Study on the Potential Mechanism of Synsepalum dulcificum Interfering with Tumor Immunity
-
摘要: 目的:研究神秘果干预肿瘤免疫药效学物质基础依据以及可能机制。方法:利用液相-质谱分析明确神秘果的成分,之后利用网络药理学以及生物信息学方法系统研究了神秘果作用于肿瘤免疫的作用靶点,同时明确神秘果药食同源的可能性。结果:液相-质谱实验得到神秘果的可能含有成分360种,与肿瘤免疫数据库对比后得到可干预肿瘤免疫相关成分42个,干预相关靶点55个,筛选后得到靶点网络中关键靶点为TSHR、TP53、MAPK1、HIF1A、CA9。进一步收集文献中实验信息及分子对接实验验证神秘果的不同成分可影响选取靶点进而干预肿瘤免疫。结论:通过以上实验发现神秘果成分与肿瘤免疫相关靶点可能具有相互作用,为神秘果直接作用肿瘤研究铺垫基础。Abstract: Objective: To study the material basis and possible mechanism of the pharmacodynamics of Synsepalum dulcificum's intervention in tumor immunity. Methods: The study used liquid phase-mass spectrometry analysis to clarify the composition of Synsepalum dulcificum, and then used the network pharmacology and bioinformatics methods to systematically study the specific impact of Synsepalum dulcificum on tumor immunity. At the same time, it was clear that Synsepalum dulcificum medicine and food were homologous.Results: The liquid-mass spectrometry experiment revealed that Synsepalum dulcificum may contain 360 components. After comparing with the tumor immunity database, 42 components related to tumor immunity and 55 related targets were obtained. After screening, key targets in the target network were obtained. These were TSHR, TP53, MAPK1, HIF1A, CA9. Further collection of experimental information in the literature and molecular docking experiments verified that the different components of Synsepalum dulcificum could affect the selection of targets and interfere with tumor immunity. Conclusion: Through the above experiments, it is found that the components of Synsepalum dulcificum may interact with tumor immune-related targets, paving the way for the research on the direct effect of Synsepalum dulcificum on tumors.
-
癌症是全球第二大死亡原因[1]。研究认为癌症的起始与发展不仅与遗传改变相关。不合理饮食、久坐的生活方式和异常肥胖也被认为是肿瘤发生危险因素[2]。目前,尚未发现癌症的明确成因以及显著有效治疗手段,一线治疗方法仍为手术及放射性疗法,但对人体有严重副作用,比如身体衰弱、免疫功能下降及神经毒性等副作用。机体免疫功能与肿瘤发生发展密切相关,人体可以通过不同的免疫途径启动抗肿瘤免疫应答,从而达到抗肿瘤功能。肿瘤免疫治疗相比其他治疗方式具有治疗范围广,临床预后好,可适用于各种癌症类型等优点。某些天然活性物质有提高免疫功能,具有预防肿瘤的发生,减少转移和复发等特点[3]。因此探究具有抗肿瘤活性且低毒副作用的天然活性物质迫在眉睫。
神秘果(Synsepalum dulcificum)是热带西非的土产水果,因果肉可以将酸味转变为甜味的特点被称神秘果。神秘果因其成分丰富具有巨大潜在保健价值,目前研究发现其果实、种子及叶等部位的提取物均有不同药理活性,比如降血脂、抗尿酸、抑菌、抗惊厥、抗疲劳等活性[4]。具有诸多生物活性的原因是其果肉中富含酚类化合物(15.8%)和类黄酮(11.9%),果皮含有更高含量的酚类化合物(36.7%)和类黄酮(51.9%)[5]。一般认为酚类和类黄酮含量与抗氧化活性及抗肿瘤之间存在密切关系,高含量的酚类及类黄酮使得神秘果具有干预肿瘤的重要潜在能力[6]。已经报道神秘果提取物可以在体外减少某些恶性肿瘤的增殖,如减少人黑色素瘤、结直肠癌细胞系的增殖和抗人慢性髓系白血病细胞(K562)活性[7-8]。即使神秘果可以干预肿瘤发生发展,但是对于这种果实的抗肿瘤的机制研究仍鲜有报道。
本研究通过液相-质谱联用技术及网络药理学研究手段探究神秘果在肿瘤免疫方面可能发挥的作用,为神秘果的开发利用铺垫基础,同样为抗肿瘤食药开发提供参考。
1. 材料与方法
1.1 材料及仪器
神秘果 购自广东江门,经内蒙古医科大学研究员鉴定为山揽科神秘果属植物,−80 ℃保存;甲醇、乙腈、甲酸 色谱纯;其他试剂 均为分析纯,天津大茂化学试剂厂。
Triple TOF 5600/6600质谱仪 美国AB SCIEX;Agilent 1290 Infinity LC超高压液相色谱仪 美国Agilent;Eppendorf5430R低温高速离心机 德国Eppendorf;色谱柱:ACQUITY UPLC BEH Amide (1.7 µm,2.1 mm×100 mm column)、ACQUITY UPLC HSS T3 (1.8 µm,2.1 mm×100 mm column) 美国Waters。
1.2 实验方法
1.2.1 液相色谱-质谱方法获得成分
1.2.1.1 样品预处理
取神秘果样品适量,去核,冻干后成粉供精密称取,−80 ℃保存。
上机样本制备:−80 ℃取出样本,称量80 mg,加入200 μL水匀浆,涡旋60 s,加入800 μL甲醇乙腈溶液(1:1, v/v),涡旋60 s,低温超声30 min,2次,−20 ℃放置1 h沉淀蛋白,14000 ×g,4 ℃离心20 min,取上清冷冻干燥,−80 ℃保存,上机样品平行两次实验。
1.2.1.2 液相色谱-质谱分析
液相色谱条件:样品采用Agilent 1290 Infinity LC超高效液相色谱系统(UHPLC)HILIC色谱柱进行分离;柱温25 ℃;流速0.3 mL/min;流动相组成A:水+25 mol/L乙酸铵+25 mmol/L氨水,B:乙腈;梯度洗脱程序如下:0~0.5 min,95% B;0.5~7 min,B从95%线性变化至65%;7~8 min,B从65%线性变化至40%;8~9 min,B维持在40%;9~9.1 min,B从40%线性变化至95%;9.1~12 min,B维持在95%;整个分析过程中样品置于4 ℃自动进样器中。为避免仪器检测信号波动而造成的影响,采用随机顺序进行样本的连续分析。
质谱条件:分别采用电喷雾电离(ESI)正离子和负离子模式进行检测。样品经UHPLC分离后用Agilent 6550质谱仪进行质谱分析。ESI源条件如下:气体温度:250 ℃,干燥气体:16 L/min,雾化气压力:20 Psig,壳气温度:400 ℃,壳气流量:12 L/min,Vcap:3000 V,喷嘴电压:0 V,Fragment:175 V,质量范围:50~1200,采集速率:4 Hz,循环时间:250 ms。样本检测完毕后,采用AB Triple TOF 6600质谱仪对代谢物进行鉴定,采集样品的一级、二级谱图。ESI源条件如下:辅助气压1(Gas1):40,辅助气压2(Gas2):80,气帘气体:30,离子源温度:650 ℃,喷雾电压±5000 V(正负两种模式);二级质谱采用信息关联扫描获得,并且采用高灵敏度模式,定性离子对去簇电压:±60 V(正负两种模式),碰撞能量:(35±15)eV,IDA设置:不包括同位素4 Da,周期监测的候选离子:10。数据采集是按质量范围进行分段:50~300、290~600、590~900、890~1200,从而扩大二级谱图的采集率,每个方法每段采集四个重复。所采集获得的数据,分别使用MetDDA和LipDDA方法,进行代谢物的结构鉴定。
1.3 神秘果干预肿瘤免疫相关成分
为明确神秘果影响肿瘤免疫的物质基础,首先将液相-质谱技术获得的化合物在PubChem Compound (https://www.ncbi.nlm.nih.gov/pccompound)中搜索转化对应唯一标识符Smiles,借助中药肿瘤免疫数据库TCMIO[9] (http://tcmio.xielab.net/)寻找神秘果成分中含有干预肿瘤免疫相关化合物,明确具体免疫相关化学成分及收录对应靶点。
1.4 靶点生物学功能信息
为更好地了解神秘果中相关免疫化合物靶点的生物学功能,将整理得到的化合物靶点信息利用R语言包clusterProfiler[10]富集分析,经过合理统计学阈值明确对应的生物学功能,包括生物过程(biologicalprocess,BP)、分子功能(molecular function,MF)和细胞成分(cellular component,CC)。
1.5 化合物-靶点关系分析
根据成分与相关靶点的关系可构建神秘果作用肿瘤免疫的生物关系网络拓扑图,通常在生物关系网络中,居于关键位置的基因起到的调控的作用更加重要,借助Cytoscape软件构建化合物-靶点互作网络,并从网络中选取核心靶点进一步分析。
1.6 核心靶点在不同肿瘤的表达差异
挑选出调控网络的关键基因后,为明确目标靶点在不同肿瘤中的表达差异,借助GEPIA数据库[11] (http://gepia.cancer-pku.cn/)记录的33种肿瘤中与正常样本数据进行对比,统计条件为表达值经过对数处理,差异统计方法采用方差分析,|Log2FC|>1,P<0.01,对比数据包括TCGA及GTEx数据。
1.7 化合物影响通路
进一步了解神秘果影响肿瘤免疫的信号通路,通过CTD数据库[12](http://ctdbase.org/)富集相关化合物的信号通路,以P<0.01进行筛选。
1.8 神秘果中成分与靶点的相互作用
前面工作得到在肿瘤中有明显表达差异的基因,为明确神秘果相关成分对得到的靶点具体相互作用,通过文献及分子对接技术进行验证。神秘果含有化合物可能直接参与靶点基因的表达过程以及产物的翻译修饰途径,还可能与蛋白质直接相互作用进而影响蛋白质功能,借助TCMSP数据库收录的化学物质结构信息,下载神秘果相关化学成分结构并保存为MOL2格式,使用Discovery Studio(DS)软件进行化学成分配体的处理。使用RCSB数据库(http://www1.rcsb.org/)下载蛋白质结构文件。用DS软件进行蛋白预处理,包括清除原配体,去水加氢,施加力场等,LibDock执行分子对接计算并分析所得结果。
2. 结果与分析
2.1 神秘果肉成分的鉴定
平行两组样品经UHPLC-Q-TOF MS分析后得到的典型TIC图谱,如图1所示。通过保留时间(tR),裂解规律以及数据库的搜索得到样品共有成分为181种,数据未给出。
2.2 神秘果果肉干预肿瘤免疫相关成分信息
经过搜索对比,在TCMIO数据库中得到神秘果可干预肿瘤免疫相关成分42个,同时整理其干预相关靶点55个。化合物详细信息表1。
表 1 神秘果干预肿瘤免疫相关成分信息Table 1. Information on relevant components of Synsepalum dulcificum 's intervention in tumor immunity序号 化合物 结构 Smiles Inchikey CAS 1 Naringenin 
[C@H]1(CC(=O)c2c(cc(cc2O1)O)O)c1ccc(cc1)O FTVWIRXFELQLPI-ZDUSSCGKSA-N 480-41-1 2 1,3,5-Benzenetriol 
c1(cc(cc(c1)O)O)O QCDYQQDYXPDABM-UHFFFAOYSA-N 108-73-6 3 Anthranilic acid (Vitamin L1) 
c1(c(cccc1)N)C(=O)O RWZYAGGXGHYGMB-UHFFFAOYSA-N 118-92-3 4 Choline 
C(CO)[N](C)(C)C GDPPXFUBIJJIKR-UHFFFAOYSA-N 62-49-7 5 Quercetin 
c1(cc(c2c(c1)oc(c(c2=O)O)c1ccc(c(c1)O)O)O)O REFJWTPEDVJJIY-UHFFFAOYSA-N 117-39-5 6 Morin 
c12c(oc(c(c1=O)O)c1c(cc(cc1)O)O)cc(cc2O)O YXOLAZRVSSWPPT-UHFFFAOYSA-N 480-16-0 7 Phenylacetic acid 
c1cccc(c1)CC(=O)O WLJVXDMOQOGPHL-UHFFFAOYSA-N 103-82-2 8 Palmitic acid 
C(=O)(CCCCCCCCCCCCCCC)O IPCSVZSSVZVIGE-UHFFFAOYSA-N 57-10-3 9 Stearic acid 
C(CCCCCCCCCCC)CCCCCC(=O)O QIQXTHQIDYTFRH-UHFFFAOYSA-N 57-11-4 10 Guanosine 
c12n([C@H]3O[C@@H]([C@H]([C@H]
3O)O)CO)cnc1c(=O)[nH]c(n2)NNYHBQMYGNKIUIF-BDXYJKHTSA-N 118-00-3 11 Uracil 
[nH]1c(=O)[nH]c(=O)cc1 ISAKRJDGNUQOIC-UHFFFAOYSA-N 66-22-8 12 Dihydrouracil 
N1C(=O)CCNC1=O OIVLITBTBDPEFK-UHFFFAOYSA-N 504-07-4 13 Dopamine 
Oc1cc(CCN)ccc1O VYFYYTLLBUKUHU-UHFFFAOYSA-N 51-61-6 14 Heptadecanoic acid 
OC(=O)CCCCCCCCCCCCCCCC KEMQGTRYUADPNZ-UHFFFAOYSA-N 506-12-7 15 Tyramine 
Oc1ccc(CCN)cc1 DZGWFCGJZKJUFP-UHFFFAOYSA-N 51-67-2 16 Procyanidin B2 
c1(cc(c2c(c1)O[C@@H]([C@@H]([C@H]2c1c
(cc(c2c1O[C@@H]([C@@H](C2)O)c1cc
(c(cc1)O)O)O)O)O)c1cc(c(cc1)O)O)O)OXFZJEEAOWLFHDH-NFJBMHMQSA-N 29106-49-8 17 Pyridoxine 
Oc1c(c(CO)cnc1C)CO LXNHXLLTXMVWPM-UHFFFAOYSA-N 65-23-6 18 Adenine 
[nH]1cnc2c(N)ncnc12 GFFGJBXGBJISGV-UHFFFAOYSA-N 73-24-5 19 Gentisic acid 
c1(c(cc(cc1)O)C(=O)O)O WXTMDXOMEHJXQO-UHFFFAOYSA-N 490-79-9 20 4-Hydroxycinnamic acid 
C(=O)(/C=C/c1ccc(cc1)O)O NGSWKAQJJWESNS-ZZXKWVIFSA-N 501-98-4 21 Sucrose 
OC[C@H]1O[C@H](O[C@@]2(CO)[C@@H]
(O)[C@H](O)[C@@H](CO)O2)[C@H](O)
[C@@H](O)[C@@H]1OCZMRCDWAGMRECN-UGDNZRGBSA-N 57-50-1 22 myo-Inositol 
C1(C(C(C(C(C1O)O)O)O)O)O CDAISMWEOUEBRE-UHFFFAOYSA-N 87-89-8 23 Myristic acid 
C(=O)(CCCCCCCCCCCCC)O TUNFSRHWOTWDNC-UHFFFAOYSA-N 544-63-8 24 Amygdalin 
c1(ccccc1)[C@H](C#N)O[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@H](O1)CO[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@H](O1)CO XUCIJNAGGSZNQT-JHSLDZJXSA-N 29883-15-6 25 Myricetin 
c1(c(cc(cc1O)c1c(O)c(=O)c2c(o1)cc(cc2O)O)O)O IKMDFBPHZNJCSN-UHFFFAOYSA-N 529-44-2 26 Maslinic Acid 
[C@@]12([C@]3(C(=CC[C@@H]1[C@@]1
([C@@H](CC2)C([C@H]([C@@H](C1)O)O)(C)
C)C)[C@H]1[C@@](CC3)(CCC(C1)(C)C)
C(=O)O)C)CMDZKJHQSJHYOHJ-LLICELPBSA-N 4373-41-5 27 N6,N6,N6-Trimethyl-L-lysine 
C[N+](C)(C)CCCC[C@@H](C(=O)[O-])N MXNRLFUSFKVQSK-QMMMGPOBSA-N 19253-88-4 28 Nicotinamide 
c1(cccnc1)C(=O)N DFPAKSUCGFBDDF-UHFFFAOYSA-N 98-92-0 29 Betaine 
OC(=O)C[N](C)(C)C GMTCLSZGPOBNLA-UHFFFAOYSA-N 107-43-7 30 Quercitrin 
c1(c(c(=O)c2c(o1)cc(cc2O)O)O[C@H]1[C@@H]([C@@H]
([C@H]([C@@H](O1)C)O)O)O)
c1cc(c(cc1)O)OOXGUCUVFOIWWQJ-HQBVPOQASA-N 522-12-3 31 L-Asparagine 
C(=O)([C@@H](N)CC(=O)N)O DCXYFEDJOCDNAF-REOHCLBHSA-N 32640-57-6 32 L-Tryptophan 
c12c(C[C@@H](C(=O)O)N)c[nH]c1cccc2 QIVBCDIJIAJPQS-VIFPVBQESA-N 73-22-3 33 L-Threonine 
O[C@@H]([C@H](N)C(=O)O)C AYFVYJQAPQTCCC-GBXIJSLDSA-N 72-19-5 34 Cytosine 
n1c(cc[nH]c1=O)N OPTASPLRGRRNAP-UHFFFAOYSA-N 71-30-7 35 L-Aspartate 
C(=O)([C@@H](N)CC(=O)O)O CKLJMWTZIZZHCS-REOHCLBHSA-N 56-84-8 36 Indole 
[nH]1c2c(cc1)cccc2 SIKJAQJRHWYJAI-UHFFFAOYSA-N 120-72-9 37 Astragalin 
c12c(oc(c(c1=O)O[C@@H]1O[C@@H]([C@H]([C@@H]([C@H]1O)O)O)CO)c1ccc(cc1)O)cc(cc2O)O JPUKWEQWGBDDQB-QSOFNFLRSA-N 480-10-4 38 2-Hydroxyadenine 
c12c(c(nc(n1)O)N)[nH]cn2 DRAVOWXCEBXPTN-UHFFFAOYSA-N 3373-53-3 39 Glutathione 
C(CC(=O)N[C@@H](CS)C(=O)NCC(=O)O)[C@@H](C(=O)O)N RWSXRVCMGQZWBV-WDSKDSINSA-N 70-18-8 40 trans-Ferulic acid 
c1(cc(c(cc1)O)OC)/C=C/C(=O)O KSEBMYQBYZTDHS-HWKANZROSA-N 1135-24-6 41 Quercetin 3'-methyl ether 
c1(c(OC)c(=O)c2c(O)cc(O)cc2o1)c1cc(O)c(O)cc1 WEPBGSIAWZTEJR-UHFFFAOYSA-N 1486-70-0 42 4-Guanidinobutyric acid 
C(=O)(CCCNC(=N)N)O TUHVEAJXIMEOSA-UHFFFAOYSA-N 463-00-3 2.3 神秘果影响肿瘤免疫靶点生物过程、分子功能、细胞成分结果
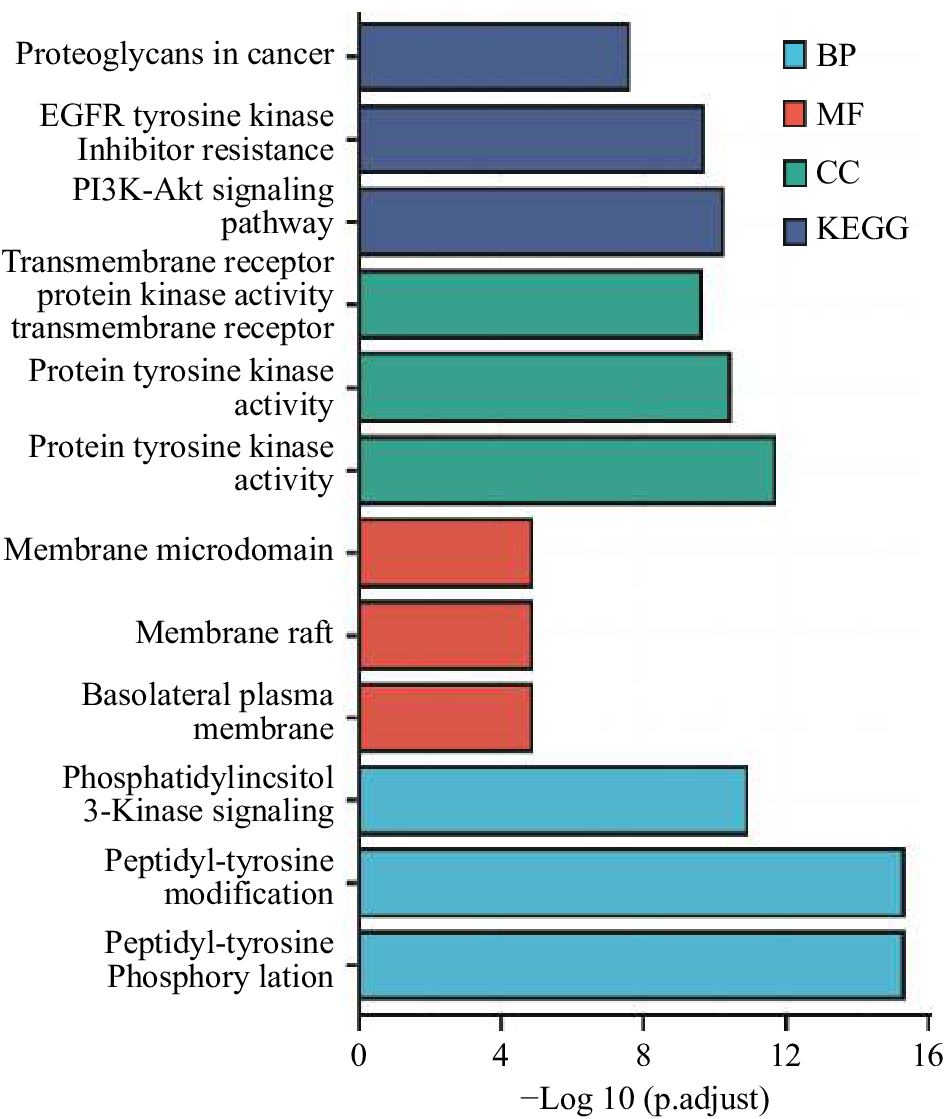
整理分析得到神秘果影响肿瘤免疫相关靶点55个,经过GO注释分析及KEGG通路分析,筛选得到P值及FDR值均小于0.01,如图2所示,各项P值前三信息,主要结果生物过程为肽酰酪氨酸磷酸化、肽酰酪氨酸修饰、磷脂酰肌醇3-激酶信号,分子功能为蛋白酪氨酸激酶活性、跨膜受体蛋白酪氨酸激酶活性、跨膜受体蛋白激酶活性,细胞定位为基底外侧质膜、膜筏、膜微区,KEGG为PI3K-AKT信号通路、EGFR酪氨酸激酶抑制剂耐药、肿瘤中蛋白聚糖相关途径。
2.4 筛选核心化合物及靶点
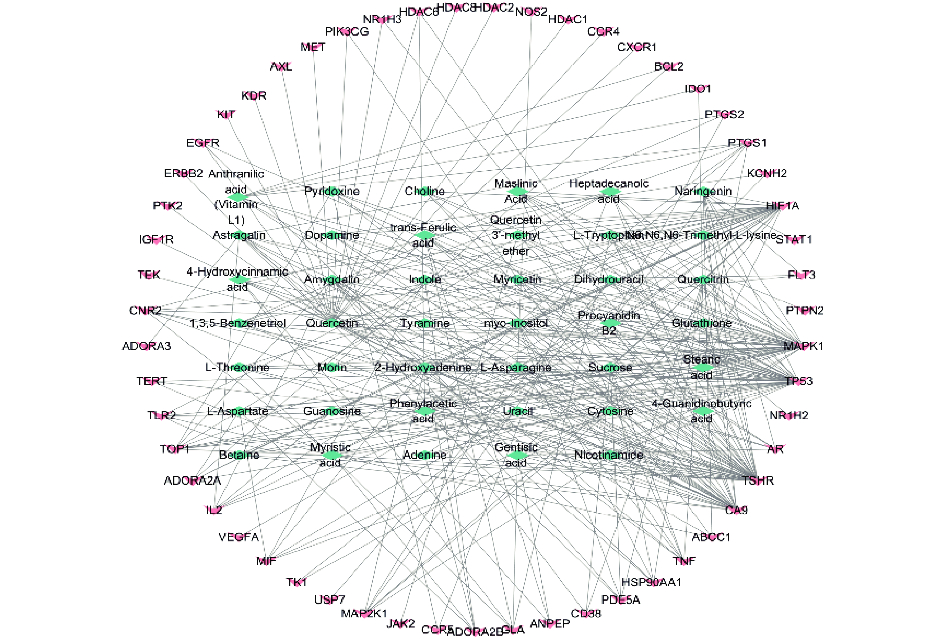
将得到的化合物及对应靶点相互关系导入Ctyoscape中,接借助Networkanalyzer计算网络拓扑参数,Degree值前五的基因为核心靶点分别为:TSHR(Degree=42)、TP53(Degree=39)、MAPK1(Degree=39)、HIF1A(Degree=30)、CA9(Degree=28)继续研究,见图3。
2.5 核心靶点的临床意义
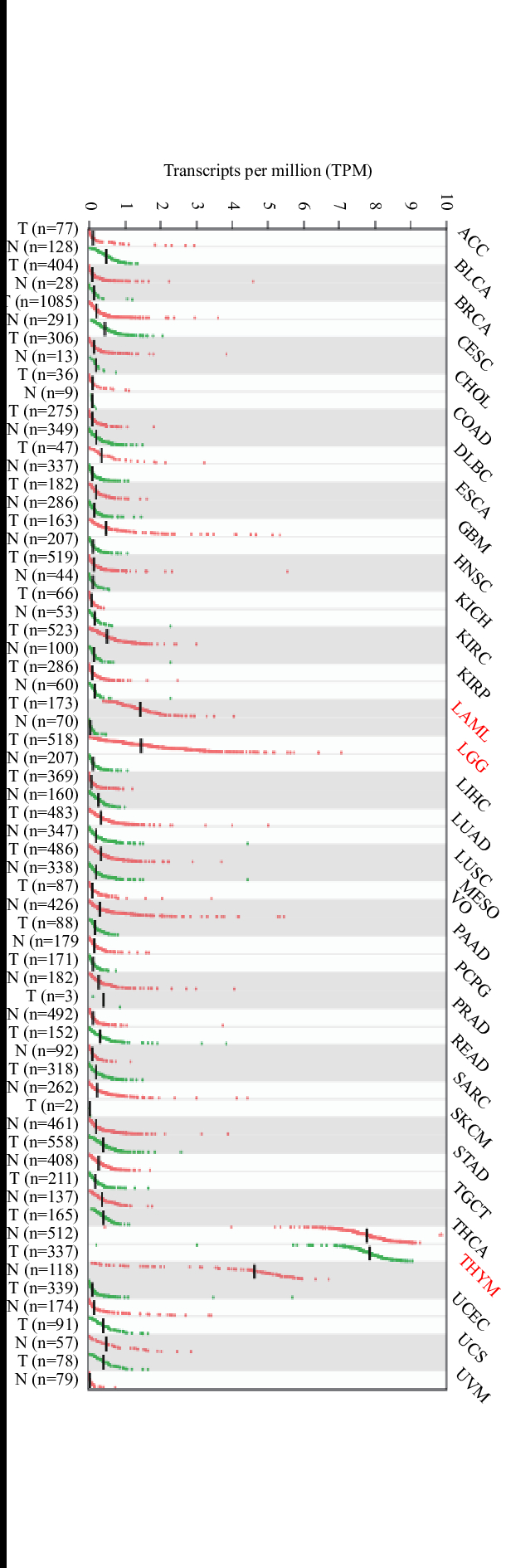
筛选得到核心靶点,也被认为神秘果影响肿瘤免疫的主要作用靶点,利用Gepia内置数据得到核心基因在不同肿瘤中差异情况不一,见图4~图8。CA9在ACC、BLCA、CESC、COAD、ESCA、GBM、HNSC、KIRC、LUAD、LUSC、OV、PAAD、READ、UCEC、UCS中高表达,而在LAML、SKCM、STAD、TGCT中低表达,HIF1A在ESCA、GBM、HNSC、LGG、PAAD、STAD高表达,MAPK1在LGG、PAAD、STAD中高表达,在ACC、DLBC、LAML低表达,TP53在COAD、DLBC、GBM、LAML、LGG、LUSC、OV、PAAD、READ、STAD、TGCT、THYM、UCEC均高表达,TSHR在LAML、LGG、THYM 高表达。发现即使同一靶点在不同肿瘤的表达趋势也不一致,证明肿瘤免疫的复杂性和多面性。
2.6 相关化合物影响的信号通路
明确神秘果相关化合物影响体内的信号通路,筛选后结果得到1479条信号通路,着重分析P值排名前十信号通路,可见神秘果相关成分影响的信号通路为免疫系统、先天免疫系统等途径(表2)。
表 2 神秘果影响肿瘤免疫相关化合物影响信号通路Table 2. Synsepalum dulcificum affects tumor immunity-related compounds affect signal pathways序号 通路 通路 ID P-value 富集基因数 基因组占比 1 Immune System REACT:R-HSA-168256 0 1391 2118/44020 genes: 4.81% 2 Innate Immune System REACT:R-HSA-168249 0 858 1298/44020 genes: 2.95% 3 Metabolic pathways KEGG:hsa01100 0 890 1270/44020 genes: 2.89% 4 Metabolism REACT:R-HSA-1430728 0 1536 2172/44020 genes: 4.93% 5 Metabolism of proteins REACT:R-HSA-392499 0 984 1623/44020 genes: 3.69% 6 Signal Transduction REACT:R-HSA-162582 0 1550 2588/44020 genes: 5.88% 7 Gene Expression REACT:R-HSA-74160 2.16e-310 969 1831/44020 genes: 4.16% 8 Metabolism of lipids and lipoproteins REACT:R-HSA-556833 6.33e -297 595 814/44020 genes: 1.85% 9 Developmental Biology REACT:R-HSA-1266738 1.94e -270 672 1076/44020 genes: 2.44% 10 Cytokine Signaling in Immune system REACT:R-HSA-1280215 9.13e -267 547 760/44020 genes: 1.73% 2.7 化合物对核心基因的影响
通过文献检索,对于上文筛选的神秘果影响肿瘤免疫的42种化合物以及筛选的关键化合物进行搜索,得到化合物与核心基因的相互关系。可见神秘果众多化合物可影响多个靶点,佐证文献见表3。
表 3 神秘果化合物对核心基因的影响Table 3. Effects of Synsepalum dulcificum compounds on hub genes核心基因 化合物 影响趋势 支持文献 实验物种 TP53 Choline Choline results in decreased expression of TP53 mRNA [13] Rattus norvegicus Dopamine [Dopamine results in increased expression of HIF1A protein] which results in increased expression of and results in increased phosphorylation of TP53 protein [14] Homo sapiens Glutathione [Buthionine Sulfoximine results in decreased abundance of Glutathione] inhibits the reaction [Benzo(a)pyrene results in increased activity of TP53 protein] [15] Homo sapiens Glutathione Dithiothreitol inhibits the reaction [Glutathione results in increased glutathionylation of TP53 protein] [16] Homo sapiens Glutathione Glutathione inhibits the reaction [[CD40LG protein co-treated with IL4 protein] results in increased expression of TP53 protein] [17] Homo sapiens morin morin inhibits the reaction [Ifosfamide results in increased expression of TP53 protein] [18] Rattus norvegicus morin morin results in increased expression of TP53 mRNA [19] Homo sapiens myricetin myricetin analog results in increased expression of TP53 protein [20] Homo sapiens myricitrin myricitrin inhibits the reaction [Hydrogen Peroxide results in increased expression of TP53 protein] [21] Homo sapiens naringenin naringenin results in increased expression of TP53 mRNA [22] Homo sapiens Quercetin Quercetin results in decreased expression of TP53 protein [23] Homo sapiens TSHR Quercetin Quercetin results in decreased expression of TSHR mRNA [24] Rattus norvegicus MAPK1 Dopamine Dopamine results in increased phosphorylation of and results in increased activity of MAPK1 protein [25] Homo sapiens Glutathione Glutathione inhibits the reaction [Hydrogen Peroxide results in increased phosphorylation of MAPK1 protein] [26] Canis lupus familiaris morin morin inhibits the reaction [Excitatory Amino Acid Agonists results in increased phosphorylation of MAPK1 protein] [27] Rattus norvegicus myricetin myricetin results in decreased activity of MAPK1 protein [28] Mus musculus naringenin naringenin results in decreased activity of MAPK1 protein [29] Homo sapiens naringenin naringenin results in decreased phosphorylation of MAPK1 protein [30] Homo sapiens Palmitic Acid Palmitic Acid results in increased phosphorylation of MAPK1 protein [31] Homo sapiens Sucrose Sucrose results in decreased phosphorylation of and results in decreased activity of MAPK1 protein [32] Mus musculus Tyramine Tyramine results in increased activity of MAPK1 protein [33] Oryctolagus cuniculus HIF1A Choline [Methionine deficiency co-treated with Choline deficiency co-treated with Folic Acid deficiency] results in increased methylation of HIF1A gene [34] Mus musculus Dopamine Dopamine results in decreased expression of HIF1A protein [35] Rattus norvegicus Dopamine Dopamine results in increased expression of HIF1A protein [14] Homo sapiens Glutathione Glutathione inhibits the reaction [15-deoxy-delta(12,14)-prostaglandin J2 results in increased expression of HIF1A protein] [36] Homo sapiens myricetin myricetin results in decreased activity of HIF1A protein [37] Homo sapiens myricetin myricetin results in increased expression of HIF1A protein [38] Homo sapiens Palmitic Acid [Palmitic Acid co-treated with Oleic Acid co-treated with TNF protein] results in increased expression of HIF1A protein [39] Homo sapiens Quercetin Quercetin inhibits the reaction [HIF1A protein binds to VHL protein] [40] Homo sapiens Quercetin Quercetin results in decreased activity of HIF1A protein [41] Homo sapiens 2.8 分子对接结果
筛选得到核心蛋白TSHR(PDB:2xwt)、TP53(PDB:3q01)、MAPK1(PDB:4fv6)、HIF1A(PDB:1l8c)、CA9(PDB:6tl5)立体结构与42种化合物进行分子对接,仅展示对接分数第一的化合物与对应靶点对接模式图,结果发现Procyanidin b2、Daturic acid及Guanosine与核心蛋白对接良好,表明筛选化合物可能直接结合蛋白质进而改变其功能,见图9。进一步分析化合物与关键靶点的互作位置及相互作用方式,进而明确结合蛋白的关键位置,将得分最高化合物以多种构象化合物与蛋白对接,结果以热图呈现,见图10。发现TP53的GLU271、GLU285、ARG273、ARG249、SER249的氨基酸位点,HIF1A的ALA34、HIS18、VAL148位点以及CA9的A200位置可以与Procyanidin b2形成氢键,MAPK1的ARG351、ARG75与Daturic acid形成氢键及静电相互作用,TSHR的PHE141、ASP143、ILE117、TYR116与Guanosine形成氢键及碳氢键。结果表明上述蛋白氨基酸残基可能在与神秘果化合物结合过程中发挥重要作用。
3. 讨论与结论
本实验探究神秘果干预肿瘤免疫进而辅助治疗肿瘤的可能性,通过液相-质谱联用技术及生物信息数据挖掘神秘果治疗肿瘤的潜在机制。发现神秘果干预肿瘤免疫过程中可能影响的主要靶点如下:TSHR为促甲状腺激素受体,正常表达于甲状腺滤泡上皮细胞的细胞膜上,属于G蛋白偶联受体,其与TSH结合后被激活,进而激活cAMP-PKA信号通路,TSHR的缺失可能会促进甲状腺乳头状癌的增殖、侵袭能力[42]。HIF1为缺氧诱导因子-1,是细胞在缺氧状态下的关键调控蛋白,调节一系列细胞因子和生长介质的表达及蛋白质合成,对肿瘤血管的形成和对肿瘤细胞的增殖、转移、侵袭、凋亡、能量代谢等作用有着重要的影响[43],HIF-1的表达程度与肿瘤的恶性程度、新生血管的表达程度及预后不良呈正相关。抑制HIF-1的表达,阻断缺氧信号的传递,已经成为肿瘤治疗的新思路,而神秘果中有诸多成分,如杨梅素、槲皮素等均可影响靶点的表达。TP53是肿瘤研究中热点靶点,有超过 50%的肿瘤中存在TP53的基因突变,其中大多数都错义突变。突变型p53蛋白,不仅失去了野生型蛋白的肿瘤抑制功能,反而能促进肿瘤的发生和进展[44]。研究同时也发现神秘果含有抗肿瘤免疫成分42种,通过文献发现,部分的化合物对肿瘤免疫已有积极意义的报道。如槲皮素可以抑制淋巴细胞活化和增殖来发挥免疫调节作用。在对白血病细胞系的研究中,槲皮素对白血病细胞系的凋亡潜能高于外周血单核细胞,同时能够抑制T细胞增殖和活化等正常免疫功能[45],同时槲皮素可诱导多种肿瘤细胞凋亡,如肝癌细胞[46]、肺癌[47],槲皮素通过灭活caspase/Cyto-c途径、抑制AP-23/h TERT、抑制NF-κB/COX-2和阻断Akt/ERKl/2信号通路而发挥抗肿瘤作用[48]。苦杏仁苷作为一种天然产生的氰化物,具有不影响正常细胞的情况下发挥抗肿瘤的作用,可通过直接抑制肾移植大鼠的免疫细胞的增殖进而干预免疫功能[49],同样对影响膀胱癌细胞(UMUC-3、RT112、TCCSUP) 的附着和迁徙[50],通过cDNA 微列分析发现苦杏仁甙能明显下调细胞周期相发挥抗肿瘤作用[51]。原花青素作为中药中广泛存在的多酚类化合物具有良好的抗肿瘤作用,可能通过增加线粒体膜通透性和细胞色素C从线粒体中释放及激活肿瘤细胞内的细胞凋亡蛋白酶和半胱天冬酶诱导肿瘤细胞凋亡,可抑制多种肿瘤细胞的生长[52],例如抗黑素瘤[53]及前列腺癌[54]。
综上,神秘果作为干预肿瘤的一种辅助治疗选择,是非常有潜力的,神秘果含有较多活性成分可以改善肿瘤患者的免疫水平进而影响肿瘤的发生发展。但以上部分实验仍为预测,需要继续实验验证以进一步确认结果,希望神秘果在肿瘤及免疫方面得到出色发挥,为抗肿瘤提供更多的选择。
-
表 1 神秘果干预肿瘤免疫相关成分信息
Table 1 Information on relevant components of Synsepalum dulcificum 's intervention in tumor immunity
序号 化合物 结构 Smiles Inchikey CAS 1 Naringenin 
[C@H]1(CC(=O)c2c(cc(cc2O1)O)O)c1ccc(cc1)O FTVWIRXFELQLPI-ZDUSSCGKSA-N 480-41-1 2 1,3,5-Benzenetriol 
c1(cc(cc(c1)O)O)O QCDYQQDYXPDABM-UHFFFAOYSA-N 108-73-6 3 Anthranilic acid (Vitamin L1) 
c1(c(cccc1)N)C(=O)O RWZYAGGXGHYGMB-UHFFFAOYSA-N 118-92-3 4 Choline 
C(CO)[N](C)(C)C GDPPXFUBIJJIKR-UHFFFAOYSA-N 62-49-7 5 Quercetin 
c1(cc(c2c(c1)oc(c(c2=O)O)c1ccc(c(c1)O)O)O)O REFJWTPEDVJJIY-UHFFFAOYSA-N 117-39-5 6 Morin 
c12c(oc(c(c1=O)O)c1c(cc(cc1)O)O)cc(cc2O)O YXOLAZRVSSWPPT-UHFFFAOYSA-N 480-16-0 7 Phenylacetic acid 
c1cccc(c1)CC(=O)O WLJVXDMOQOGPHL-UHFFFAOYSA-N 103-82-2 8 Palmitic acid 
C(=O)(CCCCCCCCCCCCCCC)O IPCSVZSSVZVIGE-UHFFFAOYSA-N 57-10-3 9 Stearic acid 
C(CCCCCCCCCCC)CCCCCC(=O)O QIQXTHQIDYTFRH-UHFFFAOYSA-N 57-11-4 10 Guanosine 
c12n([C@H]3O[C@@H]([C@H]([C@H]
3O)O)CO)cnc1c(=O)[nH]c(n2)NNYHBQMYGNKIUIF-BDXYJKHTSA-N 118-00-3 11 Uracil 
[nH]1c(=O)[nH]c(=O)cc1 ISAKRJDGNUQOIC-UHFFFAOYSA-N 66-22-8 12 Dihydrouracil 
N1C(=O)CCNC1=O OIVLITBTBDPEFK-UHFFFAOYSA-N 504-07-4 13 Dopamine 
Oc1cc(CCN)ccc1O VYFYYTLLBUKUHU-UHFFFAOYSA-N 51-61-6 14 Heptadecanoic acid 
OC(=O)CCCCCCCCCCCCCCCC KEMQGTRYUADPNZ-UHFFFAOYSA-N 506-12-7 15 Tyramine 
Oc1ccc(CCN)cc1 DZGWFCGJZKJUFP-UHFFFAOYSA-N 51-67-2 16 Procyanidin B2 
c1(cc(c2c(c1)O[C@@H]([C@@H]([C@H]2c1c
(cc(c2c1O[C@@H]([C@@H](C2)O)c1cc
(c(cc1)O)O)O)O)O)c1cc(c(cc1)O)O)O)OXFZJEEAOWLFHDH-NFJBMHMQSA-N 29106-49-8 17 Pyridoxine 
Oc1c(c(CO)cnc1C)CO LXNHXLLTXMVWPM-UHFFFAOYSA-N 65-23-6 18 Adenine 
[nH]1cnc2c(N)ncnc12 GFFGJBXGBJISGV-UHFFFAOYSA-N 73-24-5 19 Gentisic acid 
c1(c(cc(cc1)O)C(=O)O)O WXTMDXOMEHJXQO-UHFFFAOYSA-N 490-79-9 20 4-Hydroxycinnamic acid 
C(=O)(/C=C/c1ccc(cc1)O)O NGSWKAQJJWESNS-ZZXKWVIFSA-N 501-98-4 21 Sucrose 
OC[C@H]1O[C@H](O[C@@]2(CO)[C@@H]
(O)[C@H](O)[C@@H](CO)O2)[C@H](O)
[C@@H](O)[C@@H]1OCZMRCDWAGMRECN-UGDNZRGBSA-N 57-50-1 22 myo-Inositol 
C1(C(C(C(C(C1O)O)O)O)O)O CDAISMWEOUEBRE-UHFFFAOYSA-N 87-89-8 23 Myristic acid 
C(=O)(CCCCCCCCCCCCC)O TUNFSRHWOTWDNC-UHFFFAOYSA-N 544-63-8 24 Amygdalin 
c1(ccccc1)[C@H](C#N)O[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@H](O1)CO[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@H](O1)CO XUCIJNAGGSZNQT-JHSLDZJXSA-N 29883-15-6 25 Myricetin 
c1(c(cc(cc1O)c1c(O)c(=O)c2c(o1)cc(cc2O)O)O)O IKMDFBPHZNJCSN-UHFFFAOYSA-N 529-44-2 26 Maslinic Acid 
[C@@]12([C@]3(C(=CC[C@@H]1[C@@]1
([C@@H](CC2)C([C@H]([C@@H](C1)O)O)(C)
C)C)[C@H]1[C@@](CC3)(CCC(C1)(C)C)
C(=O)O)C)CMDZKJHQSJHYOHJ-LLICELPBSA-N 4373-41-5 27 N6,N6,N6-Trimethyl-L-lysine 
C[N+](C)(C)CCCC[C@@H](C(=O)[O-])N MXNRLFUSFKVQSK-QMMMGPOBSA-N 19253-88-4 28 Nicotinamide 
c1(cccnc1)C(=O)N DFPAKSUCGFBDDF-UHFFFAOYSA-N 98-92-0 29 Betaine 
OC(=O)C[N](C)(C)C GMTCLSZGPOBNLA-UHFFFAOYSA-N 107-43-7 30 Quercitrin 
c1(c(c(=O)c2c(o1)cc(cc2O)O)O[C@H]1[C@@H]([C@@H]
([C@H]([C@@H](O1)C)O)O)O)
c1cc(c(cc1)O)OOXGUCUVFOIWWQJ-HQBVPOQASA-N 522-12-3 31 L-Asparagine 
C(=O)([C@@H](N)CC(=O)N)O DCXYFEDJOCDNAF-REOHCLBHSA-N 32640-57-6 32 L-Tryptophan 
c12c(C[C@@H](C(=O)O)N)c[nH]c1cccc2 QIVBCDIJIAJPQS-VIFPVBQESA-N 73-22-3 33 L-Threonine 
O[C@@H]([C@H](N)C(=O)O)C AYFVYJQAPQTCCC-GBXIJSLDSA-N 72-19-5 34 Cytosine 
n1c(cc[nH]c1=O)N OPTASPLRGRRNAP-UHFFFAOYSA-N 71-30-7 35 L-Aspartate 
C(=O)([C@@H](N)CC(=O)O)O CKLJMWTZIZZHCS-REOHCLBHSA-N 56-84-8 36 Indole 
[nH]1c2c(cc1)cccc2 SIKJAQJRHWYJAI-UHFFFAOYSA-N 120-72-9 37 Astragalin 
c12c(oc(c(c1=O)O[C@@H]1O[C@@H]([C@H]([C@@H]([C@H]1O)O)O)CO)c1ccc(cc1)O)cc(cc2O)O JPUKWEQWGBDDQB-QSOFNFLRSA-N 480-10-4 38 2-Hydroxyadenine 
c12c(c(nc(n1)O)N)[nH]cn2 DRAVOWXCEBXPTN-UHFFFAOYSA-N 3373-53-3 39 Glutathione 
C(CC(=O)N[C@@H](CS)C(=O)NCC(=O)O)[C@@H](C(=O)O)N RWSXRVCMGQZWBV-WDSKDSINSA-N 70-18-8 40 trans-Ferulic acid 
c1(cc(c(cc1)O)OC)/C=C/C(=O)O KSEBMYQBYZTDHS-HWKANZROSA-N 1135-24-6 41 Quercetin 3'-methyl ether 
c1(c(OC)c(=O)c2c(O)cc(O)cc2o1)c1cc(O)c(O)cc1 WEPBGSIAWZTEJR-UHFFFAOYSA-N 1486-70-0 42 4-Guanidinobutyric acid 
C(=O)(CCCNC(=N)N)O TUHVEAJXIMEOSA-UHFFFAOYSA-N 463-00-3 表 2 神秘果影响肿瘤免疫相关化合物影响信号通路
Table 2 Synsepalum dulcificum affects tumor immunity-related compounds affect signal pathways
序号 通路 通路 ID P-value 富集基因数 基因组占比 1 Immune System REACT:R-HSA-168256 0 1391 2118/44020 genes: 4.81% 2 Innate Immune System REACT:R-HSA-168249 0 858 1298/44020 genes: 2.95% 3 Metabolic pathways KEGG:hsa01100 0 890 1270/44020 genes: 2.89% 4 Metabolism REACT:R-HSA-1430728 0 1536 2172/44020 genes: 4.93% 5 Metabolism of proteins REACT:R-HSA-392499 0 984 1623/44020 genes: 3.69% 6 Signal Transduction REACT:R-HSA-162582 0 1550 2588/44020 genes: 5.88% 7 Gene Expression REACT:R-HSA-74160 2.16e-310 969 1831/44020 genes: 4.16% 8 Metabolism of lipids and lipoproteins REACT:R-HSA-556833 6.33e -297 595 814/44020 genes: 1.85% 9 Developmental Biology REACT:R-HSA-1266738 1.94e -270 672 1076/44020 genes: 2.44% 10 Cytokine Signaling in Immune system REACT:R-HSA-1280215 9.13e -267 547 760/44020 genes: 1.73% 表 3 神秘果化合物对核心基因的影响
Table 3 Effects of Synsepalum dulcificum compounds on hub genes
核心基因 化合物 影响趋势 支持文献 实验物种 TP53 Choline Choline results in decreased expression of TP53 mRNA [13] Rattus norvegicus Dopamine [Dopamine results in increased expression of HIF1A protein] which results in increased expression of and results in increased phosphorylation of TP53 protein [14] Homo sapiens Glutathione [Buthionine Sulfoximine results in decreased abundance of Glutathione] inhibits the reaction [Benzo(a)pyrene results in increased activity of TP53 protein] [15] Homo sapiens Glutathione Dithiothreitol inhibits the reaction [Glutathione results in increased glutathionylation of TP53 protein] [16] Homo sapiens Glutathione Glutathione inhibits the reaction [[CD40LG protein co-treated with IL4 protein] results in increased expression of TP53 protein] [17] Homo sapiens morin morin inhibits the reaction [Ifosfamide results in increased expression of TP53 protein] [18] Rattus norvegicus morin morin results in increased expression of TP53 mRNA [19] Homo sapiens myricetin myricetin analog results in increased expression of TP53 protein [20] Homo sapiens myricitrin myricitrin inhibits the reaction [Hydrogen Peroxide results in increased expression of TP53 protein] [21] Homo sapiens naringenin naringenin results in increased expression of TP53 mRNA [22] Homo sapiens Quercetin Quercetin results in decreased expression of TP53 protein [23] Homo sapiens TSHR Quercetin Quercetin results in decreased expression of TSHR mRNA [24] Rattus norvegicus MAPK1 Dopamine Dopamine results in increased phosphorylation of and results in increased activity of MAPK1 protein [25] Homo sapiens Glutathione Glutathione inhibits the reaction [Hydrogen Peroxide results in increased phosphorylation of MAPK1 protein] [26] Canis lupus familiaris morin morin inhibits the reaction [Excitatory Amino Acid Agonists results in increased phosphorylation of MAPK1 protein] [27] Rattus norvegicus myricetin myricetin results in decreased activity of MAPK1 protein [28] Mus musculus naringenin naringenin results in decreased activity of MAPK1 protein [29] Homo sapiens naringenin naringenin results in decreased phosphorylation of MAPK1 protein [30] Homo sapiens Palmitic Acid Palmitic Acid results in increased phosphorylation of MAPK1 protein [31] Homo sapiens Sucrose Sucrose results in decreased phosphorylation of and results in decreased activity of MAPK1 protein [32] Mus musculus Tyramine Tyramine results in increased activity of MAPK1 protein [33] Oryctolagus cuniculus HIF1A Choline [Methionine deficiency co-treated with Choline deficiency co-treated with Folic Acid deficiency] results in increased methylation of HIF1A gene [34] Mus musculus Dopamine Dopamine results in decreased expression of HIF1A protein [35] Rattus norvegicus Dopamine Dopamine results in increased expression of HIF1A protein [14] Homo sapiens Glutathione Glutathione inhibits the reaction [15-deoxy-delta(12,14)-prostaglandin J2 results in increased expression of HIF1A protein] [36] Homo sapiens myricetin myricetin results in decreased activity of HIF1A protein [37] Homo sapiens myricetin myricetin results in increased expression of HIF1A protein [38] Homo sapiens Palmitic Acid [Palmitic Acid co-treated with Oleic Acid co-treated with TNF protein] results in increased expression of HIF1A protein [39] Homo sapiens Quercetin Quercetin inhibits the reaction [HIF1A protein binds to VHL protein] [40] Homo sapiens Quercetin Quercetin results in decreased activity of HIF1A protein [41] Homo sapiens -
[1] BRAY F, FERLAY J, SOERJOMATARAM I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin,2018,68(6):394−424. doi: 10.3322/caac.21492
[2] ARNOLD M, LEITZMANN M, FREISLING H, et al. Obesity and cancer: An update of the global impact[J]. Cancer Epidemiol,2016,41:8−15. doi: 10.1016/j.canep.2016.01.003
[3] LING C Q, YUE X Q, LING C. Three advantages of using traditional Chinese medicine to prevent and treat tumor[J]. Integr Med, 2014 , 12(4): 331−335.
[4] 陈琪, 王金祥, 贾颖, 等. 神秘果化学成分和生物活性研究进展[J]. 中国中医药现代远程教育,2018,16(10):149−152. [CHEN Qi, WANG Jinxiang, JIA Ying, et al. Resreach progress on the chenmical constituents and bioactivity ofSynsepalum dulcificum Daniell[J]. Chinese Medicine Modern Distance Education of China,2018,16(10):149−152. doi: 10.3969/j.issn.1672-2779.2018.10.064 [5] NTIE K F, LIFONGO L L, MBAZE L M, et al. Cameroonian medicinal plants: A bioactivity versus ethnobotanical survey and chemotaxonomic classification[J]. BMC Complement Altern Med,2013,26:147.
[6] 李玲, 汪哲, 谭宁华. 天然产物靶向肿瘤微环境的研究进展[J]. 药学学报,2021,56(6):1580−1590. [LI Ling, WANG Zhe, TAN Ninghua. Advances in natural products that target the tumor microenvironment[J]. Acta Pharmaceutica Sinica,2021,56(6):1580−1590. [7] WANG H M, CHOU Y T, HONG Z L, et al. Bioconstituents from stems of Synsepalum dulcificum Daniell (Sapotaceae) inhibit human melanoma proliferation, reduce mushroom tyrosinase activity and have antioxidant properties[J]. Taiwan Inst Chem Eng,2011,42:204−211. doi: 10.1016/j.jtice.2010.05.008
[8] SEONG J, OYONG G, CABRERA E. Synsepalum dulcificum extracts exhibit cytotoxic activity on human colorectal cancer cells and upregulate c-fos and c-jun early apoptotic gene expression[J]. Asian Pac Trop Biomed,2018,8:173−178. doi: 10.4103/2221-1691.227999
[9] LIU Z, CAI C, DU J, et al. TCMIO: A comprehensive database of traditional chinese medicine on immuno-oncology[J]. Front Pharmacol,2020,11:439.
[10] YU G, WANG L G, HAN Y, et al. ClusterProfiler: An R package for comparing biological themes among gene clusters[J]. OMICS,2012,16(5):284−287. doi: 10.1089/omi.2011.0118
[11] TANG Z, LI C, KANG B, et al. GEPIA: A web server for cancer and normal gene expression profiling and interactive analyses[J]. Nucleic Acids Res,2017,45(W1):W98−W102.
[12] DAVIS A P, WIEGERS T C, GRONDIN C J, et al. Leveraging the comparative toxicogenomics database to fill in knowledge gaps for environmental health: A test case for air pollution-induced cardiovascular disease[J]. Toxicol Sci,2020,177(2):392−404. doi: 10.1093/toxsci/kfaa113
[13] KAMENDULIS L M, KLAUNIG J E. Species differences in the induction of hepatocellular DNA synthesis by diethanolamine[J]. Toxicol Sci,2005,87(2):328−336. doi: 10.1093/toxsci/kfi252
[14] GIMÉNEZ-XAVIER P, FRANCISCO R, SANTIDRIÁN A F, et al. Effects of dopamine on LC3-II activation as a marker of autophagy in a neuroblastoma cell mode[J]. Neurotoxicology,2009,30(4):658−665. doi: 10.1016/j.neuro.2009.04.007
[15] BISWAL S, MAXWELL T, RANGASAMY T, et al Modulation of benzo[a]pyrene-induced p53 DNA activity by acrolein[J]. Carcinogenesis, 2003, 24(8): 1401−1406.
[16] VELU C S, NITURE S K, DONEANU C E, et al. Human p53 is inhibited by glutathionylation of cysteines present in the proximal DNA-binding domain during oxidative stress[J]. Biochemistry,2007,46(26):7765−7780.
[17] SAMUEL J, JAYNE S, CHEN Y, et al. Posttranscriptional upregulation of p53 by reactive oxygen species in chronic lymphocytic leukemia[J]. Cancer Res, 2016, 76(21): 6311−6319.
[18] ÇELIK H, KUCUKLER S, ÇOMAKLI S, et al. Morin attenuates ifosfamide-induced neurotoxicity in rats via suppression of oxidative stress, neuroinflammation and neuronal apoptosis[J]. Neurotoxicology,2020,76:126−137. doi: 10.1016/j.neuro.2019.11.004
[19] ZHANG Q, ZHANG F, THAKUR K, et al. Molecular mechanism of anti-cancerous potential of Morin extracted from mulberry in Hela cells[J]. Food Chem Toxicol,2018,112:466−475. doi: 10.1016/j.fct.2017.07.002
[20] AKHTAR S, NAJAFZADEH M, ISREB M, et al An in vitro investigation into the protective and genotoxic effects of myricetin bulk and nano forms in lymphocytes of MGUS patients and healthy individuals[J]. Toxicol Lett, 2020,327: 33−40.
[21] SUN G B, QIN M, YE J X, et al. Inhibitory effects of myricitrin on oxidative stress-induced endothelial damage and early atherosclerosis in ApoE-/- mice[J]. Toxicol Appl Pharmacol,2013,271(1):114−126.
[22] XU Z, HUANG B, LIU J, et al. Combinatorial anti-proliferative effects of tamoxifen and naringenin: The role of four estrogen receptor subtypes[J]. Toxicology,2018,410:231−246.
[23] WANG H, YUAN Z, CHEN Z, et al. Effect of quercetin on glioma cell U87 apoptosis and feedback regulation of MDM2-p53[J]. 2014, 34(5): 686−689.
[24] GIULIANI C, BUCCI I, DI SANTO S, et al. The flavonoid quercetin inhibits thyroid-restricted genes expression and thyroid function[J]. Food Chem Toxicol,2014,66:23−29. doi: 10.1016/j.fct.2014.01.016
[25] ANGER T, GREBE N, OSINSKI D, et al. Role of endogenous RGS proteins on endothelial ERK 1/2 activation[J]. Exp Mol Pathol,2008,85(3):165−173. doi: 10.1016/j.yexmp.2008.09.005
[26] IKARI A, ATOMI K, YAMAZAKI Y, et al. Hyperosmolarity-induced up-regulation of claudin-4 mediated by NADPH oxidase-dependent H2O2 production and Sp1/c-Jun cooperation[J]. Biochim Biophys Acta,2013,1833(12):2617−2627. doi: 10.1016/j.bbamcr.2013.06.016
[27] CAMPOS-ESPARZA M R, SÁNCHEZ-GÓMEZ M V, MATUTE C. Molecular mechanisms of neuroprotection by two natural antioxidant polyphenols[J]. Cell Calcium,2009,45(4):358−368. doi: 10.1016/j.ceca.2008.12.007
[28] LEE K W, KANG N J, ROGOZIN E A, et al. Myricetin is a novel natural inhibitor of neoplastic cell transformation and MEK1[J]. Carcinogenesis,2007,28(9):1918−1927. doi: 10.1093/carcin/bgm110
[29] CHEN Y Y, CHANG Y M, WANG K Y, et al. Naringenin inhibited migration and invasion of glioblastoma cells through multiple mechanisms[J]. Environ Toxicol,2019,34(3):233−239. doi: 10.1002/tox.22677
[30] KIM B, JUNG N, LEE S, et al. Apigenin inhibits cancer stem cell-like phenotypes in human glioblastoma cells via suppression of c-met signaling[J]. Phytother Res,2016,30(11):1833−1840. doi: 10.1002/ptr.5689
[31] LI J, BI L, HULKE M, et al. Fish oil and fenofibrate prevented phosphorylation-dependent hepatic sortilin 1 degradation in Western diet-fed mice[J]. Biol Chem, 2014, 289(32): 22437−22449.
[32] FRIIS M B, FRIBORG C R, SCHNEIDER L, et al. Cell shrinkage as a signal to apoptosis in NIH 3T3 fibroblasts [J]. Physiol, 2005, 567(Pt 2): 427−443.
[33] COATRIEUX C, SANSON M, NEGRE-SALVAYRE A, et al. MAO-A-induced mitogenic signaling is mediated by reactive oxygen species, MMP-2, and the sphingolipid pathway[J]. Free Radic Biol Med,2007,43(1):80−89.
[34] TRYNDYAK V P, HAN T, MUSKHELISHVILI L, et al. Coupling global methylation and gene expression profiles reveal key pathophysiological events in liver injury induced by a methyl-deficient diet[J]. Mol Nutr Food Res,2011,55(3):411−418. doi: 10.1002/mnfr.201000300
[35] QIN T, WANG C, CHEN X, et al. Dopamine induces growth inhibition and vascular normalization through reprogramming M2-polarized macrophages in rat C6 glioma[J]. Toxicol Appl Pharmacol,2015,286(2):112−123.
[36] OLMOS G, CONDE I, ARENAS I, et al. Accumulation of hypoxia-inducible factor-1alpha through a novel electrophilic, thiol antioxidant-sensitive mechanism[J]. Cell Signal,2007,19(10):2098−2105. doi: 10.1016/j.cellsig.2007.06.004
[37] BACH A, BENDER-SIGEL J, SCHRENK D, et al. The antioxidant quercetin inhibits cellular proliferation via HIF-1-dependent induction of p21WAF[J]. Antioxid Redox Signal,2010,13(4):437−448.
[38] HONG K S, PARK J I, KIM M J, et al. Involvement of SIRT1 in hypoxic down-regulation of c-Myc andβ-catenin and hypoxic preconditioning effect of polyphenols[J]. Toxicol Appl Pharmacol,2012,259(2):210−218.
[39] BREHER-ESCH S, SAHINI N, TRINCONE A, et al. Genomics of lipid-laden human hepatocyte cultures enables drug target screening for the treatment of non-alcoholic fatty liver disease[J]. BMC Med Genomics,2018,11(1):111.
[40] PARK S S, BAE I, LEE Y J. Flavonoids-induced accumulation of hypoxia-inducible factor (HIF)-1alpha/2alpha is mediated through chelation of iron [J]. Cell Biochem, 2008,103(6): 1989−1998.
[41] OH S J, KIM O, LEE J S, et al. Inhibition of angiogenesis by quercetin in tamoxifen-resistant breast cancer cells[J]. Food Chem Toxicol,2010,48(11):3227−3234. doi: 10.1016/j.fct.2010.08.028
[42] SOARES H P, CUSNIR M, SCHWARTZ M A, et al. Treatment of taste alterations in chemotherapy patients using the “miracle fruit”: Preliminary analysis of a pilot study[J]. Journal of Clinical Oncology,2010,28(Suppl.15):19523.
[43] KARTAL K, ONDER S, KOSEMEHMETOGLU K, et al Methylation status of TSHr in well-differentiated thyroid cancer by using cytologic material[J]. BMC Cancer, 2015,15: 824.
[44] ROUAULT-PIERRE K, HAMILTON A, BONNET D. Effect of hypoxia-inducible factors in normal and leukemic stem cell regulation and their potential therapeutic impact[J]. Expert Opin Biol Ther,2016,16(4):463−476. doi: 10.1517/14712598.2016.1133582
[45] 许杰, 郝牧. p53蛋白异常与肿瘤发生关系的研究进展[J]. 肿瘤研究与临床,2019,31(9):633−636. [(XU Jie, HAO Mu. Research progress on the relationship between p53 protein abnormality and tumorigenesis[J]. Cancer Research and Clinic,2019,31(9):633−636. [46] LUGLI E, FERRARESI R, ROAT E, et al. Quercetin inhibits lymphocyte activation and proliferation without inducing apoptosis in peripheral mononuclear cells[J]. Leuk Res,2009,33(1):140−150. doi: 10.1016/j.leukres.2008.07.025
[47] BRITO A F, RIBEIRO M, ABRANTES A M, et al. New approach for treatment of primary liver tumors: The role of quercetin[J]. Nutr Cancer, 2016, 68(2): 250−266.
[48] KLIMASZEWSKA-WIŚNIEWSKA A, HAŁAS-WIŚNIEWSKA M, IZDEBSKA M, et al. Antiproliferative and antimetastatic action of quercetin on A549 non-small cell lung cancer cells through its effect on the cytoskeleton[J]. Acta Histochem, 2017, 119(2): 99−112.
[49] 刘晟文, 刘建英. 槲皮素药理学作用的研究进展[J]. 中华肺部疾病杂志(电子版), 2020, 13(1): 1041−06 LIU Shengwen, LIU Jianying. Research progress on pharmacological effects of quercetin[J]. Chinese Journal of Lung Diseases(Electronic Edition), 2020, 13(1): 104−106.
[50] 郭君其, 王灵杰, 叶永峰, 等. 苦杏仁甙对肾脏移植大鼠存活情况的影响[J]. 中国中西医结合肾病杂志,2008,9(1):22−24. [GUO Junqi, WANG Linghuo, YE Yongfeng, et al. Effect of amygdalin on the survival of kidney transplanted rats[J]. Chinese Journal of Integrated Traditional and Western Nephrology,2008,9(1):22−24. doi: 10.3969/j.issn.1009-587X.2008.01.007 [51] MAKAREVIĆ J, RUTZ J, JUENGEL E, et al. Amygdalin influences bladder cancer cell adhesion and invasion in vitro[J]. PLoS One, 2014, 9(10): e110244.
[52] PARK H J, YOON S H, HAN L S, et al. Amygdalin inhibits genes related to cell cycle in SNU-C4 human colon cancer cells[J]. World Gastroenterol, 2005,11(33): 5156−5161.
[53] MIURA T, CHIBA M, KASAI K, et al. Apple procyanidins induce tumor cell apoptosis through mitochondrial pathway activation of caspase-3[J]. Carcinogenesis,2008,29(3):585−593.
[54] 周密, 段玉清, 王文兵, 等. 莲原花青素抗黑色素瘤的研究[J]. 食品与科学, 2009, 30(11): 223−226. ZHOU Mi, DUAN Yuqing, WANG Wenbing, et al. Study on the anti-melanoma effect of lotus proanthocyanidins[J]. Food Science, 2009, 30(11): 223−226.
-
期刊类型引用(6)
1. 李思涵,倪庆圆,耿相玉,李秀凉. 纳豆抗氧化肽对H_2O_2诱导HEK293细胞氧化应激损伤的保护作用. 食品安全质量检测学报. 2024(06): 161-169 .  百度学术
百度学术
2. 廖彭莹,张天丰,邓纭宁,庞云娟,孙雪芹,樊文研. 鳖各部位的成分和药理作用研究进展. 中药材. 2024(04): 1053-1061 .  百度学术
百度学术
3. 黄可承,包建强. 中华鳖营养成分分析与功效研究进展. 上海农业学报. 2023(01): 145-152 .  百度学术
百度学术
4. 屈美华,张彦龙,李元敬,曾伟民,孟鑫,雷虹. 蓝靛果原浆对果蝇寿命及其体内抗氧化活性的影响. 黑龙江大学自然科学学报. 2023(06): 713-720 .  百度学术
百度学术
5. 吴俊豪,王晶,KHO SETHYKUN,覃川,王倩倩,余鹏,冯凤琴. 龟肉蛋白肽延缓果蝇衰老的作用及机制研究. 食品工业科技. 2022(17): 394-401 .  本站查看
本站查看
6. 刘睿,武文星,朱悦,郭盛,赵明,曹鹏,段金廒. 动物药现代研究方法学进展与展望. 南京中医药大学学报. 2022(10): 857-869 .  百度学术
百度学术
其他类型引用(7)






 下载:
下载:






 下载:
下载:



