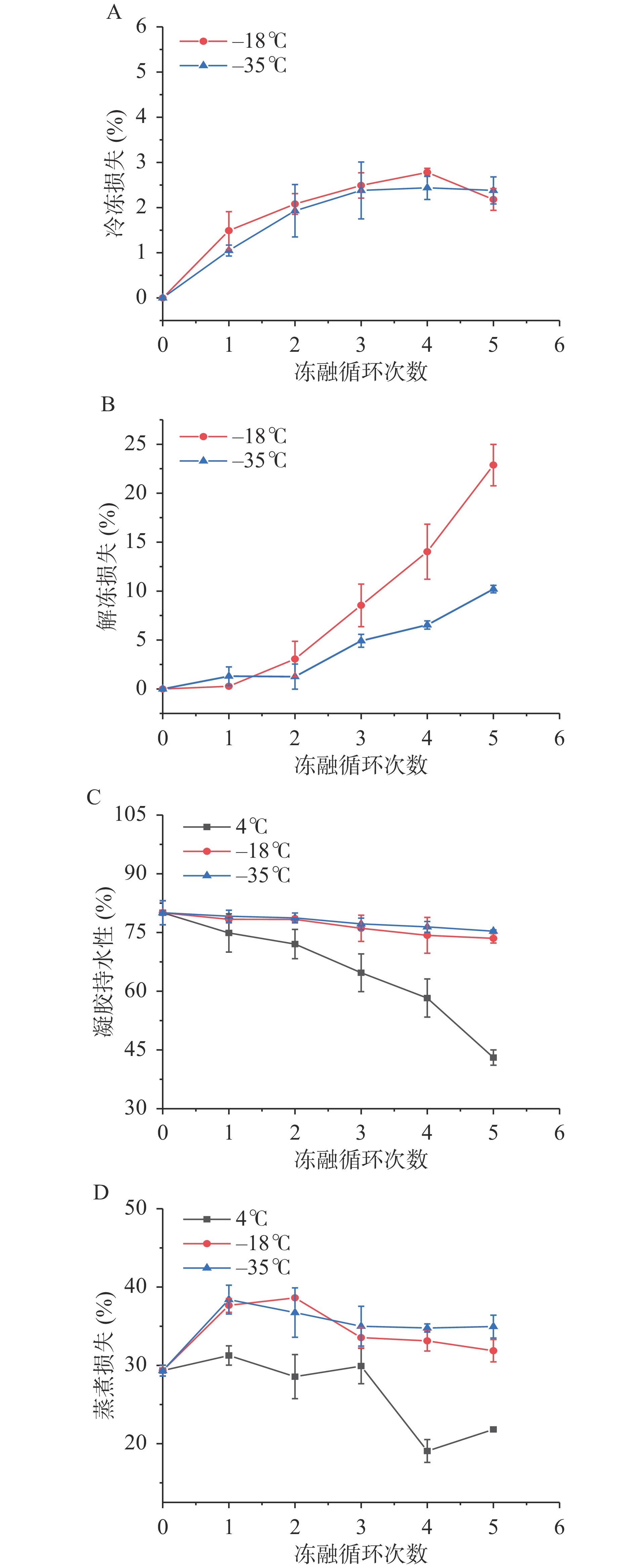
进一步对不同贮藏温度下鱼糜的冷冻损失、解冻损失、持水性、蒸煮损失进行分析,结果见图4。在冷冻过程中,当鱼糜表面的水蒸气压低于周围空气时,冰从鱼糜表面升华到周围空气中会导致冷冻损失的产生[37],由图4A所示,−18 ℃和−35 ℃条件下冻融循环5次后冷冻损失分别为2.18%和2.38%,且无明显差异。解冻损失越低表明样品品质越好[38-39]。由图4B所示,解冻损失随着冻融次数的增加而增大,经5次冻融后−18 ℃和−35 ℃下解冻损失分别为22.87%和10.20%,−18 ℃下其解冻损失显著高于−35 ℃(P<0.05),这是由于−35 ℃超低温冷冻可以使鱼糜更快的达到最大冰晶生成区,形成的小冰晶对组织的机械损伤较小。
鱼糜凝胶持水性可以反映出凝胶结构致密程度[40],与凝胶含水量以及网络结构孔隙大小等多种因素有关,含水量越高、结构越松散,对水分的保持能力就越弱[41]。由图4C所示,−18 ℃和−35 ℃条件下,凝胶持水性随冻融循环次数的增加略微下降,经5次冻融循环后分别下降至73.51%和75.33%,表明低温冷冻可以较好的抑制水分的流失。4 ℃条件下经5次冻融循环后凝胶持水性由80.04%显著下降至43.05%(P<0.05),这是由于蛋白结构改变损害了凝胶基质,与ZHANG等[42]及LEYGONIE等[43]研究结果相一致。由图4D所示,−18 ℃和−35 ℃冻藏条件下,草鱼糜在第一次冻融循环后蒸煮损失迅速增加,而后微降并逐渐趋于稳定,说明冻藏可以有效地降低鱼糜在冻藏过程中水分的流失。4 ℃冷藏条件下蒸煮损失有所下降,这是由于冷藏致使鱼糜解冻损失、冷冻损失严重,水分含量降低所致。
2.4 质构及凝胶特性分析
质构和凝胶强度可直观反映鱼糜凝胶品质的好坏,主要受贮藏温度和冻融循环次数的影响。由表1可知,不同贮藏温度下草鱼鱼糜的弹性在冻融循环2次后无显著变化(P>0.05),但到第5次冻融循环后,4 ℃贮藏条件下的草鱼鱼糜的弹性显著降低(P<0.05),并显著低于冻藏组,SUN等[44]认为鱼糜样品在4 ℃贮藏第8 d时质构特性及营养价值显著降低且失去了商品价值。在−18 ℃和−35 ℃贮藏过程中,随着冻融循环次数的增加,硬度、咀嚼性和凝胶强度大致呈先降后升趋势,下降是因为冷冻贮藏过程中发生的蛋白质变性和凝胶持水性的丢失以及肌球蛋白和氨基酸的氧化变化,阻碍了蛋白质在凝胶化过程中的交联,导致形成孔隙和较低弹性的不良凝胶网络[45],与WANG等[46]研究结果相一致。后期增加是由于水分的流失使得蛋白浓度相对提高所致[47]。CHEN等[48]也证实了反复冻融过程中水分含量的降低会导致肉类制品硬度、咀嚼度的升高。
表 1 不同贮藏温度下鱼糜凝胶的质构特性和凝胶强度Table 1. Texture properties and gel strength of surimi gel at different storage temperature样品 硬度(g) 弹性 咀嚼性 凝胶强度(g·cm) 4-0 344.50±12.73a 0.98±0.00a 285.44±10.69a 768.70±73.77a 4-1 296.22±7.79b 0.97±0.01a 248.47±4.34b 734.41±25.59a 4-2 300.08±2.91b 0.97±0.01a 246.11±5.71b 345.48±30.49b 4-3 275.13±12.76c 0.96±0.01a 219.34±6.95c 279.13±22.13c 4-4 249.95±7.33d 0.97±0.01a 189.10±4.34d 212.94±7.73d 4-5 192.66±7.55e 0.87±0.22b 130.66±6.30e 206.95±11.96d −18-0 344.50±12.73bc 0.98±0.00ab 285.44±10.69b 768.70±73.77a −18-1 372.40±9.72a 0.98±0.01a 310.35±6.17a 740.80±46.43ab −18-2 315.30±6.96c 0.97±0.00ab 269.74±5.90c 736.15±95.58abc −18-3 332.61±7.71d 0.97±0.00b 287.54±6.94b 713.57±40.61abc −18-4 376.65±3.08a 0.98±0.01ab 317.82±6.78a 663.16±40.36c −18-5 350.79±5.93b 0.97±0.01ab 295.58±3.61b 675.26±45.46bc −35-0 344.5±12.73b 0.98±0.00a 285.44±10.69b 768.70±73.77a −35-1 334.45±13.49bc 0.98±0.00a 283.98±0.97b 777.47±18.10a −35-2 294.31±8.84d 0.96±0.01a 259.78±5.02c 679.34±69.98b −35-3 319.44±15.09c 0.98±0.01a 269.67±20.39bc 692.18±53.74b −35-4 323.33±10.99bc 0.96±0.02a 281.81±14.46b 677.57±37.26b −35-5 384.23±12.14a 0.97±0.00a 329.46±6.30a 690.43±23.93b 注:结果以平均值±标准偏差表示,每列中不同的字母表示各温度下不同低温冻融循环次数之间存在显著性差异(P<0.05)。 2.5 微观结构分析
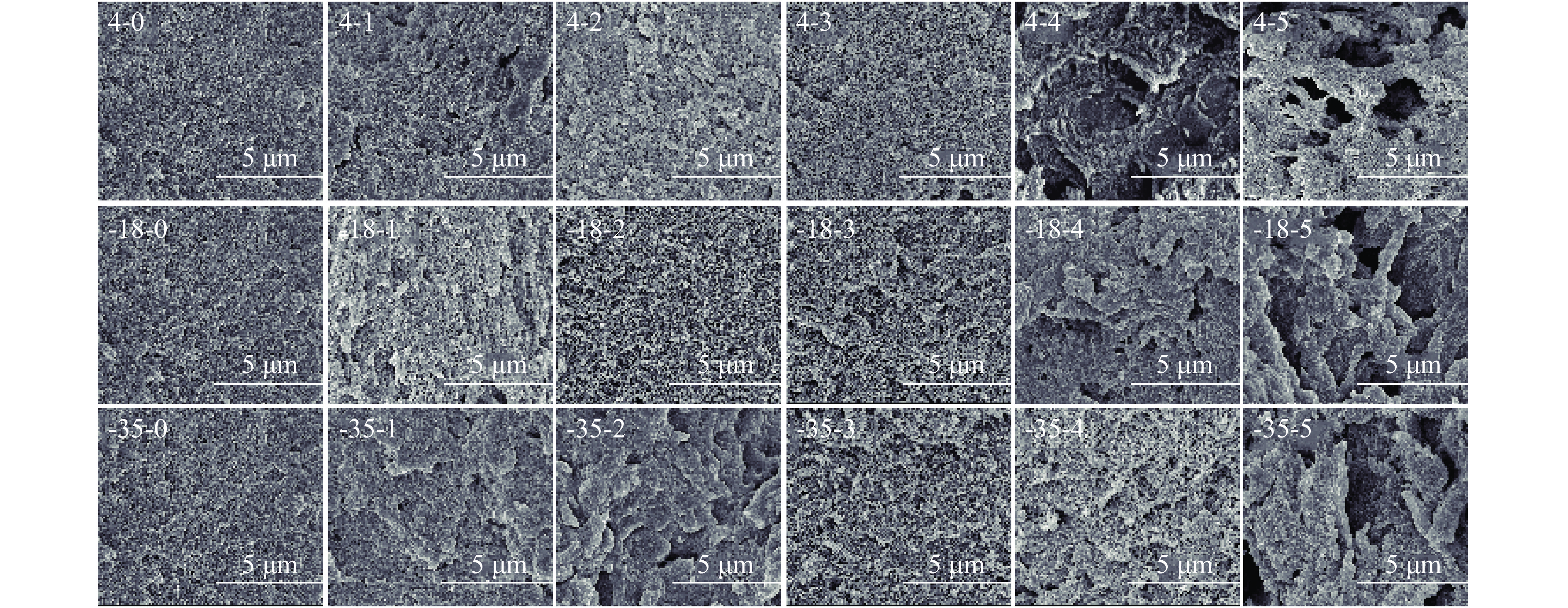
扫描电子显微镜是观察蛋白质微观结构的常用手段之一[45]。从图5中可以看出,新鲜的鱼糜凝胶表面比较平整,排列整齐,形成了致密的网络结构。随着冻融循环次数的增加,各组鱼糜空洞均有增加,且孔隙大小不一,这可能是因为在贮藏过程中,鱼糜蛋白质在微生物以及内源酶的作用下发生氧化变性,导致鱼糜凝胶品质发生劣变[49]。−18 ℃和−35 ℃冻藏条件下鱼糜凝胶在第4次冻融循环后其结构仍然较为均匀,只出现了部分小空洞,且−35 ℃冻藏条件下的凝胶结构更为完整,这也与冻藏过程中冰晶的形成以及重结晶有关,冻藏温度越低,冻结速度越快,形成的冰晶小而致密,对凝胶结构的破坏就越小。相较于冻藏组,4 ℃条件下贮藏的鱼糜制成的凝胶空洞较为明显,结构变得松散不均匀,其凝胶品质下降较为严重。CAI等[50]也研究发现,冷冻储存会导致凝胶结构排列松散,损伤严重时出现凹坑和裂缝现象。
2.6 聚类分析
为了进一步可视化低温冻融条件下贮藏温度与鱼糜品质之间的关系,对3种贮藏温度下的样品进行了PCA聚类分析。结果如图6所示,4 ℃低温条件下,冻融循环4次和5次的样品可聚为一类,4 ℃低温条件下冻融循环2次和3次以及−18 ℃冻藏条件下冻融循环5次的可聚为一类,其余样品可另聚为一类。这表明贮藏温度和冻融循环次数是影响鱼糜贮藏品质的重要因素。
2.7 相关性分析
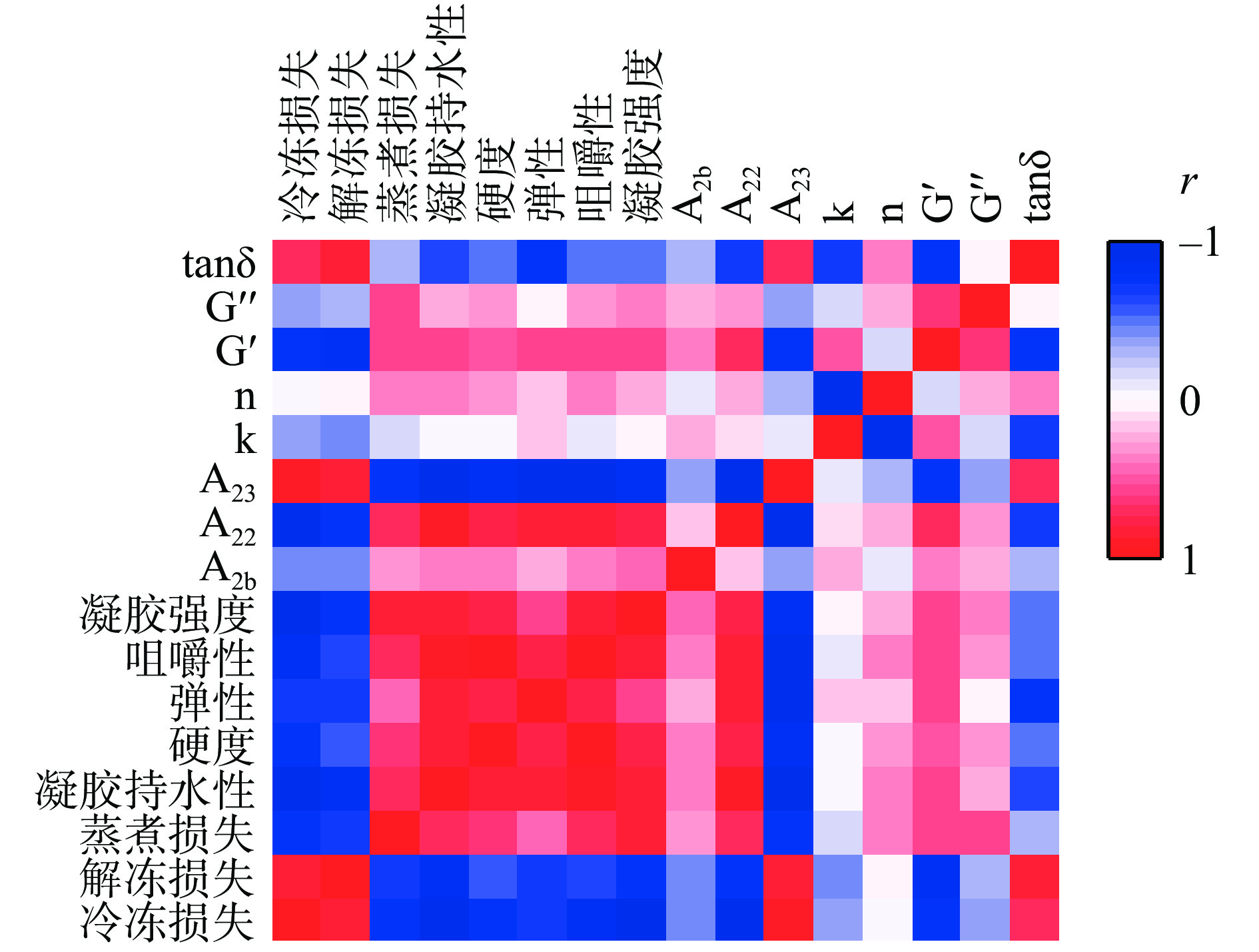
本研究旨在评估低温冻融循环条件下贮藏温度对鱼糜质构特性和加工特性的影响,而不同的贮藏温度导致水分有不同程度的流失,而水分的流失严重影响鱼糜品质,为此,建立了鱼糜流变和凝胶特性等与鱼糜水分相关指标的相关性分析。图7表示皮尔逊相关系数(r)在-1和1之间的变化,并使用颜色尺度更好的解释其相关程度。在图中,鱼糜凝胶强度、咀嚼性、弹性、硬度、G’和G”与凝胶持水性、蒸煮损失、A22呈现正相关,与冷冻损失、解冻损失、A23呈现负相关,tanδ与其相关性相反。与G’、G”和tanδ相比,鱼糜凝胶强度、咀嚼性、硬度与凝胶持水性(r>0.8)、蒸煮损失(r>0.65)、A22(r>0.80)、冷冻损失(r<−0.75)、解冻损失(r<−0.65)、A23(r<−0.80)的相关性较高,这说明贮藏导致的鱼糜水分流失对其凝胶的质构特性影响较大。
3. 结论
本研究探讨了冻融循环条件下鱼糜及其凝胶在不同贮藏温度下的品质变化规律。研究表明,4 ℃冻融循环条件下鱼糜品质劣变最显著;不同贮藏温度下随低温冻融循环次数的增加鱼糜凝胶黏度下降,G’和G”减小、tanδ增大,硬度、咀嚼性、凝胶强度大致呈现先下降后增加的趋势,冷冻损失、解冻损失增加,鱼糜持水性降低;鱼糜质构特性与水分之间存在很强的相关性,而冻融循环条件下贮藏温度直接影响水分流失的速度,贮藏温度越高,水分流失越严重。本研究建立了贮藏温度与鱼糜流变特性、凝胶特性、加工特性之间的关系,说明温度的波动导致的水分流失是影响鱼糜品质劣变的重要因素,因此降低贮藏温度,减少温度波动和水分流失是保持鱼糜质量的重要措施,本研究为鱼糜及鱼糜制品的保鲜提供了有益的信息。
-
表 1 不同贮藏温度下鱼糜凝胶的质构特性和凝胶强度
Table 1 Texture properties and gel strength of surimi gel at different storage temperature
样品 硬度(g) 弹性 咀嚼性 凝胶强度(g·cm) 4-0 344.50±12.73a 0.98±0.00a 285.44±10.69a 768.70±73.77a 4-1 296.22±7.79b 0.97±0.01a 248.47±4.34b 734.41±25.59a 4-2 300.08±2.91b 0.97±0.01a 246.11±5.71b 345.48±30.49b 4-3 275.13±12.76c 0.96±0.01a 219.34±6.95c 279.13±22.13c 4-4 249.95±7.33d 0.97±0.01a 189.10±4.34d 212.94±7.73d 4-5 192.66±7.55e 0.87±0.22b 130.66±6.30e 206.95±11.96d −18-0 344.50±12.73bc 0.98±0.00ab 285.44±10.69b 768.70±73.77a −18-1 372.40±9.72a 0.98±0.01a 310.35±6.17a 740.80±46.43ab −18-2 315.30±6.96c 0.97±0.00ab 269.74±5.90c 736.15±95.58abc −18-3 332.61±7.71d 0.97±0.00b 287.54±6.94b 713.57±40.61abc −18-4 376.65±3.08a 0.98±0.01ab 317.82±6.78a 663.16±40.36c −18-5 350.79±5.93b 0.97±0.01ab 295.58±3.61b 675.26±45.46bc −35-0 344.5±12.73b 0.98±0.00a 285.44±10.69b 768.70±73.77a −35-1 334.45±13.49bc 0.98±0.00a 283.98±0.97b 777.47±18.10a −35-2 294.31±8.84d 0.96±0.01a 259.78±5.02c 679.34±69.98b −35-3 319.44±15.09c 0.98±0.01a 269.67±20.39bc 692.18±53.74b −35-4 323.33±10.99bc 0.96±0.02a 281.81±14.46b 677.57±37.26b −35-5 384.23±12.14a 0.97±0.00a 329.46±6.30a 690.43±23.93b 注:结果以平均值±标准偏差表示,每列中不同的字母表示各温度下不同低温冻融循环次数之间存在显著性差异(P<0.05)。 -
[1] ZHANG L T, LI Q, SHI J, et al. Changes in chemical interactions and gel properties of heat-induced surimi gels from silver carp (Hypophthalmichthys molitrix) fillets during setting and heating: Effects of different washing solutions[J]. Food Hydrocolloids,2018,75:116−124. doi: 10.1016/j.foodhyd.2017.09.007
[2] DU X, DENG S, CHANG P, et al. Quality deterior ation mechanism and control technology of freezing surimi[J]. Science & Technology of Food Industry,2018,39(16):306−312.
[3] SHI L, WANG X F, CHANG T, et al. Effects of vegetable oils on gel properties of surimi gels[J]. LWT-Food Science and Technology,2014,57(2):586−593. doi: 10.1016/j.lwt.2014.02.003
[4] ZHOU F, WANG X C. Assessing the gelling properties of the silver carp surimi gel prepared with large yellow croaker processing by-product in freeze-thaw cycles[J]. Journal of Food Processing and Preservation,2021,45(5):1−10.
[5] TANG S W, FENG G X, CUI W X, et al. Effect of α-tocopherol on the physicochemical properties of sturgeon surimi during frozen storage[J]. Molecules,2019,24(4):1−13.
[6] BUENO M, RESCONI V C, CAMPO M M, et al. Effect of freezing method and frozen storage duration on odor-active compounds and sensory perception of lamb[J]. Food Research International,2013,54(1):772−780. doi: 10.1016/j.foodres.2013.08.003
[7] YU L H, CHEN X, CAI X X, et al. Research progress of change rules in freezing denaturation of surimi protein and its regulatory methods[J]. Food & Machinery,2020,36(8):1−8.
[8] AN Y Q, YOU J, XIONG S B, et al. Short-term frozen storage enhances cross-linking that was induced by transglutaminase in surimi gels from silver carp (Hypophthalmichthys molitrix)[J]. Food Chemistry,2018,257:216−222. doi: 10.1016/j.foodchem.2018.02.140
[9] ZHAO Y, CHEN Z G, WU T. Cryogelation of alginate improved the freeze-thaw stability of oil-in-water emulsions[J]. Carbohydrate Polymers,2018,198:26−33. doi: 10.1016/j.carbpol.2018.06.013
[10] WALAYAT N, XIONG Z, XIONG H, et al. Cryoprotective effect of egg white proteins and xylooligosaccharides mixture on oxidative and structural changes in myofibrillar proteins of Culter alburnus during frozen storage[J]. International Journal of Biological Macromolecules,2020,158:865−874. doi: 10.1016/j.ijbiomac.2020.04.093
[11] CHEN X, WU J H, CAI X X, et al. Production, structure-function relationships, mechanisms, and applications of antifreeze peptides[J]. Comprehensive Reviews in Food Science and Food Safety,2021,20(1):542−562. doi: 10.1111/1541-4337.12655
[12] ZHU Z W, ZHOU Q Y, SUN D W. Measuring and controlling ice crystallization in frozen foods: A review of recent developments[J]. Trends in Food Science & Technology,2019,90:13−25.
[13] TAN M T, XIE J. Exploring the effect of dehydration on water migrating property and protein changes of large yellow croaker (Pseudosciaena crocea) during frozen storage[J]. Foods,2021,10(4):1−14.
[14] LIU X, PENG H, LI Y, et al. Effect of storage temperature on protein characteristics and sensory quality of frozen catfish fi-llets[J]. Journal of Chinese Institute of Food Science and Technology,2019,19(1):141−147.
[15] EGELANDSDAL B, ABIE S M, BJARNADOTTIR S, et al. Detectability of the degree of freeze damage in meat analytic-tool depends on selection[J]. Meat Science,2019,152:8−19. doi: 10.1016/j.meatsci.2019.02.002
[16] ZHANG B, MAO J L, YAO H, et al. Label-free based proteomics analysis of protein changes in frozen whiteleg shrimp (Litopenaeus vannamei) pre-soaked with sodium trimetaphosphate[J]. Food Research International,2020,137:1−8.
[17] GAO X, XIE Y R, YIN T, et al. Effect of high intensity ultrasound on gelation properties of silver carp surimi with different salt contents[J]. Ultrasonics Sonochemistry,2021,70:1−8.
[18] ZHOU Y G, LIU J J H, KANG Y, et al. Effects of acid and alkaline treatments on physicochemical and rheological properties of tilapia surimi prepared by pH shift method during cold storage[J]. Food Research International,2021,145:1−13.
[19] WU X, SUN W, YANG H, et al. Effect of repeated freeze-thaw cycles on quality properties of frozen surimis of grass carp and common carp[J]. Food Science,2012,33(20):323−327.
[20] WANG L, ZHANG M, BHANDARI B, et al. Investigation on fish surimi gel as promising food material for 3D printing[J]. Journal of Food Engineering,2018,220:101−108. doi: 10.1016/j.jfoodeng.2017.02.029
[21] LIU H, CHEN X, SONG L, et al. Effect of different thawing methods on freshness and quality of Scomber japonicus[J]. Food Science,2016,37(10):259−265.
[22] YANG Z, WANG W, WANG H Y, et al. Effects of a highly resistant rice starch and pre-incubation temperatures on the physicochemical properties of surimi gel from grass carp (Ctenopharyn odon idellus)[J]. Food Chemistry,2014,145:212−219. doi: 10.1016/j.foodchem.2013.08.040
[23] CAO H W, FAN D M, JIAO X D, et al. Effects of microwave combined with conduction heating on surimi quality and morphology[J]. Journal of Food Engineering,2018,228:1−11. doi: 10.1016/j.jfoodeng.2018.01.021
[24] LIU Y, TIAN M, LI X, et al. Effects of silver carp hydrolysate on quality and in vitro protein digestibility of freeze-thawed surimi products[J]. Food and Machinery,2018,34(10):118−123.
[25] YU Y, YI S, XU Y, et al. Gel properties of mixed surimi from silver carp and Nemipterus virgatus[J]. Food Science,2016,37(5):17−22.
[26] SWEENEY M, CAMPBELL L, HANSON J, et al. Characterizing the feasibility of processing wet granular materials to improve rheology for 3D printing[J]. Journal of Materials Science,2017,52(22):13040−13053. doi: 10.1007/s10853-017-1404-z
[27] CHEN H Z, ZHANG M, YANG C H. Comparative analysis of 3D printability and rheological properties of surimi gels via LF-NMR and dielectric characteristics[J]. Journal of Food Engineering,2021,292:1−9.
[28] TASKAYA L, CHEN Y C, JACZYNSKI J. Color improvement by titanium dioxide and its effect on gelation and texture of proteins recovered from whole fish using isoelectric solubilization/precipitation[J]. LWT-Food Science and Technology,2010,43(3):401−408. doi: 10.1016/j.lwt.2009.08.021
[29] ZHANG T, XUE Y, LI Z, et al. Effects of deacetylation of konjac glucomannan on Alaska Pollock surimi gels subjected to high-temperature (120 ℃) treatment[J]. Food Hydrocolloids,2015,43:125−131. doi: 10.1016/j.foodhyd.2014.05.008
[30] ZHUANG X, ZHANG W, LIU R, et al. Improved gel functionality of myofibrillar proteins incorporation with sugarcane dietary fiber[J]. Food Research International,2017,100:586−594. doi: 10.1016/j.foodres.2017.07.063
[31] PEREZ-MATEOS M, SOLAS T, MONTERO P. Carrageenans and alginate effects on properties of combined pressure and temperature in fish mince gels[J]. Food Hydrocolloids,2002,16(3):225−233. doi: 10.1016/S0268-005X(01)00086-8
[32] YUE K, HUANG Y, ZHANG Y, et al. The changes of rheological properties and gel structures of sea bass surimi during fro-zen storage[J]. Science & Technology of Food Industry,2016,37(8):330−334.
[33] KAEWUDOM P, BENJAKUL S, KIJROONGROJANA K. Properties of surimi gel as influenced by fish gelatin and microbial transglutaminase[J]. Food Bioscience,2013,1:39−47. doi: 10.1016/j.fbio.2013.03.001
[34] YANG R, XU A Q, CHEN Y T, et al. Effect of laver powder on textual, rheological properties and water distribution of squid (Dosidicus gigas) surimi gel[J]. Journal of Texture Studies,2020,51(6):968−978. doi: 10.1111/jtxs.12556
[35] LI F F, WANG B, KONG B H, et al. Decreased gelling properties of protein in mirror carp (Cyprinus carpio) are due to protein aggregation and structure deterioration when subjected to freeze-thaw cycles[J]. Food Hydrocolloids,2019,97:1−8.
[36] CHEN X, SHI X D, CAI X X, et al. Ice-binding proteins: A remarkable ice crystal regulator for frozen foods[J]. Critical Reviews in Food Science and Nutrition,2020:1−14.
[37] MULOT V, BENKHELIFA H, PATHIER D, et al. Measurement of food dehydration during freezing in mechanical and cryogenic freezing conditions[J]. International Journal of Refrigeration-Revue Internationale Du Froid,2019,103:329−338. doi: 10.1016/j.ijrefrig.2019.02.032
[38] LI D P, JIA S L, ZHANG L T, et al. Effect of using a high voltage electrostatic field on microbial communities, degradation of adenosine triphosphate, and water loss when thawing lightly-salted, frozen common carp (Cyprinus carpio)[J]. Journal of Food Engineering,2017,212:226−233. doi: 10.1016/j.jfoodeng.2017.06.003
[39] CHEN Z B, ZHU Z W, SUN D W. Effects of air content in radish tissue pores on freezing efficiency during ultrasound assisted immersion freezing[J]. Modern Food Science and Technology,2017,33(7):172−179.
[40] YI S M, LI Q, QIAO C P, et al. Myofibrillar protein conformation enhance gel properties of mixed surimi gels with Nemipterus virgatus and Hypophthalmichthys molitrix[J]. Food Hydrocolloids,2020,106:1−11.
[41] YANG W G, ZHANG W, WANG X, et al. Effect of salt solution rinse on properties of hairtail surimi gel by low-field nuclear magnetic resonance[J]. Transactions of the Chinese Society of Agricultural Engineering,2016,32(7):263−269.
[42] ZHANG B, ZHAO J L, CHEN S J, et al. Influence of trehalose and alginate oligosaccharides on ice crystal growth and recrystallization in whiteleg shrimp (Litopenaeus vannamei) during frozen storage with temperature fluctuations[J]. International Journal of Refrigeration-Revue Internationale Du Froid,2019,99:176−185. doi: 10.1016/j.ijrefrig.2018.11.015
[43] LEYGONIE C, BRITZ T J, HOFFMAN L C. Impact of freezing and thawing on the quality of meat: Review[J]. Meat Science,2012,91(2):93−98. doi: 10.1016/j.meatsci.2012.01.013
[44] SUN W Q, WU X, YANG H, et al. Changes of silver carp surimi quality under different low temperature storage conditions[J]. Hubei Agricultural Sciences,2013(16):3959−3962.
[45] WALAYAT N, XIONG Z, XIONG H, et al. The effectiveness of egg white protein and β-cyclodextrin during frozen storage: Functional, rheological and structural changes in the myofibrillar proteins of Culter alburnus[J]. Food Hydrocolloids,2020,105:1−11.
[46] WANG B, LI F F, PAN N, et al. Effect of ice structuring protein on the quality of quick-frozen patties subjected to multiple freeze-thaw cycles[J]. Meat Science,2021,172:1−9.
[47] CUI X Y, ZHAO X Q, LIU C Y, et al. Research progress on the effect of ice crystal morphology repeated freeze-thaw meat system[J/OL]. Food Science: 1−13[2021-08-27]. http://kns.cnki.net/kcms/detail/11.2206.TS.20210816.1513.080.html.
[48] CHEN Q M, ZHANG Y C, GUO Y H, et al. Non-destructive prediction of texture of frozen/thaw raw beef by Raman spectroscopy[J]. Journal of Food Engineering,2020,266:1−7.
[49] WANG K Y, WANG R H, LI Y J, et al. Effects of α-tocopherol and vacuum packaging on the quality of sturgeon surimi incorporated with chicken meat during frozen storage[J]. Food Science,2021,42(11):195−204.
[50] CAI L, DAI Y, CAO M. The effects of magnetic nanoparticles combined with microwave or far infrared thawing on the freshness and safety of red seabream (Pagrus major) fillets[J]. LWT,2020,128:1−9.
-
期刊类型引用(7)
1. 杭瑜瑜,张铁涛,齐丹,胡亚芹. 聚葡萄糖对罗非鱼鱼糜冻融稳定性的影响及其机制. 中国食品添加剂. 2024(10): 131-138 .  百度学术
百度学术
2. 苏婕,李佳艺,刘永乐,周劲松,黄轶群,李向红,王发祥. 冻藏和冻融条件对鲢鱼不漂洗鱼糜中α-二羰基化合物形成的影响. 食品与机械. 2024(11): 121-127 .  百度学术
百度学术
3. 汪少芸,李晓贞,陈旭,王一潇,杜明,吴金鸿,刘永乐,黄建联,蔡茜茜. 鱼源抗冻多肽对鱼糜肌原纤维蛋白的冻融保护作用. 广东海洋大学学报. 2023(01): 78-86 .  百度学术
百度学术
4. 高华倩,张瑜,吴金鸿,汪少芸,黄轶群,王发祥,刘永乐,李向红. 油脂添加对冻融鱼糜凝胶结构和大变形行为的影响. 食品科学. 2023(10): 22-29 .  百度学术
百度学术
5. 李佳艺,苏婕,王发祥,李向红,俞健,刘永乐. 未漂洗鱼糜冻藏过程中晚期糖化终末产物形成及机制. 中国食品学报. 2023(09): 233-241 .  百度学术
百度学术
6. 江文婷,陈旭,蔡茜茜,杨傅佳,黄丹,黄建联,汪少芸. 基于分子对接技术研究鱼源抗冻多肽与鱼肌球蛋白的相互作用. 食品工业科技. 2022(20): 29-38 .  本站查看
本站查看
7. 陈旭,余璐涵,蔡茜茜,吴金鸿,刘永乐,黄建联,汪少芸. 低温冷链贮藏对鱼糜凝胶化学作用力和肌原纤维蛋白结构及功能特性的影响. 食品科学. 2022(23): 194-201 .  百度学术
百度学术
其他类型引用(5)











 下载:
下载:

 下载:
下载:



